MHC Structure and Genetics
Major Histocompatibility Complex (MHC) molecules sit at the center of adaptive immunity. Their job is to bind peptide fragments and display them on the cell surface so T cells can inspect what's happening inside and around cells. Without MHC molecules, T cells would have no way to "see" antigens, and adaptive immune responses couldn't get started.
MHC genes are among the most polymorphic in the entire genome, which makes sense: the more peptide-binding variants a population carries, the harder it is for any single pathogen to evade detection by everyone. MHC molecules fall into two main classes, and the distinction between them shapes nearly everything about how antigens get processed and which T cells respond.
Structure and Function of MHC Molecules
MHC Class I
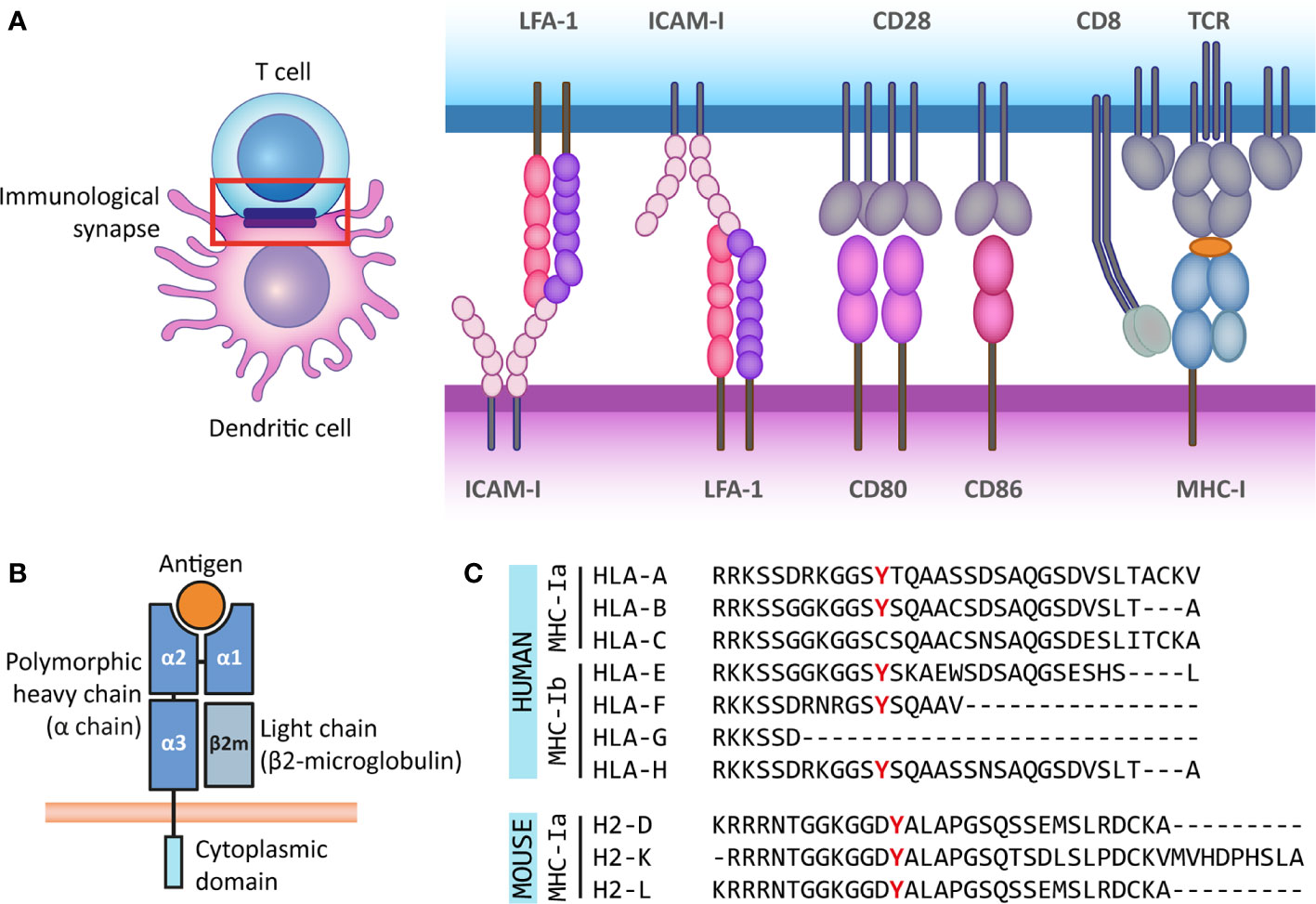
MHC class I molecules are built from a transmembrane heavy chain (α chain) with three extracellular domains (α1, α2, α3) plus a smaller protein called β2-microglobulin (β2m) that associates non-covalently. β2m doesn't span the membrane itself; it stabilizes the overall fold and is required for proper surface expression.
The peptide-binding groove is formed by the α1 and α2 domains. This groove is closed at both ends, which constrains the peptides it can hold to roughly 8–10 amino acids in length. The α3 domain contains the binding site for the CD8 co-receptor.
Functionally, MHC class I presents peptides derived from intracellular proteins. That includes normal self-proteins (which T cells learn to ignore during thymic selection), but also viral proteins and abnormal proteins from cancerous cells. CD8+ cytotoxic T cells scan these complexes and kill cells displaying foreign or aberrant peptides.
MHC Class II
MHC class II molecules consist of two transmembrane chains, α and β, each contributing two extracellular domains. The peptide-binding groove is formed by the α1 and β1 domains. Unlike class I, this groove is open at both ends, so it accommodates longer peptides, typically 13–25 amino acids.
