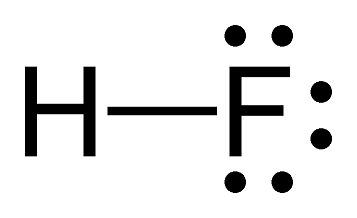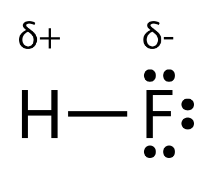We've discussed valence electrons several times in this unit's study guides already! Here is a quick recap regarding what you should know so far:
- Valence electrons are the outermost electrons in an atom.
- Valence electrons are found in the s and p orbital of the outermost shell.
- A gap in ionization energies could tell us how many valence electrons an element has.
- Elements in the same group on the periodic table have the same number of valence electrons.
Foundational Knowledge of Bonding

Valence Electrons
From the periodic table, you could tell how many valence electrons an element has.
Image Courtesy of ck12The graph above skips over the transition metals because those are much less predictable. The AP Chemistry exam is going to primarily focus on the other elements, but it is good to be familiar with several transition metals (like Co, Cu, Zn).
Just by looking at the periodic table, you could tell that oxygen has six valence electrons and carbon has four valence electrons. Remember that the number of valence electrons an atom has affects the way the element bonds with others. Therefore, elements in the same group tend to bond with similar elements and form similar compounds. Here are some examples:
- Elements in group one can all bond with chlorine: LiCl, NaCl, KCl, RbCl.
- Elements in group two can all bond with O: MgO, CaO, SrO, BaO.
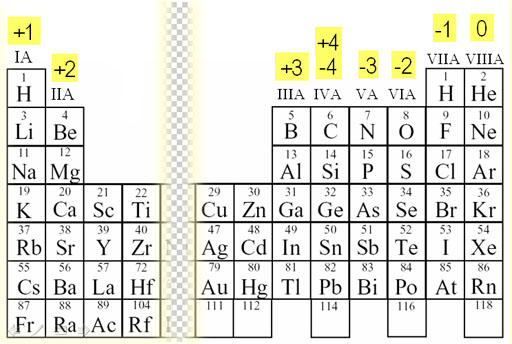
Charges of Ions
Before getting into bonds, it is also good to memorize the charges of most elements on the periodic table when they bond with another element. This will make more sense once we look into what happens when elements form a bond.
Essentially, ions are charged atoms or molecules that have gained or lost electrons. Atoms may become ions in order to achieve a more stable electron configuration. Remember, most things in chemistry try to become more stable! ⚖️
Image Courtesy of Chemistry LandAgain, the transition metals aren't included here because they have various charges and very different properties. Don't worry though, the AP won't question you about them. At most, you would have to write their electron configurations.
Types of Elements
There are three different types of elements: metals, nonmetals, and metalloids.
Image Courtesy of ck12Metals are good conductors of heat and electricity, shiny, malleable (can bend), and ductile (can be made into a wire).
Nonmetals are the complete opposite: bad conductors of heat and electricity and brittle.
Metalloids have properties of both metals and nonmetals and there are only seven of them.
Electronegativity
Electronegativity refers to how strongly a nucleus attracts electrons of another atom. This comes into play when two atoms are sharing valence electrons since the pull of the electrons depends on how electronegative the atom is!
This is one of the five essential periodic trends to know and understand for AP Chemistry. Remember that fluorine is the most electronegative element on the periodic table, with a value of 4.0. From here, you can try to compare other elements to where fluorine is located on the periodic table.
👉 Want a quick refresh on electronegativity? Check out our study guide on "Periodic Trends."
Types of Bonds
Elements bond to achieve the lowest possible energy to reach the highest stability. There are two different types of bonds you should be familiar with: ionic bonds and covalent bonds.
Ionic Bonds
Ionic bonds are formed by the transfer of electrons from one atom to another, usually from a metal to a nonmetal.
The atom that loses an electron will gain a positive charge and is called a cation (usually a metal).
The atom that gains an electron will gain a negative charge and is called an anion (usually a nonmetal).
