The harmonic oscillator model describes how atoms vibrate in crystals and provides the foundation for understanding phonons, heat capacity, and thermal transport in solids. By assuming small displacements and linear restoring forces, this deceptively simple framework captures a surprising amount of real physics. Quantum mechanics then extends it, revealing quantized energy levels and zero-point energy that directly shape material behavior at low temperatures.
Harmonic oscillator basics
Atoms in a crystal sit at equilibrium positions, and when displaced slightly, they experience a restoring force that pulls them back. The harmonic oscillator model treats each atom as if it's connected to its neighbors by ideal springs. This works well as long as displacements stay small enough that the restoring force remains proportional to displacement.
Potential energy function
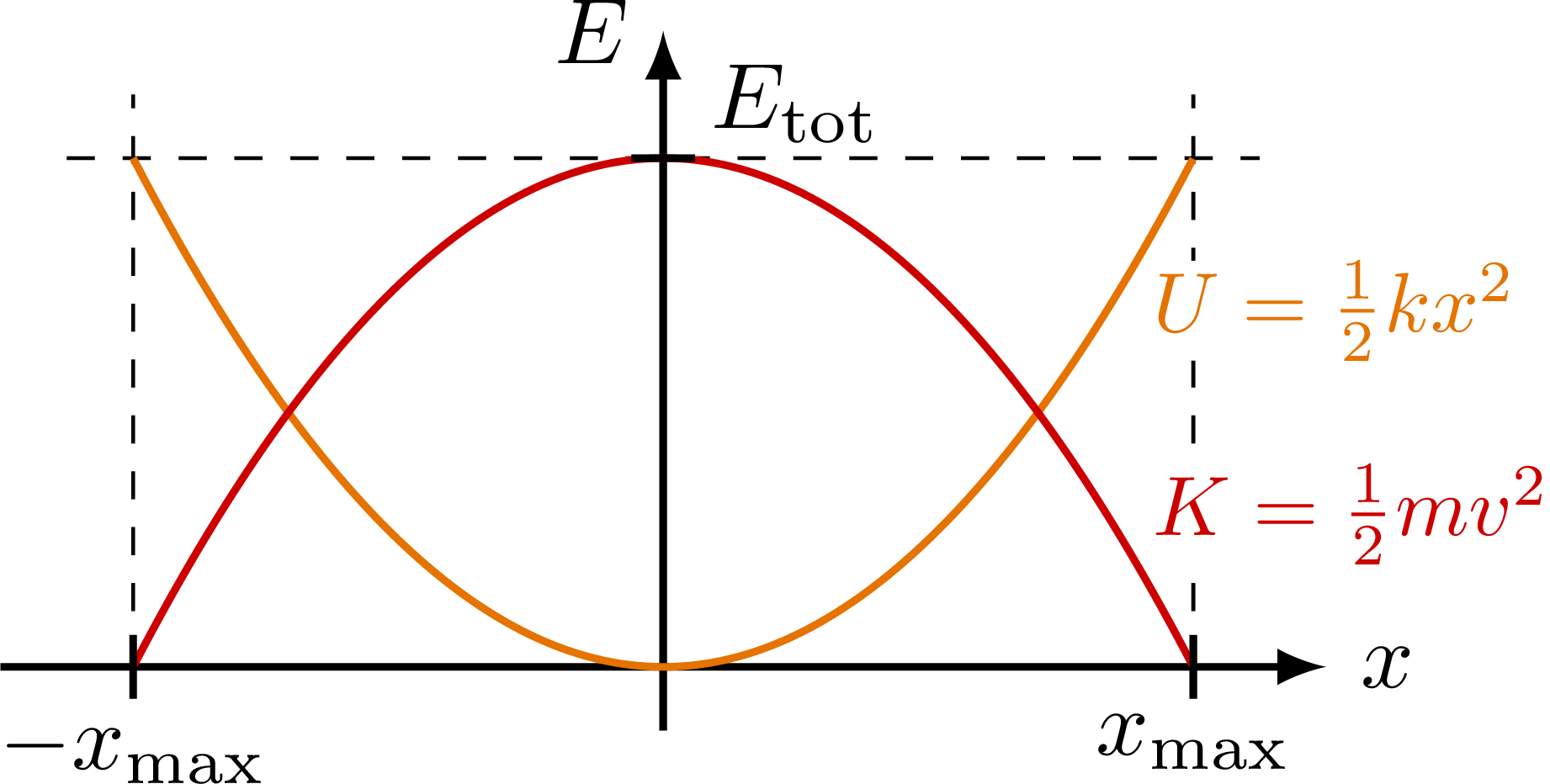
The potential energy of a harmonic oscillator depends on how far it's displaced from equilibrium:
where is the spring constant and is the displacement. This is a parabola centered at the equilibrium position, symmetric on both sides. The curvature of this parabola (set by ) determines how stiff the bond is and ultimately controls the oscillation frequency.