MHC class II presents peptides from extracellular proteins that have been internalized by endocytosis or phagocytosis. These complexes are recognized by CD4+ helper T cells, which then coordinate downstream responses: activating B cells for antibody production, licensing macrophages, or directing the type of immune response (Th1, Th2, Th17).
Quick comparison: Class I = intracellular peptides → CD8+ T cells. Class II = extracellular peptides → CD4+ T cells. This division of labor is a recurring theme throughout antigen processing and presentation.

Genetic Organization of MHC Loci
The MHC region in humans is called HLA (Human Leukocyte Antigen) and spans roughly 4 Mbp on chromosome 6p21. In mice, the equivalent region (called H-2) sits on chromosome 17 and shows conserved synteny with the human locus.
Classical MHC genes:
- Class I: HLA-A, HLA-B, HLA-C — each encodes a class I heavy chain
- Class II: HLA-DP, HLA-DQ, HLA-DR — each locus encodes both an α chain and a β chain gene (though the α chains of some loci, like DR, are essentially monomorphic)
Three genetic features make MHC diversity extraordinary:
-
Polygeny — Multiple genes encode class I and class II molecules, so each person expresses several different MHC molecules simultaneously. This broadens the range of peptides that can be presented.
-
Codominant expression — Both maternal and paternal alleles are expressed. A heterozygous individual at all three class I loci expresses six different class I molecules (two each of HLA-A, -B, and -C). This is a real advantage: heterozygotes can present a wider array of pathogen-derived peptides than homozygotes.
-
Extreme polymorphism — Some HLA loci have thousands of alleles in the human population. HLA-B alone has over 3,000 known variants. Most of this variation clusters in the residues lining the peptide-binding groove, directly affecting which peptides each allele can bind.
How does this diversity arise? Three main mechanisms contribute:
- Point mutations — Single nucleotide changes that alter groove residues
- Gene conversion — Short stretches of sequence are copied from one MHC gene to another, creating mosaic alleles
- Recombination — Shuffling of alleles at different loci during meiosis generates new haplotype combinations
MHC Function and Expression
MHC in Antigen Presentation
The pathway from intact protein to MHC-displayed peptide differs for the two classes. Here's a simplified overview (the full processing pathways are covered in later sections of this unit):
Class I pathway (cytosolic/endogenous):
- Intracellular proteins (self-proteins, viral proteins, defective ribosomal products) are degraded by the proteasome in the cytosol.
- Resulting peptides are transported into the endoplasmic reticulum (ER) by the TAP transporter (Transporter associated with Antigen Processing).
- In the ER, peptides are loaded onto MHC class I molecules with the help of chaperones including tapasin, which bridges TAP to the class I molecule and optimizes peptide selection.
- Stable peptide–MHC I complexes travel to the cell surface.
Class II pathway (endosomal/exogenous):
- Extracellular material (bacteria, soluble proteins, allergens) is internalized by endocytosis or phagocytosis.
- Proteins are degraded by acidic proteases in endosomes and lysosomes.
- MHC class II molecules are synthesized in the ER with their groove blocked by the invariant chain (Ii), which prevents premature binding of ER-resident peptides. After Ii is degraded in the endosomal compartment, a small fragment called CLIP remains in the groove.
- HLA-DM catalyzes the exchange of CLIP for antigenic peptides. HLA-DO acts as a negative regulator of HLA-DM in certain cell types (particularly B cells), adding another layer of control.
- Loaded MHC II complexes are transported to the cell surface.
T cell recognition requires the T cell receptor (TCR) to contact both the peptide and the MHC molecule simultaneously. The co-receptor (CD8 for class I, CD4 for class II) binds a conserved region on the MHC molecule, stabilizing the interaction and boosting intracellular signaling. This three-way contact (TCR + peptide-MHC + co-receptor) forms the core of the immunological synapse.
Once activated, T cells undergo clonal expansion and differentiate into effector cells. CD8+ T cells become cytotoxic lymphocytes (CTLs). CD4+ T cells differentiate into helper subsets (Th1, Th2, Th17, Tfh, etc.) depending on the cytokine environment, and these subsets direct different arms of the immune response.
Expression Patterns of MHC Classes
MHC class I is expressed on nearly all nucleated cells. Red blood cells lack it (they lack a nucleus), and a few other cell types express very low levels. Lymphocytes and macrophages express particularly high levels. This broad expression makes sense: any nucleated cell can become virally infected, so all of them need to be visible to CD8+ T cells.
MHC class II has a much more restricted distribution, found primarily on professional antigen-presenting cells (APCs):
- Dendritic cells — The most potent APCs. They're uniquely capable of activating naïve T cells and initiating primary immune responses.
- Macrophages — Present peptides from material they've phagocytosed. Particularly important for sustaining responses against intracellular bacteria.
- B cells — Capture specific antigens via their surface immunoglobulin (BCR), internalize and process them, then present peptides on MHC II to receive help from CD4+ T cells.
- Thymic epithelial cells — Express MHC II for positive and negative selection of developing T cells.
- Activated T cells (in humans, not mice) — Can upregulate MHC II, potentially presenting antigens to other T cells.
Regulation of expression:
Some MHC expression is constitutive (always on), but levels can be dynamically adjusted. The cytokine IFN-γ is a potent upregulator of both class I and class II expression. During an infection, IFN-γ released by activated T cells and NK cells increases MHC levels on surrounding cells, amplifying antigen presentation right where it's needed.
Altered expression in disease is clinically significant:
- Many viruses actively downregulate MHC class I to hide infected cells from CTLs. Examples include certain herpesvirus and HIV proteins that interfere with TAP function or redirect MHC molecules for degradation.
- Tumors frequently lose or downregulate MHC expression as an immune evasion strategy. This is one reason why NK cells are important: they're activated by the absence of MHC class I (the "missing self" hypothesis), providing a backup surveillance mechanism.