Translation is the process of converting mRNA instructions into proteins. Ribosomes and tRNAs work together to read the genetic code and assemble polypeptide chains, turning genetic information into the functional molecules that carry out nearly every job in a cell.
The process unfolds in three stages: initiation, elongation, and termination. Each stage has distinct molecular players and checkpoints that keep protein production accurate.
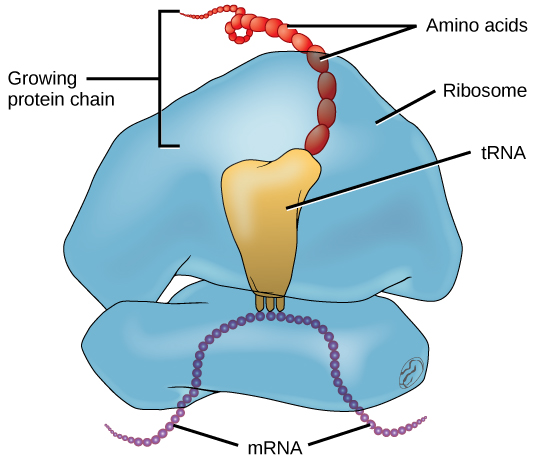
Ribosome and tRNA

Ribosomal Structure and Function
Ribosomes are the molecular machines where protein synthesis actually happens. They're made of ribosomal RNA (rRNA) and proteins, and they exist in every living cell.
Each ribosome has two parts: a small subunit and a large subunit. The small subunit reads the mRNA, while the large subunit catalyzes peptide bond formation between amino acids. These subunits only come together on an mRNA strand when translation begins.
Inside the assembled ribosome, there are three tRNA binding sites that each serve a different purpose:
- A site (aminoacyl site): where an incoming tRNA carrying its amino acid first lands
- P site (peptidyl site): where the tRNA holding the growing polypeptide chain sits
- E site (exit site): where the now-empty tRNA exits the ribosome
tRNA and Anticodon-Codon Interactions
Transfer RNA (tRNA) is the adapter molecule that physically brings amino acids to the ribosome. Think of it as a delivery vehicle: one end carries a specific amino acid, and the other end has a three-nucleotide anticodon that matches a codon on the mRNA.
The anticodon binds to its complementary mRNA codon through standard base pairing (A pairs with U, G pairs with C). This matching is what ensures the right amino acid gets added at each position. If the codon on the mRNA reads 5'-AUG-3', only a tRNA with the anticodon 3'-UAC-5' will bind there, delivering methionine.
Before a tRNA ever reaches the ribosome, an enzyme called aminoacyl-tRNA synthetase attaches the correct amino acid to it. This step uses ATP and is critical for accuracy. There's a different synthetase for each amino acid, and each one recognizes both its amino acid and the correct tRNA. If the wrong amino acid gets loaded, the entire protein could misfold.
Protein Synthesis Stages
Initiation of Translation
Initiation is where the ribosome assembles on the mRNA and positions itself at the start codon. Here's how it works step by step:
- The small ribosomal subunit binds to the 5' end of the mRNA and scans along it, looking for the start codon (AUG).
- The initiator tRNA, which carries methionine, recognizes AUG and binds to it in what will become the P site. (This is the only tRNA that ever enters the P site directly; all others enter through the A site.)
- The large ribosomal subunit joins, completing the ribosome and forming the initiation complex. The A site is now open and ready for the next tRNA.
Because every protein begins with AUG, the first amino acid in every newly made polypeptide is methionine. In many proteins, this methionine is later removed by enzymes.

Elongation and Polypeptide Formation
Elongation is the repetitive cycle where amino acids are added one at a time to the growing chain. Each cycle has three steps:
- Codon recognition: A tRNA with the correct anticodon enters the A site and base-pairs with the exposed mRNA codon.
- Peptide bond formation: The ribosome (specifically the rRNA in the large subunit, acting as a ribozyme) catalyzes a peptide bond between the amino acid in the P site and the amino acid in the A site. The growing polypeptide chain transfers to the tRNA in the A site.
- Translocation: The ribosome shifts one codon (three nucleotides) along the mRNA in the 5' → 3' direction. The tRNA that was in the A site moves to the P site, the empty tRNA in the P site moves to the E site and exits, and a new codon is exposed in the A site.
This three-step cycle repeats for every codon, and the polypeptide grows by one amino acid each round. A typical protein of 300 amino acids takes only about 20 seconds to assemble.
Termination of Translation
Termination happens when the ribosome reaches a stop codon: UAA, UAG, or UGA. No tRNA molecules have anticodons for these codons. Instead:
- A release factor protein enters the A site and recognizes the stop codon.
- The release factor triggers hydrolysis of the bond between the polypeptide and the tRNA in the P site, freeing the completed polypeptide chain.
- The ribosome disassembles into its two subunits, and both subunits release from the mRNA.
The freed polypeptide then folds into its functional three-dimensional shape, sometimes with help from chaperone proteins. The ribosomal subunits can be recycled to translate another mRNA.