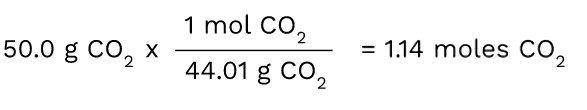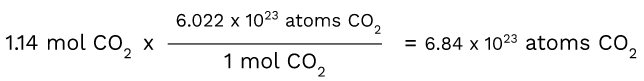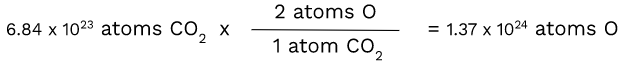Moles and molar mass are essential key concepts to understand when studying chemistry since they will allow you to accurately calculate and interpret chemical quantities and conversions. Think about it this way: how do scientists perform laboratory work when it's nearly impossible to count the atoms they are working with🤔?
The Atom
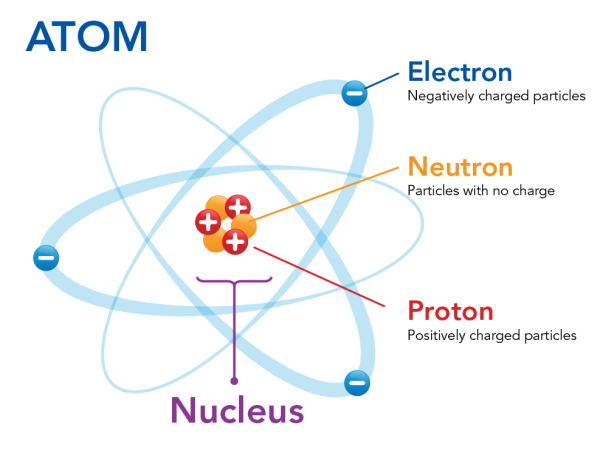
First, imagine an atom, the basic unit of matter that makes up all elements. Well, most likely you can't even begin to grasp how small an atom even is⚛️. An atom is made up of three types of subatomic particles: protons, neutrons, and electrons.

Protons and neutrons are located in the nucleus at the center of the atom, while electrons orbit the nucleus. The nucleus is a small, dense core at the center of the atom. Since protons have a + charge and neutrons are neutral, the nucleus is very overall very positive. Electrons, orbiting the nucleus, have a negative charge and counteract the positive center of the atom.
We'll discuss the atom in more depth later in this unit, but it is important to understand how small it is. This is where the concept of a mole emerged.
What is a mole?
Since scientists cannot count the tiny particles and atoms they are experimenting with, there must be a correlation between the mass of substances involved in a chemical reaction and the number of particles undergoing change. This is exactly what the mole is!
A mole relates the mass of an element to the number of particles there are. The analogy between a mole and a dozen of eggs can be helpful in understanding the concept of a mole in chemistry. Just as a dozen is a unit of measurement for eggs, a mole is a unit of measurement for particles in a substance.
Molar Mass of a Substance
What is molar mass?
The molar mass of a substance is the number of grams there are in a mole. Hence, the units for molar mass are grams/mole (or g/mol). Molar mass is important because it allows us to convert between mass, moles, and the number of particles.
Finding the molar mass of an element or compound is not as hard as it might seem: the only things that you need to know are which elements are involved and how many of them are present. This is also where the periodic table of elements comes in.
On the periodic table, each element is represented by a one-to-two-letter abbreviation. You can also see a number above and below each chemical symbol. The number above, going chronologically across the periodic table, is the atomic number. The atomic number represents the number of protons in the nucleus of an atom of that element. The number below each symbol is the element's atomic mass. This is the mass of one atom of the element in atomic mass units (amu).
👉 Cool Interactive Periodic Table
This is the periodic table that will be provided for you during the AP Chemistry Exam. You may access it online here.How do I calculate molar mass?
Let's first calculate the molar mass of water (H2O). First, break down the compound of interest. In one molecule of water, we have 2 atoms of hydrogen and 1 atom of oxygen.