In real crystals, the true interatomic potential is only approximately parabolic near the minimum. The harmonic model works because we're Taylor-expanding the real potential around equilibrium and keeping only the quadratic term.
Hooke's law
The restoring force follows directly from the potential energy:
- is the spring constant (units of N/m), measuring bond stiffness
- is the displacement from equilibrium
- The negative sign means the force always points back toward equilibrium
This linear relationship is only valid for small displacements. Push an atom too far and higher-order terms in the potential become significant, which is where anharmonic effects enter.
Restoring force
The restoring force is what drives oscillatory motion. Because it always points toward equilibrium and grows linearly with displacement, the atom overshoots equilibrium, gets pulled back, overshoots again, and so on. This produces sinusoidal motion at a natural frequency , where is the atomic mass.
Stiffer bonds (larger ) and lighter atoms (smaller ) give higher vibrational frequencies. This relationship shows up repeatedly when analyzing phonon dispersion.
Solving the harmonic oscillator
Three standard formulations from classical mechanics can solve the harmonic oscillator. Each gives the same physics but offers different advantages, especially when transitioning to quantum mechanics or handling many coupled oscillators.
Newtonian mechanics approach
Apply Newton's second law directly:
This rearranges to:
The general solution is:
where is the amplitude and is the phase. This approach is the most intuitive: you can directly read off the position and velocity as functions of time.
Lagrangian formulation
The Lagrangian is defined as kinetic minus potential energy:
Applying the Euler-Lagrange equation:
yields the same equation of motion as the Newtonian approach. The Lagrangian formulation becomes especially useful when you're dealing with many coupled oscillators or generalized coordinates, which is exactly the situation in a crystal lattice with many atoms.
Hamiltonian formulation
The Hamiltonian is the total energy expressed in terms of position and conjugate momentum :
Hamilton's equations of motion are:
This formulation is essential because it maps directly onto the quantum mechanical treatment. When you replace and with operators and , you get the quantum harmonic oscillator Hamiltonian.
Quantum harmonic oscillator
Treating the harmonic oscillator quantum mechanically reveals that vibrational energy is quantized into discrete levels. This is the conceptual origin of phonons: each quantum of vibrational energy in a lattice mode behaves like a particle.
Schrödinger equation
The time-independent Schrödinger equation for the harmonic oscillator is:
Solving this (typically through a power series method or the operator approach below) gives quantized energy levels:
The energy levels are evenly spaced by . This uniform spacing is a signature feature of the harmonic potential and has direct consequences for phonon statistics.
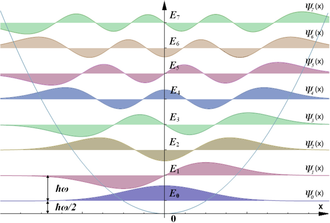
Eigenvalues and eigenfunctions
- Eigenvalues: , where
- Eigenfunctions:
Here are the Hermite polynomials and is a normalization constant. The Gaussian envelope ensures the wavefunction decays at large displacements, while the Hermite polynomial determines the number of nodes ( nodes for the -th state).
The probability density tells you where the atom is most likely to be found. For the ground state (), the atom is most likely at equilibrium. For higher states, the probability spreads out and develops nodes.
Zero-point energy
Even in the ground state (), the oscillator has energy:
This zero-point energy is a direct consequence of the Heisenberg uncertainty principle. You can't simultaneously pin down both the position and momentum of the atom to zero, so the atom must retain some minimum kinetic and potential energy.
At low temperatures, zero-point energy dominates the vibrational contribution to a solid's energy. It also means that atoms in a crystal are never truly at rest, even at absolute zero.
