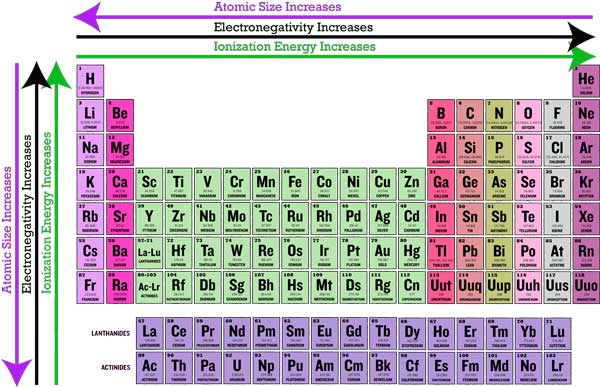
A cool thing about the periodic table is that it is organized to demonstrate different trends and properties of elements that can be explained by the pattern of electron configurations and the presence of electron-filled orbitals. The periodicity of the periodic table, or its tendency to recur at intervals, can help you estimate the properties of atoms that haven't even been discovered yet.
For the sake of the AP Chemistry exam, rather than only understanding the trends, you should be able to explain why they happen.
Foundational Concepts for Periodic Trends
In order to fully understand why the trends occur the way they do, it's important to cover the following topics:

Organization of the Periodic Table
As mentioned before, the periodic trends aren't too difficult to grasp since they follow the chronology of the periodic table. It was purposely made to group chemicals of similar properties together.
It is also important to note that the periodic table is divided into 18 columns (called groups) and 7 rows (called periods).
Periods on the Periodic Table ➡️
🤨 Properties that differ: Going horizontally, each period is organized in order of increasing atomic number. The atomic number, or the number of protons in an atom's nucleus, determines the basic chemical properties of said element. This trend contributes to the differing effective nuclear charge of elements in the same group, which we'll discuss below.
🤝 Shared properties: In each row, the elements have the same number of occupied electron shells.
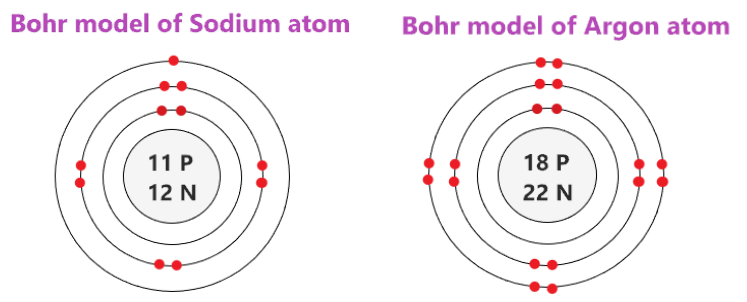
Let's compare sodium, which is the first element in period 3, with argon, the last element in period 3.
Image Courtesy of topblogtenzYou could see here that both sodium and argon have a total of three occupied electron shells, following the pattern of elements in the same period. However, sodium has 11 protons (represented by its atomic number of 11), and argon has 18 protons (represented by its atomic number of 18).
Groups on the Periodic Table ⬇️
🤨 Properties that differ: One group on the periodic table is organized so that as you move down a group, the number of occupied electron shells increase.
🤝 Shared Properties: Every element in one group has the same number of valence electrons in its outermost shell. Because these elements all have the same number of valence electrons, they can bond to other elements in similar ways. In other words, these elements tend to have similar chemical properties.
Some groups on the periodic table have a name, since the elements in a single group have similar properties. For example, the elements in group 18 are called noble gases. All noble gases are generally unreactive due to their high stability.
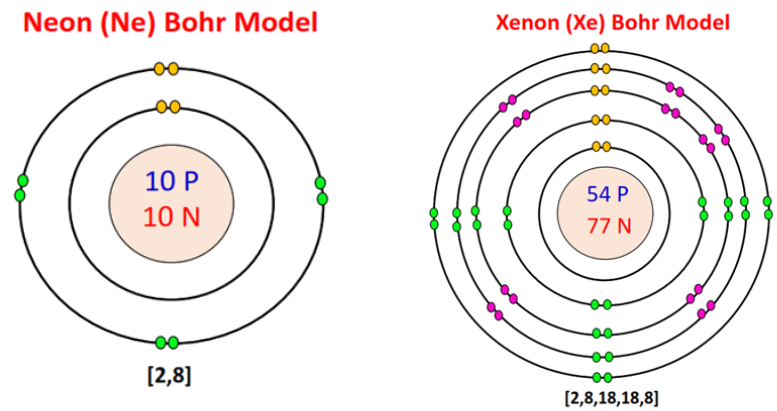
Let's now compare neon, the second noble gas in group 18, with xenon, the fifth noble gas.