Some properties of ionic compounds include very strong bonds, solubility in water, and the ability to strongly conduct heat and electricity.
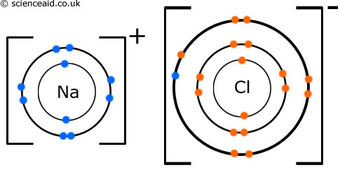
Example of an Ionic Bond - NaCl
In the ionic compound NaCl, sodium (Na) loses an electron and obtains a positive charge, while chlorine (Cl) gains an electron and therefore obtains a negative charge.
The one valence electron that was in Na was transferred to the chlorine atom in order for both ions to have a full octet. The octet rule is based on the idea that atoms are most stable when they have eight valence electrons in their valence shell.
Group one elements and group 17 elements (halogens) often bond this way to reach stability. They have the proper number of valence electrons that enable them to both reach a full octet. In fact, when forming an ionic bond with halogens, group one elements lose an entire electron shell.
When sodium and chlorine become ions, their electron configuration actually matches that of the noble gas closest to it. Here is an example:
Covalent Bonds
Covalent bonds are formed when atoms share electrons (usually two nonmetals). Some properties of covalent bonds include low melting points and weak electroconductivity abilities.
There are actually two types of covalent bonds: polar covalent bonds and nonpolar covalent bonds.
- Polar covalent bonds are a type of bonding where electrons are unequally shared between two different nonmetals. This is due to the two atoms having significantly different electronegativities.
- Nonpolar covalent bonds are a type of bonding where electrons are equally shared between, usually, two of the same nonmetal. This bond typically forms between two atoms that have a similar tendency to attract electrons, or rather, two atoms that have very similar electronegativities.
Covalent bonds are strong and stable, and they are actually responsible for the properties of many common substances that we interact with on a daily basis. Some examples include water, methane, carbon dioxide, and even proteins, DNA, and carbohydrates!
Polar-Covalent Example - HF
Hydrogen and fluorine create a polar covalent bond. Fluorine attracts electrons more strongly due to its high electronegativity of 4.0, resulting in an unequal distribution of electrons. Think about it this way: fluorine is super greedy to fulfill its full octet of eight valence electrons, so it more strongly attracts them towards it.
Image Courtesy of StudyNote the difference in representation between a covalent bond and an ionic bond. The dash here represents two shared electrons. Rather than a transfer of electrons, hydrogen is sharing one electron and fluorine is sharing one electron to enable both atoms to have their full octet.
Nonpolar Example - Cl2
In the covalent compound Cl2, two chlorine atoms bond together to share the final electron that they need to have their full octet.
Image courtesy of Wayne BreslynJust like in a polar covalent bond, they are sharing electrons with each other to obtain the full octet. However, note here that two atoms of the same element are bonding. In other words, their electronegativities are the same, so they would have the same pull on the electrons they share.
This does not mean that all nonpolar covalent bonds are between two atoms of the same nonmetal. The carbon-oxygen bonds in carbon dioxide (CO2), as well as the carbon-hydrogen bonds in methane (CH4) are also nonpolar covalent. Take a look at their electronegativities:
- Electronegativity of hydrogen: 2.2
- Electronegativity of carbon: 2.55
- Electronegativity of oxygen: 3.44
Charges and Partial Charges
When two atoms interact with each other in a bond, there is often a distribution of charge. Let's take a look at the different charge distributions we can see.
In a nonpolar covalent bond, the electronegativities of the two atoms are very similar, if not the same. Therefore, the electrons in the bond will be shared equally, resulting in a neutral charge distribution. The positive charge of the two nuclei is balanced by the negative charge of the shared electrons.
When there is a large gap in electronegativities between the two atoms in a bond, we start to see charges. In a polar covalent bond, two atoms, such as fluorine and hydrogen, share electrons unevenly. Fluorine has an electronegativity of 4.0, while hydrogen has that of 2.2. The atom with the higher electronegativity (F) will have a partial negative charge, while the atom with the lower electronegativity (H) will have a partial positive charge. This is because the very negative electrons are shifted towards the fluorine, creating an abundance of negative charge, and away from the hydrogen, creating a "loss" of positive charge.