Creation and annihilation operators
The ladder operator (also called second quantization) approach provides an elegant algebraic way to handle the quantum harmonic oscillator. Define:
- Annihilation operator:
- Creation operator:
These operators step you down or up one energy level at a time. They satisfy the commutation relation:
The Hamiltonian can be rewritten compactly as:
where is the number operator, whose eigenvalue gives the quantum number . This formalism is not just mathematically convenient; it's the natural language for describing phonons. Creating a phonon in a lattice mode is literally applying to that mode's state.
Applications in solid state physics
The single harmonic oscillator is a building block. A real crystal contains atoms, and the harmonic model extends to this many-body system by decomposing the coupled vibrations into independent normal modes, each of which behaves as its own quantum harmonic oscillator.
Lattice vibrations
Atoms in a crystal are coupled to their neighbors through interatomic forces. Treating these as harmonic springs, you can write equations of motion for all atoms and diagonalize them to find normal modes: collective oscillation patterns where every atom moves at the same frequency.
Each normal mode is characterized by a wavevector and a frequency . The relationship is the dispersion relation, which encodes how vibrational frequency depends on wavelength. The dispersion relation determines elastic properties, sound velocities, and heat capacity.
Phonons
Phonons are the quanta of these normal modes. Each phonon carries energy and crystal momentum . They obey Bose-Einstein statistics since they're bosonic quasiparticles.
Two main types:
- Acoustic phonons: All atoms in the unit cell move roughly in phase. At long wavelengths, these correspond to sound waves. Their dispersion starts linearly: near .
- Optical phonons: Atoms within the unit cell move out of phase. These have a finite frequency even at and can interact with electromagnetic radiation (hence "optical").
A crystal with atoms per unit cell has 3 acoustic branches and optical branches.
Einstein vs Debye models
Both models estimate the phonon contribution to heat capacity, but they make different assumptions about the phonon spectrum:
- Einstein model: Assumes all oscillators vibrate at the same frequency . This gives a heat capacity that correctly goes to zero at (unlike the classical Dulong-Petit value of ), but it drops off too fast because it ignores low-frequency modes.
- Debye model: Treats the solid as a continuum with a linear dispersion up to a cutoff frequency (the Debye frequency). This correctly reproduces the dependence of heat capacity at low temperatures, which matches experiment well.
The Debye temperature is a characteristic scale: for , the heat capacity approaches the classical limit ; for , quantum effects dominate and .
Thermal properties of solids
- Heat capacity is determined by how phonon modes are thermally populated. At high temperatures all modes are excited (Dulong-Petit). At low temperatures only long-wavelength acoustic modes contribute.
- Thermal expansion cannot be explained by a purely harmonic model (a symmetric parabola gives no net shift in average position with temperature). It requires anharmonic corrections.
- Thermal conductivity depends on phonon transport: phonons carry energy through the lattice, and their mean free path is limited by scattering from defects, boundaries, and other phonons.
Anharmonic effects
The harmonic approximation breaks down when displacements become large enough that higher-order terms in the interatomic potential matter. Anharmonic effects are not just corrections; they're responsible for phenomena that the harmonic model simply cannot explain.
Morse potential
The Morse potential provides a more realistic description of interatomic bonding:
- is the well depth (bond dissociation energy)
- controls the width of the potential well
- is the equilibrium bond length
Unlike the harmonic parabola, the Morse potential is asymmetric: it's steeper on the compression side and flattens out at large separations (approaching the dissociation limit ). This asymmetry means the energy levels are no longer evenly spaced; they get closer together at higher energies, and eventually the bond breaks.
Thermal expansion
A purely harmonic potential is symmetric, so the average atomic position stays at the equilibrium point regardless of temperature. Thermal expansion requires the asymmetry of an anharmonic potential.
In an anharmonic well, as temperature increases and atoms explore larger displacements, they spend more time at larger separations (where the potential is shallower) than at smaller separations. The average interatomic distance therefore increases with temperature, producing thermal expansion.
The linear thermal expansion coefficient is directly related to the degree of anharmonicity in the interatomic potential.
Phonon-phonon interactions
In a perfectly harmonic crystal, phonons don't interact with each other at all; they propagate independently forever. Anharmonicity introduces phonon-phonon scattering, which is critical for thermal transport.
