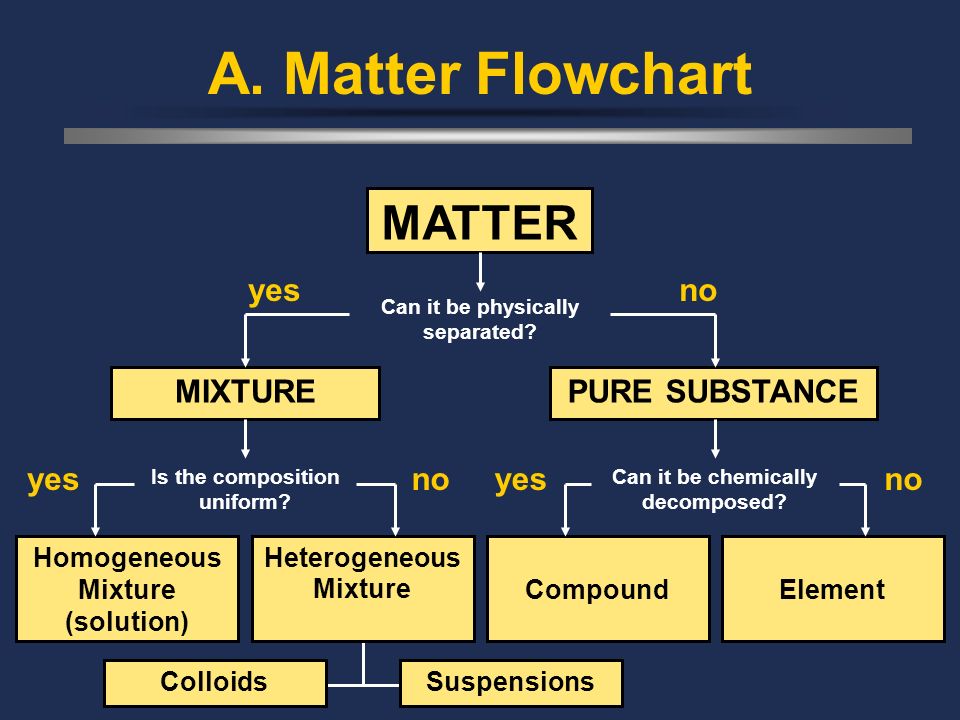
If you recall from the last section, matter can be categorized according to state or composition. The three states of matter will be elaborated upon in unit three of this AP Chemistry course, but we will delve deeper into categorizing matter by composition in this guide.
Remember that the first division in the classification of matter by composition is between a pure substance and a mixture. A pure substance is composed of a single type of atom or molecule, whereas a mixture is a composition of two or more elements or compounds.
What are Mixtures?
As discussed, mixtures are materials made up of two or more substances that are physically combined.

Formula Units: Pure Substances and Mixtures
To better clarify the difference between pure substances and mixtures, let's discuss them in terms of formula units. As a review, a formula unit represents the lowest whole number ratio of atoms that can be used to describe a compound.
While pure substances contain atoms or formula units of a single type, mixtures contain atoms or formula units of two or more types (and their proportions vary).
Different Types of Mixtures
Just like there are different types of pure substances, there are different types of mixtures!
💭 Remember that a pure substance can be either an element or a compound. Both are composed of a single type of atom or molecule, but compounds can be chemically decomposed into smaller parts (elements).
Mixtures are separated into two categories: homogeneous and heterogeneous.
- Homogeneous mixtures are composed of two or more substances but are uniform in composition. In other words, if you take a look at a homogeneous mixture, you will not be able to see the parts it is made up of.
- 🌊 Salt water is made up of different compounds (water + salt) but you can't see ALL of the elements; it all looks like one mixture of just water.
- 💨 Air is a homogeneous mixture made up of gases. So many different gases make up air, but it is impossible to see its composition.- An important characteristic of homogeneous mixtures is that they are difficult to separate. Imagine trying to take a bottle of soda and separating it, how would that even work?
