Physiological Changes in the Female Body During Pregnancy
Pregnancy triggers a coordinated set of adaptations across nearly every organ system. These changes support fetal growth, prepare the body for delivery, and maintain maternal health under significantly increased physiological demands.
Reproductive System Adaptations
The menstrual cycle ceases because sustained high levels of progesterone and estrogen maintain the uterine lining instead of allowing it to shed. The uterus itself undergoes dramatic enlargement, growing from roughly 70 g to about 1,100 g by term to accommodate the fetus, placenta, and amniotic fluid.
The cervix forms a mucus plug early in pregnancy. This thick barrier seals the cervical canal and helps protect the uterine environment from bacteria and other external contaminants.
Breast and Endocrine Changes
- Breast tissue increases in size and sensitivity as it prepares for lactation. Ducts and lobules proliferate under the influence of estrogen and progesterone.
- Areolas darken and enlarge, which is thought to help the newborn locate the nipple for feeding.
- Hormonal fluctuations, particularly rising hCG, contribute to morning sickness (nausea and vomiting most common in the first trimester) and mood changes.
Cardiovascular and Respiratory Adaptations
Blood volume increases by approximately 50% over the course of pregnancy. This extra volume supports placental perfusion and meets the oxygen demands of the growing fetus. Cardiac output rises accordingly, driven by increases in both heart rate and stroke volume.
The respiratory system compensates for higher oxygen demand by increasing tidal volume (the amount of air per breath) rather than breathing rate alone. Progesterone acts on the brainstem respiratory centers to drive this change. A slight elevation in basal body temperature also occurs due to the increased metabolic rate.
Metabolic and Gastrointestinal Changes
- Metabolic rate increases by roughly 15–20%, requiring higher caloric intake to fuel both fetal growth and maternal energy needs.
- Progesterone slows gastrointestinal motility, which can lead to constipation and heartburn (as the lower esophageal sphincter relaxes and the growing uterus pushes upward on the stomach).
- Increased water retention, driven by hormonal changes and greater blood volume, commonly causes swelling in the extremities (edema).
Stages of Embryonic and Fetal Development
Human development from fertilization to birth spans about 38 weeks and is divided into distinct phases, each with characteristic events. The earliest weeks are when the basic body plan forms; the later months focus on growth and maturation.
Early Embryonic Development
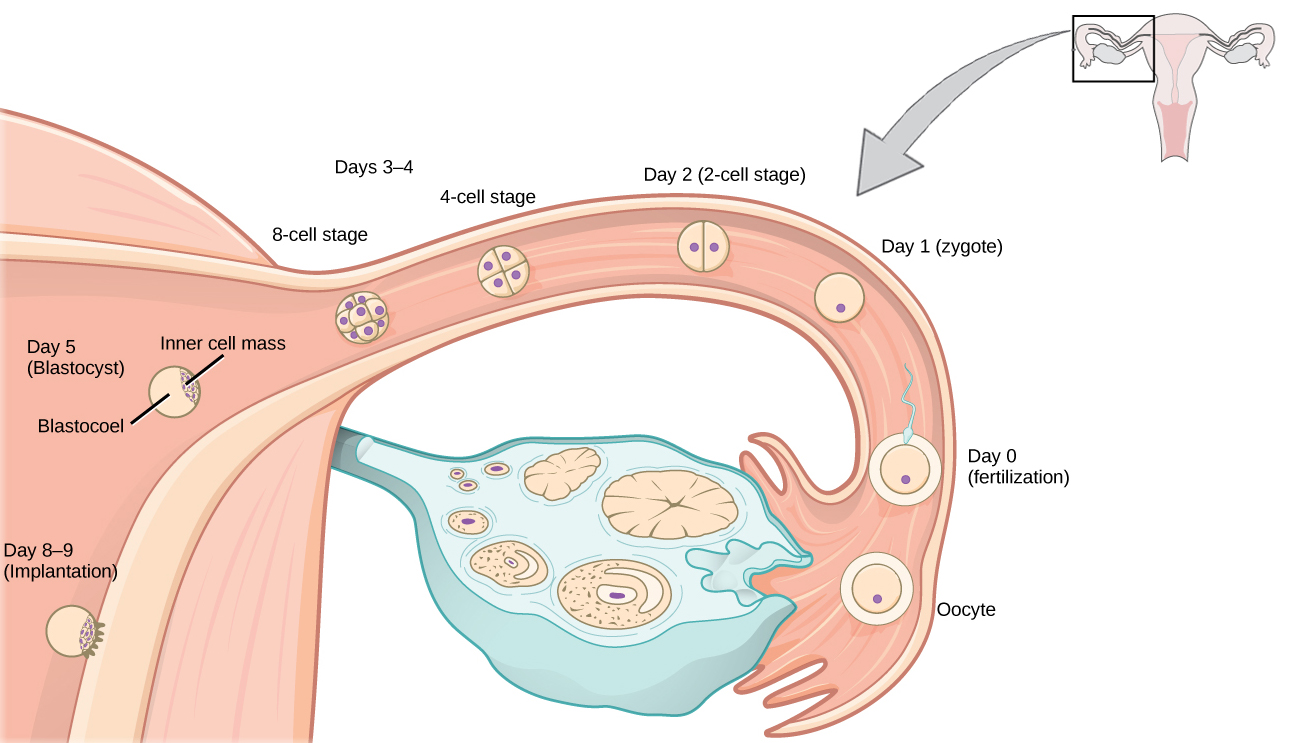
- Fertilization occurs when a sperm penetrates the egg, forming a single-celled zygote. The zygote immediately begins rapid mitotic divisions called cleavage as it travels down the fallopian tube toward the uterus.
- By about day 5, the dividing cells form a hollow ball called a blastocyst. The outer cell layer (trophoblast) will contribute to the placenta, while the inner cell mass becomes the embryo.
- Implantation occurs around days 6–10 post-fertilization, when the blastocyst embeds into the uterine wall (endometrium). This event marks the true beginning of pregnancy.