Image Courtesy of Chem LibreTextsThe Greek letter delta (δ) is used to indicate an atom that has either a positive or negative partial charge.
In an ionic bond, the transfer of electrons forms ions with complete opposite charges. Take a look at the transfer of electrons between the sodium and chlorine atoms above.
The loss of an electron causes the sodium to be a little extra positive (hence its +1 charge), whereas the gain of an electron causes the chlorine to be a little extra negative (hence its -1 charge). The reason why metals are typically the atoms to donate an electron is that they have very low electronegativities. Sodium has an electronegativity of 0.93, while chlorine has an electronegativity of 3.16. Compare this difference to that between hydrogen and fluorine! This is why the charges are not partial.
Atoms with low electronegativities are more likely to donate electrons and form cations, while atoms with high electronegativities are more likely to accept electrons and form anions.
Check your Understanding
Try these little questions on your own and see how you do! It's just to see how well you understood this key topic:
Atoms of Ca combine with atoms of Br to form an ionic bond.
- What ratio would they combine in?
- What other compounds have this same ratio with Ca?
- What elements could form an ionic bond with sulfur?
Answers
- Calcium and bromine bond in a 1:2 ratio, creating the compound CaBr2. This is because Ca has a +2 charge and Br has a -1 charge. In order for the two to bond together and form a neutral compound, there must be two Br atoms.
- We need other elements that have a -1 charge in order for them to bond in this 1:2 ratio with calcium. This includes all of the group 17 elements on the periodic table, such as fluorine, chlorine, bromine, and iodine.
- In a 1-to-1 ratio, any elements in group two would form an ionic bond with sulfur. Some examples include MgS, CaS, and BaS. In a 2-to-1 ratio, any elements in group one would form an ionic bond with sulfur. An example is Na2S.
🎥 Watch AP Chemistry teacher Mónica Gracida discuss how to do electron configurations of transition metals and how to quickly know the valence electrons for atoms.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
chemical bond | An attractive force between atoms that holds them together in a compound. |
ionic charge | The net electrical charge of an ion, determined by the number of valence electrons and the element's position on the periodic table. |
ionic compound | Compounds formed by the electrostatic attraction between positively charged cations and negatively charged anions. |
periodic table | A systematic arrangement of elements organized by atomic number and grouped by similar chemical properties. |
periodicity | The recurring pattern of properties in elements that repeats across periods and groups in the periodic table. |
reactivity | The tendency of an element to undergo chemical reactions and form bonds with other elements. |
valence electrons | Electrons in the outermost shell of an atom that participate in bonding and determine many properties of substances. |
Frequently Asked Questions
What are valence electrons and why are they important for bonding?
Valence electrons are the electrons in an atom’s outermost energy level—the ones that interact with other atoms. They matter because chemical bonds form from attractions between valence electrons and nuclei (CED 1.8.A.1). Atoms gain, lose, or share valence electrons to reach a stable octet (or duet for H/He), which explains why alkali metals form +1 cations and halogens form −1 anions (CED 1.8.A.3). Trends in ionization energy, electron affinity, and electronegativity (and a element’s column on the periodic table) predict whether atoms will form ionic bonds (electron transfer → cation + anion) or covalent bonds (electron sharing). Understanding valence electrons helps you predict formulas, charges, ionic radii, and reactivity—exactly the stuff tested in Unit 1/Topic 1.8 on the AP exam (see the study guide for Topic 1.8 for a quick review: https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx). For extra practice, try AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I figure out how many valence electrons an element has?