- Heterogeneous mixtures are visibly composed of more than one element or compound. Their composition is not uniform, so the proportions of different compounds can vary!
- 🍨 Rocky road ice cream is a prime example of a heterogeneous mixture. You could see each of the "compounds" (chocolate ice cream, nuts, and marshmallows).
- 🥗 Salad is another heterogeneous mixture since you can see all of the ingredients you physically combined in a bowl.- Unlike homogenous mixtures, heterogeneous mixtures are often separable. Their components are physically distinct from one another, so it is pretty easy to disassemble them. Think about taking a salad apart!
Separating Mixtures
Chemists often want to separate a mixture into its components in chemical experiments. There are several ways to separate mixtures, but the most common ways in chemistry are distillation and filtration. These techniques use different physical or chemical properties of the components in a mixture in order to separate them.
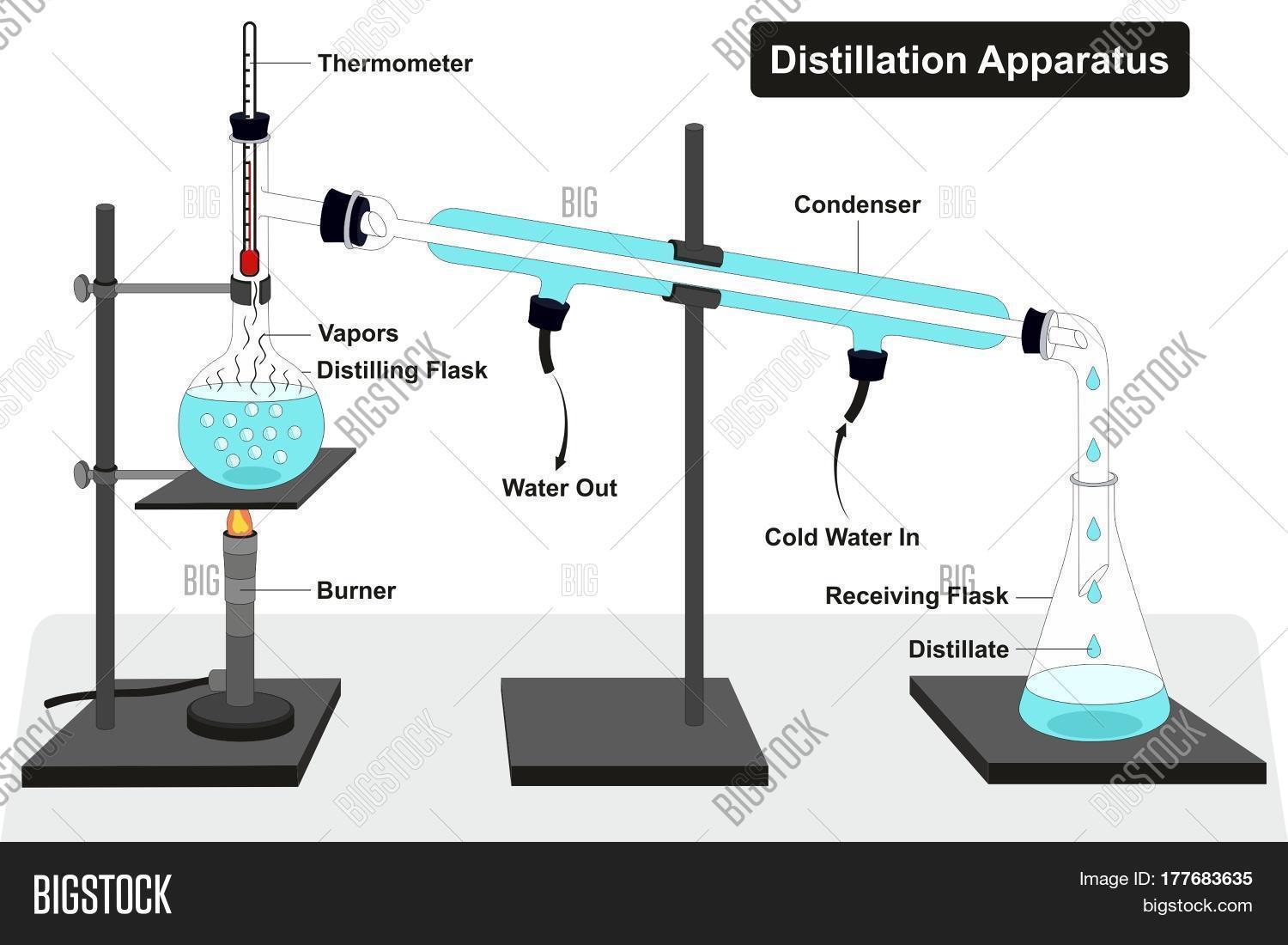
Distillation
Distillation separates components in liquid mixtures by evaporating the most volatile substance first, usually the substance with the lowest boiling temperature. The most important thing to know is that it separates based on differences in boiling points.