From here, we have to take a look at the periodic table and find out how much each atom of hydrogen and oxygen weighs. This is where we have to take a look at the atomic mass of an element. The atomic mass of hydrogen is 1.008 g/mol and the atomic mass of oxygen is 16.00 g/mol. Since there are two atoms of hydrogen and one atom of oxygen in water, we must multiply 1.008 by 2, and then add that product to 16.00. This is how you can calculate the molar mass of water: 18.02 g/mol! 🥳
Calculating Molar Mass: Carbon Dioxide
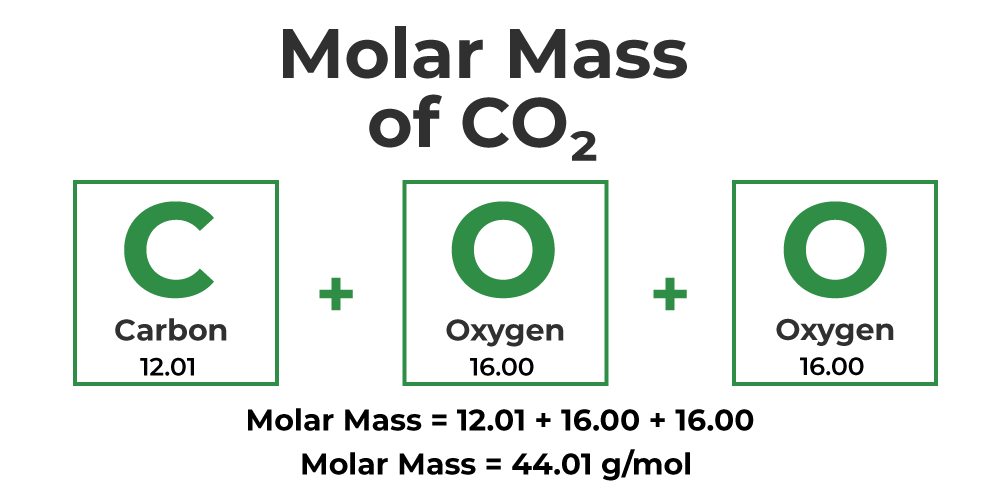
Let's say we now have the compound CO2 or carbon dioxide. Remember, to calculate the molar mass, you simply have to multiply the atomic mass of each specific element by its subscript, and then add it all together.
Carbon has a subscript of 1 and an atomic mass of 12.01 grams according to the periodic table. Oxygen has a subscript of 2 in this compound and has an atomic mass of 15.99 grams. Always multiply the subscript by the atomic mass of the element:
Carbon: 1 x 12.01 = 12.01 g
Oxygen: 2 x 16.00 = 32.00 g
Finally, we add 32.00 grams to 12.01 grams to get 44.01 grams. Therefore, CO2 has a molar mass of 44.01 grams per mole.
Image Courtesy of GeeksforGeeksAvogadro's Number
Remember the analogy between a mole and a dozen? Just as a dozen eggs contains 12 eggs, a mole of a substance contains 6.022 x 10^23 particles. This very large number is Avogadro's number. To put this into perspective, a mole of hockey pucks would be equal to the mass of the Moon. In chemistry, this number is relevant when converting from grams to moles to atoms.
Dimensional Analysis
One of the most fundamental takeaways from this unit is dimensional analysis, and you'll be using it throughout the rest of this course! This is a technique used to convert between different units of measurement, and you've probably implicitly done it before.
For example, you can use dimensional analysis to convert from miles per hour to meters per second, or from inches to centimeters.
Dimensional analysis is going to be so useful throughout this course, especially when you forgot a formula that is essential to solving the question!When doing dimensional analysis, you start by identifying the units you are trying to convert from and the units you want to convert to. Then, you write down the conversion factor that will allow you to make the conversion. Finally, you multiply the value you are trying to convert by the conversion factor to get the final result.
A conversion factor is a ratio of equivalent units that can be used to convert one set of physical units to another. These are usually known facts, such as 1 foot = 12 inches and 1 meter = 3.28 feet.