Two types of three-phonon processes:
- Normal (N) processes: Total crystal momentum is conserved. These redistribute energy among phonon modes but don't directly resist heat flow.
- Umklapp (U) processes: The total crystal momentum changes by a reciprocal lattice vector. These are the dominant source of thermal resistance in pure crystals at moderate temperatures.
The thermal conductivity of a crystal decreases at high temperatures primarily because Umklapp scattering becomes more frequent as more phonons are thermally excited.
Experimental techniques
Several experimental methods probe the vibrational properties of solids. Each technique has different selection rules and is sensitive to different parts of the phonon spectrum.
Raman spectroscopy
Raman spectroscopy measures the inelastic scattering of visible light by phonons. When a photon scatters off a crystal, it can create a phonon (Stokes process, photon loses energy) or absorb one (anti-Stokes process, photon gains energy). The frequency shift of the scattered light gives the phonon frequency.
Raman scattering is governed by changes in polarizability during the vibration. Only modes that change the polarizability tensor are Raman-active. Because visible light has very small wavevectors compared to the Brillouin zone, Raman spectroscopy primarily probes zone-center () optical phonons.
Infrared spectroscopy
Infrared (IR) spectroscopy measures the direct absorption of infrared light by phonons. A vibrational mode is IR-active if it produces a change in the dipole moment of the unit cell.
Raman and IR selection rules are complementary. In crystals with inversion symmetry, modes that are Raman-active are IR-inactive and vice versa (this is the rule of mutual exclusion). Like Raman, IR spectroscopy probes near zone-center optical phonons due to the small photon wavevector.
Inelastic neutron scattering
Inelastic neutron scattering (INS) is the most comprehensive technique for mapping phonon dispersion relations. Thermal neutrons have wavelengths comparable to interatomic spacings and energies comparable to phonon energies, so they can probe phonons across the entire Brillouin zone.
By measuring the change in energy and momentum of scattered neutrons, you can map out for all phonon branches. INS has no optical selection rules, so it can detect both acoustic and optical modes regardless of symmetry. The main limitation is that it requires large single crystals and access to a neutron source (reactor or spallation facility).
Advanced topics
These topics build on the harmonic oscillator framework by incorporating interactions between phonons and electrons, leading to some of the most important phenomena in condensed matter physics.
Polaron theory
When an electron moves through an ionic or polar crystal, it distorts the surrounding lattice. The electron plus its accompanying lattice distortion is called a polaron. The distortion follows the electron, increasing its effective mass and reducing its mobility.
- Large polarons extend over many lattice sites and can be treated perturbatively (Fröhlich model).
- Small polarons are localized to roughly one lattice site and move by thermally activated hopping.
Polaron effects are significant in materials like transition metal oxides, organic semiconductors, and halide perovskites.
Electron-phonon interactions
Electrons in a solid scatter off lattice vibrations, exchanging energy and momentum with phonons. The strength of this coupling is quantified by the electron-phonon coupling constant .
Key consequences:
- Electrical resistivity in metals at room temperature is dominated by electron-phonon scattering (resistivity at high temperatures).
- Renormalization of the electronic band structure: phonon interactions shift and broaden electronic energy levels.
- Cooper pairing in conventional superconductors (see below).
Techniques like angle-resolved photoemission spectroscopy (ARPES) can reveal kinks in the electronic dispersion caused by electron-phonon coupling.
Superconductivity mechanisms
In conventional (BCS) superconductors, phonons mediate an attractive interaction between electrons. The mechanism works as follows:
- An electron moves through the lattice and attracts nearby positive ions, creating a region of slightly higher positive charge density.
- A second electron is attracted to this region before the lattice relaxes.
- This phonon-mediated attraction overcomes the Coulomb repulsion at low temperatures, binding the two electrons into a Cooper pair.
- Cooper pairs condense into a macroscopic quantum state with zero resistance below the critical temperature .
BCS theory predicts that depends on the phonon spectrum and the electron-phonon coupling strength. The isotope effect (, where is the isotopic mass) provided early evidence that phonons are involved.
Unconventional superconductors (high- cuprates, iron-based superconductors) may involve pairing mechanisms beyond simple electron-phonon coupling, such as spin fluctuations, though lattice effects can still play a role.