Think about a mixture of water and alcohol. Since the boiling point of alcohol is lower than that of water, the alcohol is going to evaporate first. Therefore, the vapor collected will contain more alcohol than water.
Filtration
Filtration separates mixtures by using a mesh or filter to separate solids from liquids. However, this only works with heterogeneous mixtures.
Imagine you are filtering a solution of salt, water, and sand. Can you guess what will get caught up in the filter paper? Only the sand will. This is because the salt will dissolve in the water since it is soluble.
Only insoluble substances will be filtered with filtration.
The filtrate would then be both salt and water, or salt water. Therefore, filtration isn't effective if you want to fully separate all of these elements. After filtering the sand out, you would have to evaporate the water out of the salt water solution to separate the salt from the water.
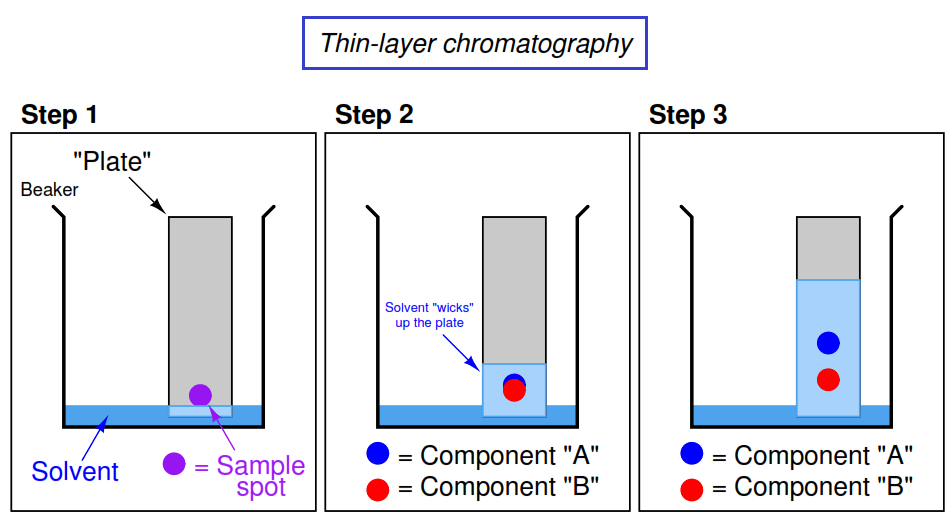
Thin-Layer Chromatography (TLC)
Chromatography is a method used to identify and compare mixtures based on their attraction to solids and liquids or differences in polarity (polarity is further explained in future units).
Image Courtesy of Instrumentation ToolsIn the first step, someone drew a dot with a marker. Most of the ink in markers are a mixture of a number of substances and thin-layer chromatography can separate these substances.
In step two, the solvent climbed up the filter paper📄 and began to separate the sample spot by its substances.