Image Courtesy of topblogtenzBoth neon and xenon have eight valence electrons in their outermost shell, but neon only has two occupied electron shells, while xenon has five occupied electron shells. The fact that they both have a full octet, or eight valence electrons in their outermost shell, makes them both noble gases. Having a full octet makes these elements very stable, and therefore unreactive.
Effective Nuclear Charge
What two subatomic particles make up the nucleus? Protons and neutrons, right? Since neutrons are neutral, protons are the particles that contribute to the positive charge of the nucleus, or the actual nuclear charge (Z).
Now, try to connect this to Coulomb's law which calculates the attraction between two atoms. Each electron orbiting the nucleus experiences both an attraction to the nucleus and a repulsion from the atom's other electrons.
In order to fully comprehend the forces electrons feel, remember that opposite charges attract. Electrons are negatively charged, and the nucleus is positively charged.
Electrons that are in the outer shells of an atom may be shielded by the innermost electrons because of the electron-electron repulsion present. In order to accurately represent the nuclear charge of a nucleus, we must account for both the actual nuclear charge and the charge shielded by other electrons (S).
You do not need to know this formula for the AP exam, but it may help you better understand nuclear charge. Effective nuclear charge is equal to the actual nuclear charge (Z) - the charge shielded by other electrons (S).
👉 Want to read more about Coulomb's law? Check out this study guide on Atomic Structure and Electron Configuration.
5 Periodic Trends to Know for AP Chemistry
Let's try to apply the concepts above to the five periodic trends that you should learn and understand for the AP Chemistry exam. The best way to conceptualize this information is to think about it through the concepts we went over above. When in doubt, think about nuclear charge and the periodicity of the periodic table.
Atomic Radius
The atomic radius of an atom is the distance between an atom's nucleus and its valence electrons.
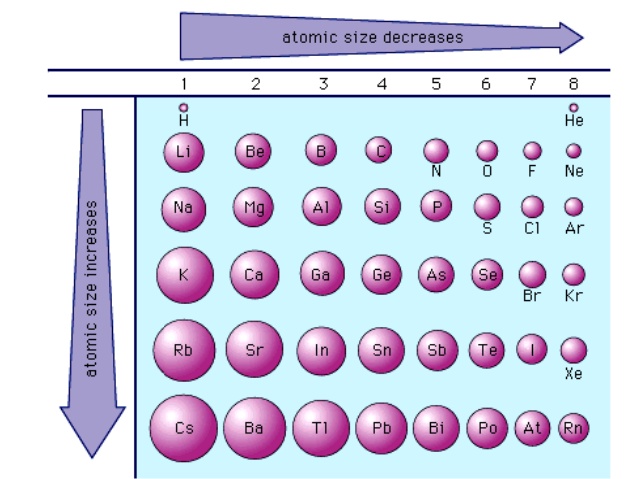
Across a Period - Smaller ⬇️
Going from left to right on the periodic table, the atomic radii get smaller. As you go right, the atomic numbers increase. This means that there is a higher nuclear charge which increases the pull the nucleus has on the electrons. The closer the electrons are to the nucleus, the smaller the distance.
This trend can also be explained by the fact that all elements in a period have the same number of shells. For example, both Li and F have 2 shells, like Na and Ar both have 3 shells.
Down a Group - Larger ⬆️
As you go down a group on the periodic table, the atomic radii increase. This is because the number of occupied shells increases. For example in group 1, Li has 2 occupied shells while Cs has 6 occupied electron shells (similar to the trend explained above with neon and xenon).
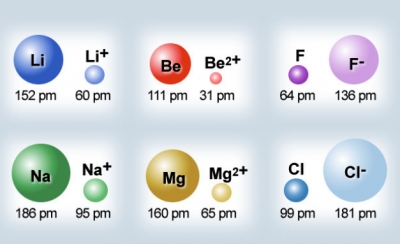
Image Courtesy of ThivyaapriyaIonic Radius
The ionic radius is the distance between the nucleus of an ion and the valence electrons of that said ion.
➕ Ions < Atoms
- When metals ionize, they lose an electron and become positive ions. Losing an electron makes the ion decrease in size. There is also less shielding and electron-electron repulsion present, allowing the remaining valence electrons to be closer to the nucleus. - Sometimes, metals lose their entire valence shell, significantly decreasing their size.