For main-group elements (Groups 1, 2 and 13–18) you can usually get valence electrons straight from the group number: - Group 1 → 1 valence e–, Group 2 → 2, Groups 13–18 → 3–8 (e.g., Group 17 halogens = 7). Another way is to write the electron configuration and count the electrons in the highest occupied principal energy level (the outermost s and p orbitals). Those counted are the valence electrons—this matters for predicting ionic charges (octet rule → metals lose to form cations, nonmetals gain to form anions). Transition metals are trickier: their valence electrons include outer s and sometimes d electrons, so charges are less predictable and often given (learn common charges: Fe2+/3+, Cu+/2+, etc.). This skill maps directly to CED Topic 1.8 (valence electrons → ionic compounds, cation/anion predictions). For a quick refresher tied to AP-style practice, see the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do elements in the same group have similar chemical properties?
Elements in the same group (same column) have similar chemical properties because they have the same number of valence electrons. Valence electrons determine how atoms interact—how easily they lose or gain electrons (ionization energy, electron affinity, electronegativity) and the typical charge they form in ionic compounds (CED 1.8.A.1, 1.8.A.3). For example, Group 1 atoms have one valence electron and commonly form +1 cations; halogens (Group 17) have seven valence electrons and typically form −1 anions. Because bonding behavior depends on valence electrons and the nucleus’ attraction to them, elements with the same valence shell form analogous compounds and show periodic trends in reactivity (CED 1.8.A.2). For AP review, focus on valence-electron patterns, common ionic charges, and trends like ionization energy—see the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try practice problems (https://library.fiveable.me/practice/ap-chemistry) to solidify this.
What's the difference between valence electrons and core electrons?
Valence electrons are the outermost electrons of an atom—the ones in the highest principal energy level—and they’re the ones that interact in bonding, determine typical ionic charges, and control reactivity (think alkali metals giving 1 e–, halogens accepting 1 e–). Core electrons are all the inner electrons (those in filled lower-energy shells); they don’t participate in bonding but they shield valence electrons from the nucleus and affect properties like ionization energy and atomic radius. Example: neutral Na has electron configuration 1s2 2s2 2p6 3s1—the single 3s electron is the valence electron; the 1s–2p electrons are core. On the AP exam, you’ll use this idea to predict ionic charges, explain periodic trends, and justify reactivity (CED 1.8.A, keywords: valence electrons, cation/anions, ionization energy). For a focused review, see the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try related practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about how to predict the charge of ions - can someone explain?
Think of ion charge as “how many electrons an atom will lose or gain to get a full valence shell (an octet).” Look at the group (column) on the periodic table and count valence electrons: - Group 1 (alkali): 1 valence → lose 1 → +1 (e.g., Na+) - Group 2 (alkaline earth): 2 → lose 2 → +2 (e.g., Ca2+) - Group 16: 6 → gain 2 → −2 (e.g., O2−) - Group 17 (halogens): 7 → gain 1 → −1 (e.g., Cl−) For main-group elements, the typical ionic charge ≈ group number (for metals) or group number −8 (for nonmetals). Transition metals can have multiple charges (use given info or look up common oxidation states). This prediction follows the octet rule and the CED Essential Knowledge 1.8.A.3 (valence electrons govern typical ionic charges). Want practice? Review the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try problems at Fiveable’s unit page (https://library.fiveable.me/ap-chemistry/unit-1) or the practice bank (https://library.fiveable.me/practice/ap-chemistry).
How do valence electrons determine if two elements will bond together?
Valence electrons control bonding because they’re the ones atoms can lose, gain, or share. Atoms with 1–3 valence electrons (like alkali/alkaline earth metals) tend to lose electrons (low ionization energy) and form cations; atoms with 5–7 valence electrons (like halogens) tend to gain electrons (high electron affinity) and form anions. When one element easily gives electrons and the other easily accepts them, an ionic bond forms (Na → Na+ and Cl + e– → Cl– → NaCl). If both atoms have similar electronegativities and unpaired valence electrons, they share electrons to satisfy octets and make covalent bonds (C, O, H). You can predict typical ionic charges from periodic table location (CED 1.8.A.1 & 1.8.A.3) and expect elements in the same column to form analogous compounds (CED 1.8.A.2). For a quick AP-aligned review, see the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try practice problems on Fiveable (https://library.fiveable.me/practice/ap-chemistry).