A quick rundown of polarity to explain TLC:
Polarity is one way to compare solvents or substances. A substance may be polar or nonpolar.
You may hear the phrase, "Like dissolves like" a lot. This means polar substances dissolve in polar solvents and nonpolar substances dissolve in nonpolar solvents.
Do you know how oil and water never mix? Water is a polar substance and oil is a nonpolar substance. Since they aren't the same polarity, they never mix.
Most TLC plates are made up of polar silica, which you can think of as a very small powder. This silica is considered the stationary phase, while the solvent in the TLC chamber is considered the mobile phase. A general rule of thumb is that polar compounds are more strongly attracted to the stationary phase, and will move less, while nonpolar compounds are more readily eluted with the solvent.
☝️Let's refer back to the image above. If the solvent is nonpolar, could you guess which component is more nonpolar? If Compound A traveled farther up the TLC plate, that means it was more readily eluted with the solvent. With this being said, Compound A is less polar than Compound B.
AP NOTE: There are other types of chromatography as well but this one is the most commonly tested on the AP Chemistry Exam.
AP Practice Question - 2018 #2
This question is adapted from the 2018 AP Chemistry Exam posted on the College Board website.
There are questions like this one on several past AP exams. This is only part a, but it's good to recognize what they are asking here. They basically want you to draw the reactant mixture given the information you are provided with.
The two things you want to note in this question are:
- The types of compounds that should be drawn in the reactant mixture
- How much of each element is present in the product mixture Step 1) This question introduces the reaction and tells us that the two reactants are NO and O2.
Step 2) If you count how many atoms of each element are present, you would get 8 N atoms and 12 O atoms.
Since you have fewer Nitrogen atoms, it'd be good to draw the NO reactant first. Draw as many as you can until you run out of nitrogen atoms. This would get us to have 8 NO molecules. Now, we have 4 O atoms remaining. Since each O2 compound contains two atoms of oxygen, we could only draw 2 O2 molecules with the remaining oxygen.
A sample response looks like the following:
Make sure you use the key they gave you when drawing the molecules! You should always look out for requirements and directions on how to draw diagrams. The orientation at which you draw the molecules doesn't matter, as long as you draw them correctly.
AP Practice Question - 2017 #4
This question is adapted from the 2017 AP Chemistry Exam posted on the College Board website.
What You Know
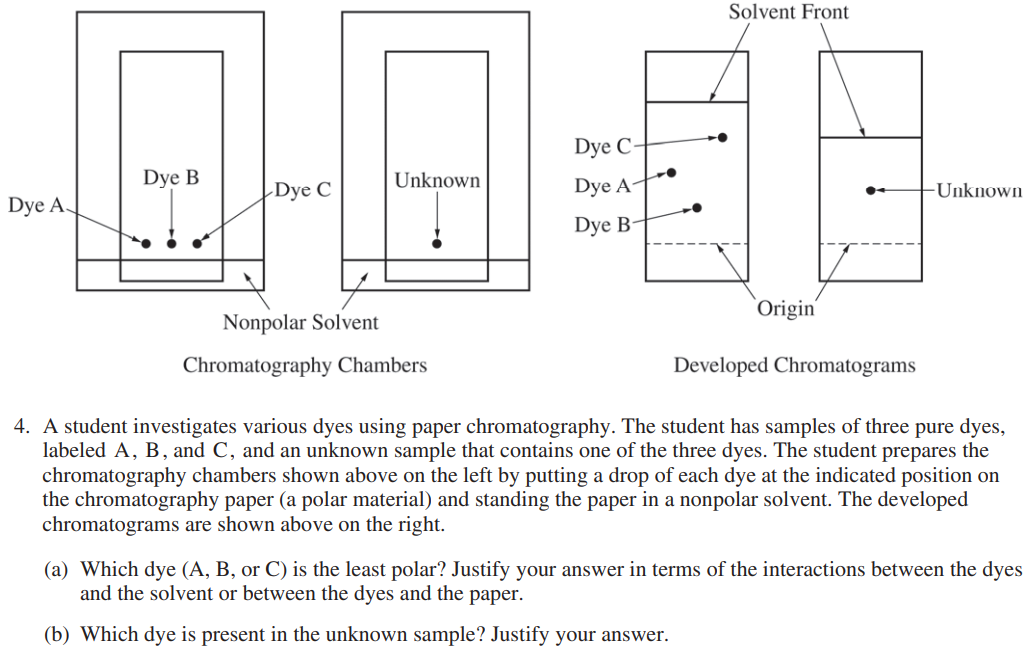
This is an AP free-response question testing your knowledge about thin layer chromatography! In this question, they are giving you several pieces of information:
- You have two TLC plates developing: one with labeled and known dots, and one with an unknown dot.