- During the embryonic stage (weeks 3–8), cells rapidly differentiate and all major organ systems begin to form. The neural tube (precursor to the brain and spinal cord) and a primitive heart are among the first structures to develop.
Fetal Development Milestones
The fetal stage runs from week 9 until birth. Organ systems that formed during the embryonic period now grow, refine, and begin to function. Key milestones include:
- Week 12: External genitalia begin to differentiate, allowing sex determination on ultrasound soon after.
- Week 16: Limb movements become more coordinated; bones begin to ossify.
- Week 20: Quickening occurs, meaning the mother can feel fetal movements for the first time.
- Week 24: Lungs begin producing surfactant, a substance that reduces surface tension in the alveoli and is critical for breathing. This is a major threshold for viability outside the womb.
- Week 28: Rapid brain growth accelerates, and the fetus develops recognizable sleep-wake cycles.
A pregnancy is considered full-term at 37 weeks, though continued maturation (especially of the lungs and brain) occurs right up until birth.
Critical Periods and Developmental Processes
Certain windows during development are especially sensitive to disruption:
- Neurulation (weeks 3–4) is the formation of the neural tube. Failure to close properly can result in defects like spina bifida or anencephaly. Adequate maternal folate intake is protective during this period.
- Organogenesis (weeks 3–8) is when all major organ systems take shape. This is the period of greatest vulnerability to teratogens, which are substances (certain drugs, infections, chemicals) that can cause birth defects.
- After week 8, the fetal period focuses on growth and functional maturation of already-formed structures. Teratogens can still cause harm, but the risk of major structural malformations decreases.
Role of the Placenta in Fetal Development
The placenta is a temporary organ that serves as the interface between maternal and fetal circulations. It handles gas exchange, nutrient delivery, waste removal, hormone production, and immune protection. Maternal and fetal blood do not mix directly; exchange occurs across thin membranes in the placenta.
Placental Formation and Structure
The placenta develops from both fetal and maternal tissues. The trophoblast cells of the blastocyst give rise to finger-like projections called chorionic villi, which extend into the maternal endometrium. These villi dramatically increase the surface area available for exchange. By the end of the first trimester, the placenta is fully functional.

Hormonal Functions
The placenta acts as a major endocrine organ, producing hormones essential for maintaining pregnancy:
- Human chorionic gonadotropin (hCG): Maintains the corpus luteum during early pregnancy so it continues producing progesterone. hCG is the hormone detected by pregnancy tests, and its levels peak around weeks 8–10.
- Estrogen: Promotes uterine growth, stimulates breast development, and supports fetal organ maturation.
- Progesterone: Maintains the uterine lining and suppresses uterine contractions, preventing premature labor. After the first trimester, the placenta takes over progesterone production from the corpus luteum.