Why do metals tend to lose electrons while nonmetals tend to gain them?
Metals tend to lose electrons because they have few valence electrons (usually 1–3) held relatively loosely—low ionization energy—so losing electrons to reach a full shell (the octet) is energetically favorable. Nonmetals have more valence electrons (usually 4–7), higher electronegativity, and often high (more negative) electron affinity, so gaining electrons to complete an octet releases energy and is favored. Periodic trends explain this: left-side elements (alkali, alkaline earth metals) form cations (+1, +2) easily; right-side elements (halogens, oxygen group) form anions (−1, −2) by gaining electrons. These ideas tie directly to Topic 1.8 (valence electrons, ionic bonds, cations/anions) in the CED and show why ionic compounds form predictable charges. For extra review and practice, check the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and lots of practice questions (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when they say elements in the same column form analogous compounds?
It means elements in the same column (group) have the same number of valence electrons, so they tend to lose/gain the same number of electrons and make compounds with the same formulas and similar properties. For example, group 1 (alkali metals) all form M+ ions and make halides like NaCl, KCl, etc.; group 2 form M2+ and give MCl2 salts. Because valence electrons and typical ionic charges are predictable from group number, the bonding type (ionic), stoichiometry (formula), and general reactivity patterns are analogous across a column. This idea is exactly what the CED calls “periodicity” and links to reactivity and typical charges (1.8.A.1–1.8.A.3). For more examples and AP-style practice, see the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
How do I use the periodic table to predict ionic charges?
Use the periodic table to count valence electrons and apply the octet idea. For main-group elements, the group number tells you valence electrons (use the A groups): metals on the left lose electrons to reach an octet and become cations; nonmetals on the right gain electrons and become anions. Quick rules you should memorize for AP-style predictions: - Group 1 → +1, Group 2 → +2 - Group 13 → +3 (often), Group 14 → ±4 (C usually covalent; heavier metals may be +2 or +4) - Group 15 → −3, Group 16 → −2, Group 17 (halogens) → −1 Transition metals have variable charges—use common oxidation states (Fe²⁺/Fe³⁺, Cu⁺/Cu²⁺) or given clues. Remember this is tied to valence electrons, ionization energy, and electron affinity (CED 1.8.A.1–3). The AP exam gives a periodic table; practice predicting charges with the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and drill problems at (https://library.fiveable.me/practice/ap-chemistry).
Why are noble gases so unreactive compared to other elements?
Noble gases are unreactive because their valence shells are already full (He: 1s2; Ne, Ar, etc.: full octets). That means they don’t need to gain, lose, or share electrons to reach a lower-energy, stable state (the octet rule). Because removing an electron would break that stable configuration, noble gases have very high ionization energies; because they’re already stable, their electron affinities are near zero or even positive—they don’t strongly attract extra electrons. Together, these factors (valence electron configuration, ionization energy, and electron affinity) explain the low chemical reactivity you see across the rightmost column of the periodic table—a clear example of periodicity from the CED (1.8.A, 1.8.A.1). For more on valence electrons and ionic behavior, check the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain the relationship between electron configuration and reactivity?
Electron configuration tells you how many valence electrons an atom has and where they sit, and that directly controls reactivity. Atoms try to reach a stable (often octet) arrangement, so: - Metals (left side, e.g., alkali metals with 1 valence e–) lose electrons easily because low ionization energy → form cations and react strongly. - Nonmetals (right side, e.g., halogens with 7 valence e–) gain an electron readily because high electron affinity → form anions and are very reactive. Periodic trends matter: as you move down a group, valence electrons are farther from the nucleus (bigger ionic radius), so metals get more reactive; across a period, increasing nuclear charge raises ionization energy and lowers metallic reactivity. Predict ionic charges from valence electrons (e.g., Group 1 → +1, Group 17 → −1). These ideas link to ionization energy, electron affinity, electronegativity, lattice energy, and the octet rule in the CED (1.8.A). For extra practice and AP-style questions, check the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and the practice set (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how the number of valence electrons affects an atom's charge in ionic compounds.