- You are developing your TLC plates in a nonpolar substance.
- You are told where the dots began, so you can compare them with regard to the distance they traveled from the origin.
Breaking down part a:
Part (a) is asking you which of the known dyes (A, B, or C) is the least polar and to justify your response. Since they explicitly asked, your justification must involve the interactions between the dyes and the solvent or the dyes and the paper.
You should automatically be able to eliminate A as the least polar dye, since it travels a distance between C and B. You can see that dye C travels the farthest in a nonpolar solvent, while dye B has traveled the least. Remember "like dissolves like," so the least polar dye, or the most nonpolar dye, will travel the farthest in a nonpolar solvent!
Therefore, your answer to part a should be similar to one of the following:
"Dye C is the least polar dye because it moved the farthest on the TLC plate. This is because nonpolar dyes are more attracted to the nonpolar solvent."
"Dye C is the least polar dye because it moved the farthest on the TLC plate. This is because nonpolar dyes are less strongly attracted by the polar TLC plates."
Both of these responses would earn you the full two points on this part of #4.
Breaking down part b:
Part (b) asks you to figure out which dye is present in the unknown sample and justify. This question can easily check many students if they aren't paying close attention to the difference in the TLC plates.
The solvent in the developed TLC plates with the dyes has traveled much farther than the one with the unknown. This is important to note because in order to accurately tell which dye is in the unknown, the solvent fronts must be in the same location. Therefore, we have to use proportions to answer this question.
The unknown dye seems to have traveled half the distance that the solvent did, so we have to find which dye did the same on the other TLC plate. It looks like dye A followed the same behavior, so it must be in the unknown.
According to the scoring guidelines, either of the following responses would earn you a full two points:
"Dye A is present in the unknown sample. The unknown sample moves to a position that is midway between the origin and the solvent front, and so does dye A"
Dye A has a retention factor (Rf) that is close to 0.50 on the chromatogram with the three dyes, and the unknown also has a retention factor close to 0.50."
We haven't discussed Rf yet, but it is basically a measure of the distance traveled by the dye relative to the distance traveled by the solvent. This number is always going to be between 0 to 1.
Rf = (distance traveled by the component) / (distance traveled by the solvent)
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
elemental analysis | An analytical technique used to determine the relative numbers of atoms of each element in a substance and to assess its purity. |
elemental composition by mass | The percentage or proportion of each element present in a substance, expressed as a mass fraction or mass percentage. |
mixture | Materials that contain atoms, molecules, or formula units of two or more types, whose relative proportions can vary. |
pure substance | A material with a fixed, definite composition and consistent properties throughout. |
purity | The degree to which a substance contains only one type of atom, molecule, or formula unit without contamination from other substances. |
Frequently Asked Questions
What's the difference between a pure substance and a mixture?