Exchange Functions
The placenta facilitates the transfer of substances between mother and fetus without direct blood mixing:
- Oxygen diffuses from maternal blood into fetal blood; carbon dioxide moves in the opposite direction.
- Nutrients such as glucose, amino acids, and fatty acids cross from maternal to fetal circulation.
- Fetal waste products (urea, creatinine) transfer into maternal blood for excretion by the mother's kidneys.
Protective Functions
- The placenta acts as a selective barrier, blocking many (but not all) pathogens and harmful substances from reaching the fetus.
- Maternal IgG antibodies cross the placenta, providing the fetus with passive immunity that lasts for several months after birth.
- The placenta can metabolize certain drugs and toxins, partially reducing fetal exposure, though many substances still cross.
Hormonal Changes and Effects During Pregnancy
Pregnancy hormones shift dramatically across trimesters. Understanding which hormones dominate at each stage, and what they do, is essential for making sense of the physiological changes described above.
Early Pregnancy Hormones
- hCG rises rapidly after implantation, peaking around weeks 8–10. Its primary role is maintaining the corpus luteum so progesterone production continues. In male fetuses, hCG also stimulates fetal testosterone production, which drives male sexual differentiation.
- Progesterone is initially produced by the corpus luteum, then increasingly by the placenta (the "luteal-placental shift" occurs around weeks 8–12). It maintains the endometrium and suppresses uterine contractions.
- Estrogen levels climb steadily throughout pregnancy, promoting uterine growth, breast development, and fetal organ maturation.
Mid to Late Pregnancy Hormones
- Human placental lactogen (hPL) promotes mammary gland development and alters maternal metabolism. It increases maternal insulin resistance, which helps ensure glucose availability for the fetus.
- Relaxin, produced by the corpus luteum and placenta, softens the cervix and relaxes pelvic ligaments in preparation for delivery.
- Cortisol levels rise gradually. In late pregnancy, cortisol plays a key role in promoting fetal lung maturation (stimulating surfactant production) and preparing the maternal body for the stress of labor.
Labor and Postpartum Hormones
- Oxytocin levels increase near term, stimulating rhythmic uterine contractions during labor. After delivery, oxytocin promotes milk ejection (the "let-down reflex") and maternal bonding.
- Prolactin rises throughout pregnancy, preparing breast tissue for milk production. However, high progesterone and estrogen levels during pregnancy actually inhibit milk secretion.
- The rapid drop in progesterone and estrogen after delivery of the placenta removes this inhibition, allowing prolactin to trigger milk production.
Potential Complications During Pregnancy
Metabolic and Cardiovascular Complications
- Gestational diabetes: Abnormal glucose metabolism that develops during pregnancy, often related to the insulin resistance caused by hPL. Managed through diet, exercise, and sometimes insulin therapy. Usually resolves after delivery but increases the mother's long-term risk of type 2 diabetes.
- Preeclampsia: Characterized by new-onset high blood pressure and protein in the urine (proteinuria) after 20 weeks of gestation. Can progress to life-threatening eclampsia (seizures) if untreated. Requires close monitoring and may necessitate early delivery.
- Deep vein thrombosis (DVT): Pregnancy creates a hypercoagulable state (blood clots more easily), increasing the risk of clots in deep veins. Managed with anticoagulants and compression stockings.
Placental and Uterine Complications
- Placenta previa: The placenta implants over or near the cervical opening, potentially causing painless bleeding in the second or third trimester. Often requires cesarean delivery.
- Placental abruption: Premature separation of the placenta from the uterine wall before delivery. Causes painful bleeding and can lead to fetal distress due to interrupted blood supply.
- Ectopic pregnancy: The blastocyst implants outside the uterus, most commonly in a fallopian tube. This is not viable and requires immediate medical intervention because a growing ectopic pregnancy can rupture and cause life-threatening hemorrhage.
Fetal and Pregnancy Loss Complications
- Miscarriage (spontaneous abortion): Loss of pregnancy before 20 weeks, most commonly due to chromosomal abnormalities. May be managed with supportive care or medical/surgical intervention.
- Stillbirth: Fetal death after 20 weeks of gestation. Delivery is still required, and causes are often investigated.
- Intrauterine growth restriction (IUGR): The fetus grows more slowly than expected for gestational age, often due to placental insufficiency. Managed through close monitoring and possible early delivery.