Dimensional Analysis with Moles and Molar Mass
Now that we've discussed the fundamental concepts of moles and molar mass, let's try converting a sample of 50.0 grams of CO2 between units.
CO2 - Converting from Grams to Moles
Since we know we have to convert from grams to moles, we have to figure out what conversion factor can help us do this. First, put the number that is given to you in the problem, which in this case, is 50.0 grams of CO2.
Then, you want to multiply 50.0 by the molar mass in order to convert it to the moles of CO2. The unit that you have (grams of CO2) should always be on the bottom of the next ratio in order for the units to cancel out. Here, the grams of CO2 cancel out and you are left with a measurement in moles.
Tip: It is good to memorize that moles = grams/molar mass. The conversion factor in this problem is actually using this concept since you are ultimately dividing the number of grams you have by the molar mass to get the number of moles.
CO2 - Converting from Moles to Atoms
Now let's convert 1.14 moles of CO2 into atoms using Avogadro's number.
Here, you are once again taking the number that you have and putting it first. Then, you are putting the unit of measurement that you want over the unit of measurement that you have, making that step the conversion factor. This enables the moles of CO2 to cancel out, leaving you with just 6.84x10^23 atoms of CO2.
Keep in mind that the concept of Avogadro's number serves as the conversion factor when going from moles to atoms.
CO2 - Finding the number of atoms of C
Since the subscript on Carbon is 1, the number of atoms of CO2 is equivalent to the number of carbon atoms in CO2. There is nothing to multiply by because of this 1-to-1 ratio; therefore the number of carbon atoms in this 50.0g sample of CO2 is 6.84x10^23.
CO2 - Finding the number of atoms of O
Unlike carbon, oxygen has a subscript of 2. This makes the ratio of CO2 atoms to oxygen atoms 1:2. Therefore, we have to use dimensional analysis again:
Since there are two atoms of O in one atom of CO2, we had to multiply by 2 to get the number of atoms of O.
You got this! Once you practice multiple problems involving dimensional analysis, it'll seem like a piece of cake. Sadly, these problems become more difficult as the course progresses but as always, practice makes perfect.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
atomic mass units | A unit of mass used to express the mass of individual atoms or molecules, where one amu is defined relative to carbon-12. |
Avogadro's number | The constant 6.022 × 10^23 mol^-1 that represents the number of particles (atoms, molecules, or formula units) in one mole of a substance. |
constituent particles | The individual atoms, molecules, or ions that make up a substance. |
dimensional analysis | A mathematical technique for converting between different units of measurement by using conversion factors and canceling units. |
formula unit | The smallest unit of a compound that shows the fixed proportions of atoms or ions held together. |
molar mass | The mass in grams of one mole of a substance, numerically equal to the average mass of one particle in atomic mass units. |
mole concept | A fundamental chemistry concept that relates the number of particles (atoms, molecules, or ions) to measurable quantities through Avogadro's number. |
Frequently Asked Questions
What is a mole in chemistry and why do we use it?
A mole is a counting unit in chemistry—1 mole = 6.022 × 10^23 representative particles (Avogadro’s number). We use it because atoms and molecules are tiny and unimaginably numerous, so you can’t count them one-by-one in the lab. The mole connects the number of particles to measurable mass: the average mass of one particle in amu is numerically equal to the molar mass in grams per mole, so n = m/M lets you convert mass ↔ moles ↔ number of particles with dimensional analysis (an AP-tested skill). Practically, you use moles to relate reactants and products in stoichiometry, find empirical formulas, and convert between grams and molecules for calculations on the exam. For a quick refresher and practice problems on this exact topic, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and the AP Chem practice bank (https://library.fiveable.me/practice/ap-chemistry).
How do you calculate molar mass from the periodic table?