A pure substance contains only one kind of atom, molecule, or formula unit in a sample—its composition is fixed (e.g., elemental Cu or pure NaCl). A mixture contains two or more different atoms/molecules/formula units and the relative proportions can vary (e.g., air, saltwater). Mixtures can be homogeneous (uniform composition, like a salt solution) or heterogeneous (nonuniform, like sand in water). On the AP CED this difference matters because elemental analysis and percent composition by mass, mole fraction, and mass fraction are used to determine purity or the composition of mixtures (1.4.A.1–1.4.A.2). Practically: if composition is constant and describable by one formula, it’s a pure substance; if proportions can change, it’s a mixture. For more review on Topic 1.4 see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do you calculate the percent composition of elements in a compound?
Percent composition by mass tells you what percent of a compound’s mass comes from each element. Steps: 1. Write the formula and find each element’s total mass in one mole: (# of atoms of element) × (atomic mass from periodic table). 2. Find the molar mass of the whole compound (sum of all element masses). 3. Percent composition for an element = (mass of that element in 1 mol ÷ molar mass of compound) × 100%. Quick example—H2O: H mass = 2 × 1.008 = 2.016 g; O = 16.00 g; molar mass = 18.016 g. %H = (2.016/18.016)×100 = 11.19%; %O = 88.81%. This skill is part of elemental analysis and mass-percent composition in the AP CED (Topic 1.4 keywords: mass percent composition, mass fraction, empirical/molecular formula). On the exam you may be asked to “calculate” these values, so show your work and units. For more practice and a refresher, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and the large practice set (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about elemental analysis - what does it actually tell us?
Elemental analysis tells you the relative amounts (by mass) of each element in a sample. From those mass percents you convert to moles, get mole ratios, and determine an empirical formula (and with molar mass, a molecular formula). It’s also used to assess purity: if measured composition deviates from the expected percent composition, there’s an impurity or mixture. Common methods include combustion analysis (for organics) and gravimetric analysis (precipitating and weighing a component). On the AP exam you’ll be asked to calculate mass percent, mole fraction, empirical formulas, and sometimes use elemental analysis to judge purity—so practice converting mass % → moles → smallest whole-number ratio. For a focused review see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l), the Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1), and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to know the mass percent of each element in a mixture?
You need mass percent because it’s the bridge between what you actually weigh and the chemical identity/behavior of a mixture. Mass percent tells you each element’s fraction of the total mass so you can: - convert masses into moles (via molar mass) to find empirical/molecular formulas or mole fractions (CED keywords: mass percent composition, mole fraction, empirical formula); - assess purity or quantify impurities (gravimetric/combustion analysis), which matters for labs and industry; - do stoichiometry for reactions involving mixtures (knowing mass % lets you calculate limiting reactants and yields); - predict colligative or mixture properties that depend on composition. On the AP exam you’ll be expected to calculate percent composition and use it to get mole ratios (see Topic 1.4 in the CED). For guided review and examples, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens to the composition when you mix two pure substances together?
If you mix two pure substances, the result is a mixture—not a new pure substance—whose composition depends on the relative amounts you mixed. The mixture can be homogeneous (uniform, e.g., two miscible liquids) or heterogeneous (not uniform, e.g., sand + salt). Quantitatively you describe composition with mass fraction or mass percent, and with mole fraction (useful for gases and stoichiometry). Elemental mass composition of each pure component stays the same, but the overall elemental composition of the mixture is a weighted average (mass balance). That means you can calculate the mixture’s percent composition by mass or mole fraction from the amounts you started with—a skill AP Chem tests under Topic 1.4 (CED 1.4.A). For review, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How can you tell if a sample is pure or if it's a mixture?
You can tell a sample is pure vs a mixture two ways: qualitatively and quantitatively. - Qualitative: look/feel (heterogeneous mixtures show visible phases; homogeneous look uniform). Use simple tests—filtration, paper/column chromatography, or measuring a sharp melting/boiling point (a pure substance has a sharp, reproducible melting/boiling point; mixtures broaden or depress it). - Quantitative (AP-style, CED-aligned): do elemental analysis (mass percent composition, combustion or gravimetric analysis) and calculate mole or mass fractions. Compare measured mass % or empirical formula to the theoretical values for the pure substance; if they match within experimental error the sample is likely pure (purity assessment). Chromatography or spectroscopy can also show multiple components. Elemental analysis is explicitly named in the CED as a method to determine purity (Topic 1.4). For a quick review, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l). For more practice, check Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1) and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain how to find the empirical formula from percent composition?