➖ Ions > Atoms
- When nonmetals ionize, they gain an electron and become negative ions. Gaining an electron makes the ion increase in size. There is also more electron-electron repulsion present due to the increased number of negatively charged particles.
Electronegativity
Electronegativity refers to how strongly a nucleus attracts electrons of another atom.
Across a Period - Increases ↖️
This is because the elements on the right side of the periodic table (such as noble gases in group 18) have more protons in their nuclei, which gives them a greater positive charge. Having a greater nuclear charge makes the nuclei more effective at attracting electrons.
Down a Group - Decreases ↘️
As you go down a group, the atomic size of an atom increases. Therefore, the nucleus of one atom is farther away from the electrons of another atom, and the attraction between the two is weaker.
Tip - Fluorine is the most electronegative element on the periodic table, with a value of 4.0. Just remember that and try to compare other elements to where fluorine is located on the periodic table.
Ionization Energy
Ionization energy is the amount of energy needed to remove the valence electrons of an atom. Since there are often multiple valence electrons, there are multiple ionization energies. The first I.E. is the amount required to remove the most loosely held electron and the second I.E. is the amount required to remove the second most loosely held electron.
Across a Period - Increases ↖️
Since size decreases across a period, the nucleus and the electrons are more closely attracted to each other. This stronger attraction makes it harder to remove a valence electron. Thus, it takes more energy to do so.
Down a Group - Decreases ↘️
As you go down a group, the amount of occupied electron shells increases. The valence electrons that are farther away are more loosely attracted to the nucleus. Therefore, it takes less energy to remove them.
Information to Note
- The 2nd I.E. will always be greater than the first since inner electrons are more strongly attracted to the nucleus.
- There are some exceptions to this trend!- 1st I.E. for group 15 > 1st I.E. for group 16.
- This highlighted electron is in an already occupied orbital. The shielding experienced will lower the required energy to remove the outermost electron, making the 1st I.E. lower than expected for S.- I.E. of Be>B and Mg>Al because of something called quantum tunneling.
- Quantum Tunneling - The 2p electron in B is easier to remove than a 2s electron in Be. This is because the 2p electron spends more time away from the nucleus while the 2s electron may tunnel towards the nucleus for enough time to make it more difficult to remove.
Valence Electrons
- Using the ionization energies given below, determine the number of valence electrons this element has.1. I1 = 5001. I2 = 15001. I3 = 70001. I4 = 9000 When you are given this type of question, just look for the huge jump in ionization energies. This gap occurs because of how much more energy it takes to remove electrons closer to the nucleus.
Therefore, this element has 2 valence electrons!
Electron Affinity
Electron affinity is the energy change when an electron is added to an atom in the gaseous state.
Across a Period - more negative ➖
Down a Group - more positive ➕
The more negative the energy, the more energy is released! Electron affinity is typically negative just because an atom releases energy when it gains an electron. However, how negative depends on this trend. You may be able to explain this trend by thinking about electronegativity.
Because of this, you may expect flourine to have the highest magnitude of electron affinity. However, chlorine does! Flourine is too small of an atom and the electrons are so close together that they would repel, which takes energy.
Overview Image of Periodic Trends
Image Courtesy of SciencetuteVocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
atomic properties | Characteristics of atoms such as size, ionization energy, electron affinity, and electronegativity that vary predictably across the periodic table. |
atomic radius | The size of an atom, typically measured as the distance from the nucleus to the outermost electrons. |
Coulomb's law | The principle that the electrostatic force between charged particles is proportional to the product of their charges and inversely proportional to the square of the distance between them. |
effective nuclear charge | The net positive charge experienced by an electron in an atom, accounting for shielding by inner electrons. |
electron affinity | The energy change that occurs when an electron is added to an atom in the gas phase. |
electron configuration | The arrangement of electrons in an atom or ion, describing which orbitals and subshells are occupied and how many electrons are in each. |
electronegativity | A measure of an atom's ability to attract valence electrons in a chemical bond; increases across a period and decreases down a group in the periodic table. |
electronic structure | The arrangement of electrons in an atom, including the distribution across shells and subshells. |
ionic radius | The size of an ion, which differs from the neutral atom due to the gain or loss of electrons. |
ionization energy | The energy required to remove an electron from an atom in the gas phase. |
periodicity | The recurring pattern of properties in elements that repeats across periods and groups in the periodic table. |
shell | Energy levels in an atom that contain electrons, designated by principal quantum numbers (n = 1, 2, 3, etc.). |
shell model | A model of atomic structure in which electrons occupy discrete energy levels or shells around the nucleus. |
shielding | The reduction in the effective nuclear charge experienced by outer electrons due to repulsion from inner electrons. |
subshell | A subdivision of an electron shell characterized by a specific orbital type (s, p, d, or f) and containing orbitals of similar energy. |
unpaired electrons | Electrons that occupy orbitals alone rather than in pairs, affecting an atom's magnetic and chemical properties. |
Frequently Asked Questions
What is effective nuclear charge and why does it matter for periodic trends?