Use the atomic masses on the periodic table (these are the average masses in amu and equal the molar mass in g·mol⁻¹). Steps: 1) For an element, read its atomic mass from the table (e.g., O ≈ 16.00, H ≈ 1.008). 2) For a compound, multiply each element’s atomic mass by its subscript and add them. Example: H2O → 2(1.008) + 16.00 = 18.016 g·mol⁻¹. 3) Use the molar mass in conversions with n = m/M (m in grams, M in g·mol⁻¹, n in mol). Remember Avogadro’s number (6.022×10^23 particles per mol) to convert moles ↔ particles. On the AP exam you’ll be given a periodic table and should report answers with correct units and significant figures. For a quick refresher, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about Avogadro's number - what does 6.022 x 10^23 actually represent?
Avogadro’s number (6.022 × 10^23 mol^-1) is simply the counting unit for particles in chemistry—like a dozen = 12, but for atoms/molecules. It tells you how many “representative particles” (atoms, molecules, or formula units) are in one mole. So 1.00 mol of carbon atoms = 6.022 × 10^23 C atoms. Its unit mol^-1 means “per mole,” so particles = moles × 6.022×10^23. This is the bridge the CED describes: you can’t count atoms directly in lab, so Avogadro’s number links mass to particle number via molar mass. Use n = m/M to go between grams and moles, then multiply/divide by Avogadro’s number for particles. Practice converting grams ↔ moles ↔ particles with dimensional analysis—that’s frequently tested on the AP (Topic 1.1). For extra practice and step-by-step examples, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between atomic mass units (amu) and grams per mole?
amu (atomic mass unit) and g·mol⁻¹ (grams per mole) are the same “number” but different contexts. - amu is a scale for the mass of a single atom or molecule: 1 amu = 1/12 the mass of a 12C atom ≈ 1.6605×10⁻²⁴ g. So an oxygen atom ≈ 16.00 amu. - g·mol⁻¹ (grams per mole) is the mass of one mole (6.022×10²³ particles) of those atoms or molecules. So O has a molar mass ≈ 16.00 g·mol⁻¹. Why both matter: the CED (1.1.A.3) notes that the average mass in amu of one particle is numerically equal to the molar mass in g·mol⁻¹. Use Avogadro’s number to move between the two: one particle’s mass (amu) × NA = molar mass (g·mol⁻¹). For calculations on the AP exam you’ll convert sample mass to moles using n = m / M (M in g·mol⁻¹). For extra practice and examples, check the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and more problems (https://library.fiveable.me/practice/ap-chemistry).
How do I use the formula n = m/M to solve mole problems?
n = m / M is your go-to for converting mass (g) to amount (mol). Steps: 1. Identify m (mass in grams) and M (molar mass in g·mol^-1). M comes from the periodic table (sum atomic masses for a molecule). 2. Plug into n = m / M. Units: g ÷ (g·mol^-1) → mol. 3. Use mols to get particles: N = n × NA (NA = 6.022×10^23 mol^-1). Quick example: 25.0 g H2O. M = 2(1.01) + 16.00 = 18.02 g·mol^-1. n = 25.0 g / 18.02 g·mol^-1 = 1.39 mol (3 sf). Particles = 1.39 mol × 6.022×10^23 mol^-1 = 8.36×10^23 molecules. Tips for AP: always show units (dimensional analysis), keep proper sig figs, and when asked, convert moles to particles or to volumes (use ideal gas law) as required by the CED learning objective 1.1.A. For more review and practice problems, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and Fiveable’s practice set (https://library.fiveable.me/practice/ap-chemistry).
Why can't we just count atoms directly in the lab instead of using moles?
You can't practically count atoms in the lab because individual atoms are unbelievably small and numerous—a single mole is 6.022 × 10^23 particles (Avogadro's number), so even tiny samples contain enormous numbers of atoms. Instead we measure mass (grams) and use the mole as a bridge: n = m/M (m = mass, M = molar mass in g·mol⁻¹). The molar mass numerically equals the average particle mass in amu, so grams → moles → particles (× 6.022×10^23) gives the particle count without counting atoms directly (CED 1.1.A.1–1.1.A.3). This is exactly the scheme the AP expects you to use on calculations and free-response items (use dimensional analysis and the mole concept). Need more practice converting mass ↔ moles ↔ particles? Check the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain dimensional analysis for mole conversions step by step?