Turn each percent into grams (assume 100 g sample so percent = grams). Convert grams → moles using atomic masses. Divide all mole values by the smallest mole number to get whole-number mole ratios. If a ratio isn’t whole, multiply by 2, 3, or 4 to clear halves/thirds. Write the empirical formula from those whole-number subscripts. Quick example: 40.0% C, 6.7% H, 53.3% O → assume 100 g: 40.0 g C (40.0/12.01 = 3.33 mol), 6.7 g H (6.7/1.008 = 6.65 mol), 53.3 g O (53.3/16.00 = 3.33 mol). Divide by smallest (3.33): C1 H2 O1 → empirical formula CH2O. This uses AP keywords: mass percent composition, mole ratio, empirical formula (CED 1.4.A.2). For more step-by-step review see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the point of doing elemental analysis in the lab?
Elemental analysis tells you what elements are in a sample and how much of each by mass—and that’s super useful. In the lab you use techniques like combustion or gravimetric analysis to get mass percent (or mass fraction), convert those to moles, find mole ratios, and from that get an empirical (and sometimes molecular) formula. That same data tells you purity (is your “pure” substance really pure?) and the composition of mixtures (mass percent or mole fraction of components), which is exactly what Topic 1.4 tests (LO 1.4.A in the CED). On the AP exam you’ll be expected to do the math (percent composition → moles → mole ratio) and interpret whether a sample is impure or part of a mixture. If you want a concise walkthrough and practice problems for this topic, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and thousands of practice questions (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how the relative proportions in mixtures can vary - what does that mean?
“Relative proportions can vary” just means a mixture can have different amounts of each substance—there isn’t a fixed formula like a pure compound. For example, saltwater could be 3% NaCl by mass (sea water) or 10% NaCl (brine). Both are mixtures of the same two components, but their mass percent, mass fraction, and mole fraction are different. Those quantitative measures (mass percent = mass of solute / total mass ×100, mole fraction = moles of component / total moles) are exactly what the CED expects you to use to describe mixtures (see keywords: mass percent composition, mole fraction, mass fraction, purity). Changing proportions changes properties (boiling point, density, conductivity), so many AP questions ask you to calculate composition or determine purity from analysis. If you want worked examples and practice converting between mass % and mole fraction, check the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
How do you convert from mass percent to actual grams of each element?
Pick a 100.0 g sample—that makes mass percent = grams (simple trick AP students use). Steps: 1. Convert mass % → grams: mass % of element × 100.0 g / 100 = grams of that element. (e.g., 40.0% C → 40.0 g C) 2. Convert grams → moles: grams ÷ atomic/molar mass (use periodic table). (40.0 g C ÷ 12.01 g·mol⁻¹ = 3.33 mol C) 3. If you need an empirical formula, divide all mole values by the smallest mole number to get whole-number ratios. (Example: C 3.33 mol, H 6.67 mol, O 3.33 mol → divide by 3.33 → C1 H2 O1 → empirical CH2O) 4. If you started with a different sample mass (not 100 g), multiply mass fraction (mass%/100) by that sample mass to get grams. This is exactly the elemental-analysis/mass-percent routine tested in Topic 1.4 (percent composition by mass → mole ratio → empirical formula). For a refresher and practice problems, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
Why would the composition by mass be different in mixtures vs pure substances?