Effective nuclear charge (Zeff) is the net positive charge felt by a valence electron after accounting for repulsion from inner electrons (shielding). You can think of it as Z (actual protons) minus shielding from core electrons; higher Zeff means the nucleus pulls valence electrons in more strongly (Coulomb’s law: force ∝ charge/distance²). Zeff matters because it explains AP periodic trends: across a period Zeff increases (more protons, similar shielding) → atomic radius decreases, ionization energy and electronegativity increase, electron affinity often becomes more negative. Down a group Zeff experienced by valence electrons increases only slightly while principal quantum number increases, so radii increase and ionization energies decrease. Understanding Zeff, shielding, and subshell penetration lets you qualitatively predict those properties on the exam (CED Topic 1.7). For a quick review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I remember which way atomic radius increases on the periodic table?
Think: bigger going down, bigger going left—atomic radius increases down a group and from right to left across a period. Why: going down adds electron shells (more distance from the nucleus), and inner electrons shield valence electrons, so Coulomb attraction weakens and size grows. Going across a period left → right, protons are added but electrons enter the same shell, so effective nuclear charge (Zeff) increases; stronger Zeff pulls electrons closer and the radius decreases. Memory shortcut: “Down = bigger shells; Right = tighter pull.” Or picture layers (more shells) stacking down, and a stronger nucleus pulling in to the right. This is exactly the periodic-trends reasoning in Topic 1.7 (effective nuclear charge, shielding, Coulomb’s law). For a quick review and practice problems, see the Topic 1.7 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and the Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1).
Why does ionization energy increase across a period but decrease down a group?
Ionization energy (IE) is the energy needed to remove an electron. Across a period IE increases because effective nuclear charge (Zeff) increases: protons are added to the nucleus while shielding by inner electrons stays about the same, so valence electrons feel a stronger Coulomb attraction and are held more tightly (harder to remove). Down a group IE decreases because valence electrons occupy higher principal shells (larger r) and there’s more shielding from inner shells; larger distance + more shielding reduce the nucleus’ pull (Coulomb’s law: force ∝ Z/r^2), so it’s easier to remove an electron. Use electron configurations and shell-model reasoning to justify trends on the AP exam (Topic 1.7, CED 1.7.A). For practice visualizing and questions, check the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about shielding - can someone explain how electrons shield each other?
Shielding means inner electrons reduce the full positive pull of the nucleus felt by outer (valence) electrons. Core electrons (filled shells) do most of the shielding because they sit between nucleus and valence electrons; by Coulomb’s law, more intervening negative charge lowers the effective nuclear charge (Zeff) experienced by an outer electron. Electrons in the same shell shield each other only partially because they’re at similar distances and have less penetration toward the nucleus. Subshell penetration matters too: s electrons penetrate closer and feel a higher Zeff than p or d electrons in the same shell. Practically, greater shielding → larger atomic radius, lower ionization energy and lower electronegativity. You’ll need this for AP Topic 1.7 problems connecting electron configuration, Zeff, and trends (CED keywords: effective nuclear charge, shielding, Coulomb’s law). For a focused review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and try practice questions (https://library.fiveable.me/practice/ap-chemistry) to apply these ideas.
What's the difference between atomic radius and ionic radius?
Atomic radius = the size of a neutral atom (usually defined as half the distance between nuclei in a bonded pair). Ionic radius = the size of an ion. They behave differently because changing electron count changes electron–electron repulsions, shielding, and effective nuclear charge (Zeff)—the CED keywords you should use to explain trends. Removing electrons (making a cation) reduces electron–electron repulsion and often removes an outer shell, so the ion is much smaller than its neutral atom. Adding electrons (making an anion) increases repulsion and lowers Zeff per electron, so the ion is larger. In an isoelectronic series (same electron number), radius decreases with increasing nuclear charge. These ideas connect to Coulomb’s law, shielding, and electron configuration—all listed in Topic 1.7 of the CED. For a quick review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and practice related problems at (https://library.fiveable.me/practice/ap-chemistry).