Dimensional analysis for mole work is just unit-canceling step-by-step. Start with what you have and what you want. Useful relationships: n = m / M (moles = mass ÷ molar mass), 1 mol = 6.022×10^23 particles (Avogadro’s number). Example 1—grams → moles: want moles of CO2 from 88.0 g CO2. Write 88.0 g CO2 × (1 mol CO2 / 44.01 g CO2) = 2.00 mol CO2. Units (g) cancel, leaving mol. Example 2—moles → particles: 2.00 mol CO2 × (6.022×10^23 molecules / 1 mol) = 1.20×10^24 molecules. Example 3—particles → grams: 3.01×10^22 atoms × (1 mol / 6.022×10^23 atoms) × (M g / 1 mol)—cancel atoms then mol, plug atomic/molar mass for M to get grams. Always write conversion factors as fractions, cancel units across numerator/denominator, and use correct significant figures. These steps map directly to CED Objective 1.1.A (mass-to-moles conversions). For more worked examples, check the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the connection between the mass of something and how many particles it has?
Mass tells you how many particles you have by using the mole. Convert mass (m, in grams) to moles (n) with n = m / M, where M is the molar mass (g·mol⁻¹). Then convert moles to particles with Avogadro’s number: particles = n × 6.022×10^23. So a sample’s average particle mass in atomic mass units (amu) is numerically equal to its molar mass in grams—that’s the bridge between single-particle mass and bulk mass. Example: 18.0 g of H2O → n = 18.0 g / 18.02 g·mol⁻¹ ≈ 1.00 mol → ≈ 6.02×10^23 water molecules. This is exactly the dimensional-analysis routine the AP exam expects (Topic 1.1; use n = m/M and NA). For a quick review, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I convert from grams to moles to atoms in one problem?
Do it in two clean conversion steps using n = m/M and Avogadro’s number. Start with mass → moles, then moles → particles. Step 1: grams → moles using n = m / M (m = sample mass, M = molar mass in g·mol⁻¹). Step 2: moles → atoms using N = n × NA (NA = 6.022 × 10^23 mol⁻¹). Example: 12.0 g C → how many atoms? - M(C) ≈ 12.01 g·mol⁻¹, so n = 12.0 g / 12.01 g·mol⁻¹ = 1.00 mol (3 s.f.). - Atoms = 1.00 mol × 6.022×10^23 mol⁻¹ = 6.02×10^23 C atoms. Tips for AP Chem (CED-aligned): always track units with dimensional analysis, show n = m/M, and state Avogadro’s number. Use correct sig figs and the periodic table’s molar mass (standard atomic weight). For extra practice see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and tons of problems at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why the atomic mass on the periodic table equals the molar mass - can someone explain?
Think of the atomic mass on the periodic table as the average mass of one atom measured in atomic mass units (amu). By definition, 1 amu = 1/12 the mass of a carbon-12 atom. Avogadro’s number (6.022×10^23) is the bridge between the microscopic and macroscopic: 6.022×10^23 particles weigh the same number in grams as a single particle’s mass in amu. So if an oxygen atom has an atomic mass ≈16.00 amu, one mole (6.022×10^23 atoms) of oxygen weighs 16.00 g—that’s the molar mass. This is why the average mass in amu of one particle is numerically equal to the molar mass in grams (CED 1.1.A.3). Use n = m/M (m = mass, M = molar mass) to convert between grams and moles on AP problems. For a concise review and more examples, check the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
What happens if I mess up units when doing mole calculations?