Pure substances have a fixed mass percent composition because they’re made of one formula unit (one set of atoms) repeated: for example, pure H2O is always 11.2% H and 88.8% O by mass. Mixtures, however, contain two or more different substances whose relative amounts can change, so the overall composition by mass depends on how much of each component you have. If you add more of substance A, the mass fraction and mass percent of A go up and the others go down. That’s why mixtures don’t have a single “percent composition” the way pure compounds do, and why elemental analysis is used to determine purity or the composition of a mixture (mass percent, mole fraction, mass balance). This idea maps to CED LO 1.4.A (mass percent composition, mass fraction, purity assessment). For a focused review, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between molecular formula and percent composition?
Molecular formula tells you the exact number of each atom in a molecule (e.g., C6H12O6); percent composition tells you the mass percent of each element in that molecule (e.g., %C, %H, %O). You can go both ways: given a molecular formula, compute mass percent by (mass of element in one formula unit / molar mass of the molecule) × 100. Given percent composition (from elemental analysis), convert each percent to moles (assume 100 g sample), find the smallest whole-number mole ratio to get an empirical formula, then use the molar mass (if known) to scale the empirical formula to the molecular formula. These steps are exactly what the CED lists under elemental analysis, mass percent composition, empirical formula, and molecular formula (Topic 1.4 / 1.3). If you want worked examples and practice (strongly recommended for AP exam prep), see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and the practice problem bank (https://library.fiveable.me/practice/ap-chemistry).
How do chemists actually determine what's in an unknown mixture?
Chemists identify components of an unknown mixture by separating and measuring each part, then reporting mass- or mole-based composition (mass percent, mole fraction). Common AP-level techniques: gravimetric analysis (isolate and weigh a component), elemental/combustion analysis (measure C, H, N to get mass percents and empirical formula), chromatography (separates components for qualitative ID and relative amounts), and titration (quantitative concentration of an analyte). For mixtures with known substances, you can do mass balance calculations and convert mass percent → moles → mole ratio or empirical/molecular formulas. Purity assessment uses percent composition vs. theoretical values. On the exam you’ll be expected to do those conversions and interpret data (CED Topic 1.4 keywords: elemental analysis, combustion analysis, gravimetric analysis, percent composition by mass, mole ratio). For step-by-step examples and practice problems, check the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l) and the Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1).
I missed the lab on elemental analysis - what were we supposed to learn?
You were supposed to learn how elemental analysis tells you the composition and purity of a substance and how that links to mixtures. In the lab you’d measure masses (or combustion products) to calculate percent composition by mass, convert those percents to moles to get empirical (and molecular) formulas, and use mass fraction or mole fraction to describe how a substance sits in a mixture. Practically: you practice mass-balance calculations, learn gravimetric/combustion analysis techniques, and use results to decide if a sample is pure or contains impurities. Those skills map to CED 1.4.A (mass percent composition, empirical formula, purity assessment) and show up on the exam under Unit 1 (7–9% of the exam) and related math routines. If you want to review the exact formulas, step-by-step examples, and practice problems, check the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l), the Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1), and grab practice questions (https://library.fiveable.me/practice/ap-chemistry). If you want, tell me one sample mass you missed and I’ll walk you through the calculations.
Can you have the same percent composition but different molecular formulas?
Yes. Percent composition only tells you the mass percent of each element, not how many atoms are in the actual molecule. Different molecules can have the same percent composition if they share the same empirical formula (or one is a whole-number multiple of the other). Example: glucose (C6H12O6), lactic acid (C3H6O3), and the empirical formula CH2O all have the same percent composition by mass (≈40.0% C, 6.7% H, 53.3% O), even though their molecular formulas and properties differ. Elemental analysis gives you mass percent → empirical formula; you need molar mass (or other data) to get the molecular formula. This idea is tested on the AP Exam in Topic 1.4 (elemental analysis, mass percent, empirical vs molecular formula). For a clear review, see the Topic 1.4 study guide (https://library.fiveable.me/ap-chemistry/unit-1/composition-mixtures/study-guide/dKotYK10xJPuFb5FD15l). For extra practice, check the unit overview (https://library.fiveable.me/ap-chemistry/unit-1) and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).