Why is fluorine the most electronegative element if it's so small?
Short answer: fluorine is the most electronegative because its nucleus pulls bonding electrons very strongly. On the periodic table trend, electronegativity increases across a period and up a group because effective nuclear charge (Zeff) increases while shielding stays low. F has a very high Zeff for its valence electrons and only one small shell of core electrons (minimal shielding), so Coulomb’s law gives a strong attraction at a very short distance (small atomic radius). That strong attraction outweighs the fact that adding an electron to a tiny atom has some electron–electron repulsion (which is why chlorine’s electron affinity is actually a bit more exothermic than fluorine’s), but on the Pauling electronegativity scale F still ranks highest. This answer ties to CED concepts: Zeff, shielding, Coulomb’s law, atomic radius, and electronegativity (Topic 1.7). For a quick review, see Fiveable’s Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
How does Coulomb's law explain why atoms get smaller across a period?
As you go across a period, protons are added to the nucleus but valence electrons stay in the same principal shell. Coulomb’s law (F = k q1q2 / r^2) says the attractive force between the positively charged nucleus and an electron increases when the nuclear charge (q1) increases or the distance (r) decreases. Because added protons increase the nuclear charge and inner-shell shielding doesn’t increase much for electrons in the same shell, the effective nuclear charge (Zeff) felt by valence electrons rises. Stronger attraction pulls the electron cloud closer to the nucleus, so atomic radius decreases across a period. This explanation uses CED keywords: Coulomb’s law, effective nuclear charge, shielding, shell model (Topic 1.7). For a focused review, see the Periodic Trends study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain electron affinity in simple terms - I keep mixing it up with ionization energy
Think of ionization energy (IE) and electron affinity (EA) as opposite questions about an atom’s love for electrons. - Ionization energy: How much energy do you need to remove an electron from a neutral atom? (Higher IE = electron held tightly.) IE relates to valence electrons, effective nuclear charge, shielding, and Coulomb’s law—closer electrons + higher Zeff → larger IE. - Electron affinity: How much energy is released (or required) when a neutral atom gains an extra electron? A more negative EA means the atom wants an electron more. EA depends on how well the nucleus (Zeff) attracts one more electron and whether that electron enters a new shell (more shielding → less favorable). Quick memory trick: IE = energy to pull an electron away. EA = energy change when you add one. Both are periodic: IE generally increases left→right and up→down (with exceptions); EA trends similarly but with more irregularities. For AP review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do noble gases have such high ionization energies compared to the elements next to them?
Noble gases have very high first ionization energies because their valence shells are completely filled (e.g., He: 1s2; Ne: 1s2 2s2 2p6). A full shell is especially stable, so removing an electron requires breaking that stable configuration. From the CED ideas: Coulomb’s law and effective nuclear charge (Zeff) explain this—with a full shell the nucleus’s pull on valence electrons is strong (higher Zeff) and there’s minimal additional shielding from inner electrons, so the electron is held tightly (small atomic radius → larger attraction). Neighboring elements (like the alkali metals just left of a noble gas) have one electron in a new, more weakly held valence shell with lower Zeff and greater shielding, so their ionization energies are much smaller. This is exactly the periodic-trend reasoning AP asks you to use (CED Topic 1.7: effective nuclear charge, shielding, Coulomb’s law). For a quick review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why calcium has a larger atomic radius than magnesium - aren't they in the same group?
Good question—they’re in the same group, but not the same period. Calcium’s valence electrons are in the 4th shell (n = 4) while magnesium’s are in the 3rd shell (n = 3). That extra shell puts the valence electrons farther from the nucleus, so even though Ca has more protons, the increased distance and stronger inner-electron shielding reduce the net pull on those outer electrons. By Coulomb’s law and the shell model, force ∝ q1q2/r², so increasing r (shell size) has a big effect and gives Ca a larger atomic radius. Effective nuclear charge (Zeff) felt by the valence electrons doesn’t rise enough to overcome the added shell and shielding. This is exactly the periodic trend AP Chem asks you to explain (CED 1.7.A: shielding, Zeff, Coulomb’s law). For a quick refresher, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and more practice problems at (https://library.fiveable.me/practice/ap-chemistry).
How do periodic trends help predict if an element will form ionic or covalent bonds?