If you mess up units in mole work, your answer will usually be the wrong magnitude (like 10^3 too big/small) or the wrong quantity (you give grams when the question asked for moles or particles). AP problems test dimensional analysis and the n = m/M connection, so units must cancel across every conversion (grams ↔ moles using molar mass, moles ↔ particles using Avogadro’s number 6.022×10^23 mol^-1). Quick checklist: write units on every number, use conversion factors that cancel, check the final unit (should be mol, g, or particles as asked), and verify the order of magnitude (does your mole answer make sense given the mass?). The AP CED expects correct labeling of units and significant figures for “Calculate” tasks—losing units can lose you points. For more practice and worked examples, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and the AP Chem practice bank (https://library.fiveable.me/practice/ap-chemistry).
How do you find the number of molecules in a sample if you know the mass?
Use the mole as your bridge. Steps: 1) Convert mass (m) to moles (n) with n = m / M, where M is the molar mass in g·mol⁻¹ (sum of atomic masses from the periodic table). 2) Convert moles to particles using Avogadro’s number: number of molecules = n × NA, where NA = 6.022 × 10^23 mol⁻¹. Quick example: If you have 18.0 g H2O, M = 18.0 g·mol⁻¹ so n = 18.0/18.0 = 1.00 mol. Number of molecules = 1.00 × 6.022×10^23 = 6.022×10^23 molecules. This is exactly the mass-to-moles and particles conversion AP emphasizes (CED 1.1.A, Avogadro’s number, molar mass). For extra practice and worked examples see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and more problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to memorize Avogadro's number for the AP exam?
You should memorize Avogadro’s number because it’s the bridge between particles and lab-scale mass—and you’ll use it a lot on the exam. Avogadro’s number (NA = 6.022 × 10^23 mol^-1) converts moles ↔ number of representative particles, which is required by the CED learning objective 1.1.A (mass-to-moles and particle counting). It also ties to Essential Knowledge 1.1.A.3: the average mass in amu of one particle equals the molar mass in grams, so you’ll convert grams ↔ moles ↔ particles in many problems. The AP exam expects you to perform these dimensional-analysis calculations (Practice 5: Mathematical Routines), and that constant isn’t something you should rely on being given every time—so committing it to memory saves time and prevents setup errors. For quick review and worked examples, see the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and try practice problems on Fiveable (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between formula units and molecules when counting particles?
“Molecules” and “formula units” both mean “one representative particle,” but which term you use depends on the kind of substance. Use “molecule” for covalent (molecular) substances made of discrete bonded units (H2O, CO2). Use “formula unit” for ionic solids that don’t exist as discrete molecules but as a repeating lattice (NaCl, CaCO3); the formula unit is the simplest ratio of ions (Na+ : Cl− = 1:1). For counting with the mole concept you always convert between moles and particles using Avogadro’s number (6.022×10^23 particles/mol). So 1.00 mol H2O = 6.022×10^23 molecules H2O, and 1.00 mol NaCl = 6.022×10^23 formula units NaCl. Molar mass (g/mol) still equals the mass of one mole of those representative particles (CED keywords: mole, molar mass, formula unit, representative particle). For more practice and AP-aligned examples, see the Unit 1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and extra practice problems (https://library.fiveable.me/practice/ap-chemistry).
I missed the moles lab - how do you actually measure moles in real experiments?
You can’t count atoms directly, so labs measure moles by measuring something macroscopic and converting with n = m / M or with Avogadro’s number. Common AP-level methods: - Mass (gravimetric): weigh a pure sample or a precipitate, use its molar mass to find moles (n = m/M). - Titration: react a measured volume of solution with known concentration; moles = M·V and stoichiometry gives moles of the analyte. - Gas collection: measure gas volume, use ideal-gas law (PV = nRT) to get moles of gas produced. - Gravimetric stoichiometry: convert between reactant/product masses via mole ratios from a balanced equation. In AP problems you’ll use dimensional analysis, molar mass, and Avogadro’s number (6.022×10^23) to move between grams, moles, and particles. Practice these lab types and calculations on the Topic 1.1 study guide (https://library.fiveable.me/ap-chemistry/unit-1/moles-molar-mass/study-guide/U0wdfzbGXdkv2l1LVdqP) and try more problems at (https://library.fiveable.me/practice/ap-chemistry).