Immunological Complications
- Rh incompatibility: Occurs when an Rh-negative mother carries an Rh-positive fetus. The mother's immune system may produce antibodies against fetal red blood cells, potentially causing hemolytic disease of the newborn in subsequent pregnancies. Prevented by administering Rh immunoglobulin (RhoGAM) to the mother.
- Gestational thrombocytopenia: A mild decrease in platelet count during pregnancy. Usually benign but monitored for bleeding risk.
Labor and Delivery Process
Labor is the process by which the fetus, placenta, and membranes are expelled from the uterus. It's driven by a positive feedback loop involving oxytocin and prostaglandins, and it unfolds in three defined stages.
Initiation and Stages of Labor
Labor is initiated by a complex interplay of hormonal and mechanical signals. Rising oxytocin and prostaglandin levels, combined with increased uterine stretch, trigger the onset of contractions.
- Stage 1 (Dilation):
- Latent phase: Cervical effacement (thinning) and early dilation to about 3–4 cm. Contractions are mild and irregular.
- Active phase: Rapid cervical dilation from 4 to 10 cm with regular, strong contractions. This is typically the most intense part of Stage 1.
- Stage 2 (Expulsion): Begins at full dilation (10 cm) and ends with delivery of the infant. The mother actively pushes, and this stage typically lasts 20 minutes to 2 hours.
- Stage 3 (Placental delivery): The placenta detaches from the uterine wall and is expelled, usually within 5–30 minutes after birth.
Hormonal and Physiological Mechanisms
- Oxytocin stimulates uterine smooth muscle contractions and promotes maternal-infant bonding.
- Prostaglandins contribute to cervical ripening (softening and dilation) and enhance uterine contractions.
- The Ferguson reflex is a neuroendocrine positive feedback loop: as the fetus descends and presses on the cervix, stretch receptors send signals to the hypothalamus, which triggers more oxytocin release. More oxytocin causes stronger contractions, which push the fetus further down, stimulating even more oxytocin. This cycle intensifies until delivery occurs.
Monitoring and Management During Labor
- Fetal heart rate is monitored throughout labor. The normal range is 110–160 bpm; deviations may indicate fetal distress.
- Contraction intensity and frequency are tracked to assess labor progress.
- Maternal vital signs (blood pressure, heart rate, temperature) are monitored continuously.
- Pain management options range from non-pharmacological methods (breathing techniques, positioning, hydrotherapy) to pharmacological interventions (most commonly epidural anesthesia).
Immediate Postpartum Period and Maternal Changes
After delivery, the body begins reversing the adaptations of pregnancy. This process takes roughly 6 weeks, though some changes (like breast tissue) persist longer if breastfeeding.
Uterine Involution and Bleeding
The uterus contracts rapidly after delivery to compress blood vessels at the placental site and control bleeding. This process of returning to pre-pregnancy size is called uterine involution. The uterus shrinks from about 1,000 g immediately postpartum to 50–100 g by 6 weeks.
Lochia is the vaginal discharge that follows delivery. It progresses through three phases:
- Lochia rubra (red): Days 1–3, contains blood and tissue
- Lochia serosa (pink/brown): Days 4–10, thinner and lighter
- Lochia alba (white/yellow): Days 10 through about 6 weeks, mostly white blood cells and mucus
Hormonal Shifts
- The rapid decline in estrogen and progesterone after placental delivery removes the inhibition on prolactin, triggering milk production.
- Oxytocin levels remain elevated, promoting uterine contractions (which help with involution) and the milk let-down reflex during breastfeeding.
- Prolactin levels increase further with breastfeeding, sustaining milk production.
Cardiovascular and Respiratory Changes
Blood volume and cardiac output gradually return to pre-pregnancy levels over 6–8 weeks. Respiratory function normalizes as oxygen demand decreases and the diaphragm is no longer displaced upward by the uterus. The hypercoagulable state of pregnancy persists for several weeks postpartum, so the risk of thromboembolism remains elevated during this period.
Gastrointestinal and Urinary Changes
- Gastrointestinal motility returns to normal, relieving the constipation and heartburn common during pregnancy.
- Temporary urinary retention may occur due to pelvic floor trauma, edema, and decreased bladder sensation from labor.
- Increased urinary frequency is common in the early postpartum days as the body eliminates the excess fluid retained during pregnancy.