Periodic trends tell you how atoms gain/lose/share electrons, so they're great for predicting bond type. Key clues: - Electronegativity (EN): big EN difference → electrons transfer → ionic; small difference → electron sharing → covalent. (A rough Pauling cutoff often taught is ≈1.7, but AP wants qualitative reasoning.) - Ionization energy (IE) and electron affinity (EA): low IE (left/metal) → easy cation formation; high EA/EN (right/nonmetal) → prefer anions or shared pairs. - Atomic/ionic radius & effective nuclear charge (Zeff): large atoms (low Zeff, big radius) hold valence electrons weakly and tend to form cations; small atoms with high Zeff hold electrons tightly and form anions or strong covalent bonds. So: a metal + nonmetal (large EN difference) → likely ionic; two nonmetals (similar EN) → likely covalent. Use the periodic table position and these trends (IE, EN, EA, radii, Zeff) to justify your prediction for AP-style explanations. For a quick review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and try practice questions (https://library.fiveable.me/practice/ap-chemistry).
What happens to electronegativity when you go down a group and why?
Electronegativity decreases as you go down a group. That’s because valence electrons are farther from the nucleus (larger atomic radius) and there are more inner electrons that shield the nucleus, so the effective nuclear charge felt by a bonding electron is lower. By Coulomb’s law, weaker attraction (larger r, more shielding) → lower tendency to attract shared electrons. Use AP keywords: effective nuclear charge, shielding, Coulomb’s law, valence electrons. This trend helps predict bond polarity and reactivity on the exam (Topic 1.7 in the CED). For quick review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why does sodium have a lower first ionization energy than lithium?
Sodium has a lower first ionization energy than lithium mainly because its valence electron is farther from the nucleus and is better shielded by inner electrons. Li has configuration 1s2 2s1; Na has 1s2 2s2 2p6 3s1. The 3s electron in Na is in a higher principal energy level (n = 3) so its average distance from the nucleus is larger. By Coulomb’s law, attraction falls off with distance, and the inner (core) electrons in Na shield the nucleus more effectively, reducing the effective nuclear charge felt by the 3s electron. Even though Na has one more proton than Li, the increased distance + greater shielding outweigh the extra nuclear charge, so it’s easier to remove Na’s valence electron. This explanation uses CED ideas: shell model, shielding/effective nuclear charge, and Coulomb’s law (Topic 1.7). For a concise review, see the Periodic Trends study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and try practice questions (https://library.fiveable.me/practice/ap-chemistry).
I'm studying for the AP exam - what periodic trends do I absolutely need to memorize?
You should memorize the four core periodic trends and why they happen: ionization energy, atomic/ionic radius, electron affinity, and electronegativity—and the directions across a period (increase for IE, EA, EN; decrease for atomic radius) and down a group (IE, EA, EN decrease; radius increases). Link these to effective nuclear charge and shielding (use Coulomb’s law and shell model to explain exceptions). Know that ionic radius changes when you lose/gain electrons (cations smaller, anions larger) and be able to predict relative values for given elements. The AP CED expects qualitative explanations (Topic 1.7: 1.7.A.1–1.7.A.3), not memorizing aufbau exceptions. Unit 1 is ~7–9% of the exam, so be ready to apply these ideas to predict properties and justify answers. For a concise review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe) and practice lots of problems at (https://library.fiveable.me/practice/ap-chemistry).
How do you use effective nuclear charge to predict which element has a higher ionization energy?
Effective nuclear charge (Zeff) = nuclear charge felt by an electron after shielding by other electrons (qualitatively Z_eff ≈ Z − S). Higher Zeff means the nucleus pulls valence electrons more strongly (Coulomb’s law), so those electrons are held tighter and ionization energy (IE) is larger. How to use it to predict IE: - Compare same period: more protons → larger Zeff (shielding roughly constant), so IE increases left → right (e.g., Na < Mg < Al generally). - Compare same group: Zeff changes little down a group, but valence electrons are farther out and more shielded → IE decreases down a group (e.g., Li > Na > K). - Watch exceptions due to subshell structure/penetation (e.g., 2nd → 3rd period anomalies), but AP expects qualitative reasoning with Coulomb’s law and shielding (CED 1.7.A.2). For a quick review, see the Topic 1.7 study guide (https://library.fiveable.me/ap-chemistry/unit-1/periodic-trends/study-guide/J1NnoL1NHgd6B1dG2UZe). For more practice, check Fiveable’s AP Chem practice problems (https://library.fiveable.me/practice/ap-chemistry).