Valence electrons set an atom’s “goal” for stability. Atoms with 1–3 valence electrons (metals) tend to lose them to get a filled shell and become cations (+ charge); atoms with 5–7 valence electrons (nonmetals) tend to gain electrons and become anions (− charge). The octet rule explains why: forming ions gives a noble-gas electron configuration. You can predict typical ionic charges from group number: Group 1 → +1, Group 2 → +2, Group 13 → +3; Group 17 → −1, Group 16 → −2, Group 15 → −3. Ionization energy (how hard it is to remove an electron) and electron affinity (how much an atom wants an extra electron) control whether loss or gain is favored. This is exactly what the CED wants you to explain for Topic 1.8 (1.8.A.3). For a quick refresher and examples, check the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and try practice problems at Fiveable (https://library.fiveable.me/practice/ap-chemistry).
What's the connection between an element's position on the periodic table and its typical ionic charge?
Your position in the periodic table basically tells you how many valence electrons an element has, and that predicts whether it tends to lose or gain electrons to reach an octet—which determines its typical ionic charge (CED 1.8.A, EK 1.8.A.3). Quick rules for main-group elements: - Group 1 → 1 valence e− → lose 1 → +1 - Group 2 → 2 valence e− → lose 2 → +2 - Group 13 → 3 valence e− → often +3 - Group 14 → 4 valence e− → often covalent (±4 less common as ions) - Group 15 → gain 3 → −3 - Group 16 → gain 2 → −2 - Group 17 (halogens) → gain 1 → −1 - Group 18 → full octet → typically 0 (inert) Ionic compounds are electrically neutral, so charges combine (e.g., Ca2+ + 2 Cl− → CaCl2). This connects to ionization energy, electron affinity, and lattice energy when predicting stability. For a focused review, check the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and more unit resources (https://library.fiveable.me/ap-chemistry/unit-1). For extra practice, try problems at https://library.fiveable.me/practice/ap-chemistry.
How do valence electrons and nuclear charge work together to determine bonding?
Bonding comes from a push–pull between valence electrons (the ones available to share or lose/gain) and the positive pull of nuclei (nuclear charge). If an atom has low ionization energy (weak hold on its valence electrons) and another has high electron affinity/electronegativity (strong pull on extra electrons), electrons will transfer and an ionic bond forms: the low-IE atom becomes a cation, the high-EA atom becomes an anion. If both atoms have similar electronegativities, they share valence electrons (covalent bonding) to achieve octets. Periodic trends matter: moving right increases effective nuclear charge and electronegativity, moving down increases atomic radius and lowers IE, so alkali metals readily form cations and halogens readily form anions (CED 1.8.A.1, 1.8.A.3). Lattice energy then determines ionic solid stability: smaller ions and higher charges → larger lattice energy → stronger ionic compounds. For more examples and exam-style practice, see the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1).
Why do some elements form multiple different ions with different charges?
Some elements form more than one stable ion because removing or adding different numbers of valence (and sometimes d) electrons can be energetically feasible depending on the atom and its environment. For main-group atoms, ions usually reflect reaching an octet (e.g., Ca2+), but if successive ionization energies aren’t hugely different an element can lose different numbers of electrons. Transition metals commonly show multiple charges because their ns and (n–1)d electrons have similar energies, so removing different combinations gives different stable oxidation states. Which ion actually forms depends on the balance of ionization energy, electron affinity, lattice/solvation energy, and the final electronic stability (octet, half-filled d subshells, etc.). AP relevance: this ties to 1.8.A (valence electrons predict ionic charges) and you might see questions asking you to justify likely charges using ionization energy and periodic trends. For extra practice and examples, check the Topic 1.8 study guide (https://library.fiveable.me/ap-chemistry/unit-1/valence-electrons-ionic-compounds/study-guide/XTtinEfGPR0jEJmpUuBx) and AP practice sets (https://library.fiveable.me/practice/ap-chemistry).