Types of Solids
Classifying solids by their bonding helps you predict physical properties like melting point, hardness, and electrical conductivity. The bonding type also determines the crystal structure a solid adopts, which connects directly to how we analyze these materials with X-ray diffraction and other techniques.
Ionic and Metallic Solids
Ionic solids form through electrostatic attraction between oppositely charged ions. In NaCl, for example, each ion is surrounded by ions and vice versa, creating an extended three-dimensional lattice. There are no discrete "molecules" in an ionic solid; the formula unit just gives you the simplest ratio of ions.
Metallic solids consist of metal atoms held together by metallic bonding, where valence electrons are delocalized across the entire lattice of positive metal cores. Think of it as a rigid framework of cations sitting in a "sea of electrons." This model explains why metals like Cu, Fe, and Al conduct electricity so well and why they're malleable: the electron sea can accommodate shifts in atom positions without breaking bonds.
Both ionic and metallic solids typically have high melting points, but for different reasons. Ionic solids require enough energy to overcome strong Coulombic attractions. Metallic solids require enough energy to disrupt the metallic bonding network. One key difference: ionic solids only conduct electricity when molten or dissolved (freeing the ions to move), while metals conduct in the solid state because their electrons are already mobile.
Covalent (Molecular) and Network Solids
These two categories are easy to confuse, so the distinction matters.
- Molecular solids are made of discrete molecules held together by intermolecular forces (van der Waals, dipole-dipole, or hydrogen bonding). Sugar () and ice () are classic examples. Because intermolecular forces are relatively weak compared to ionic or covalent bonds, molecular solids tend to have low melting points and are poor electrical conductors.
- Network (covalent) solids have atoms linked by covalent bonds in an extended three-dimensional framework with no individual molecules. Diamond (each C bonded tetrahedrally to four others) and quartz () are typical examples. Breaking these solids means breaking actual covalent bonds, so their melting points are extremely high (diamond sublimes above 3500 °C).
The quick test: if you can identify discrete molecular units, it's a molecular solid. If the covalent bonding extends continuously through the crystal, it's a network solid.
Alloys and Composite Materials
Alloys are solid mixtures of two or more metallic elements (or a metal with a small amount of a nonmetal). They're important because you can tune mechanical and chemical properties by changing composition.
- Substitutional alloys replace some host metal atoms with atoms of a similar size. Bronze (Cu + Sn) is a classic example. The substituting atoms must have a similar atomic radius for this to work well (the Hume-Rothery rules give the criteria).
- Interstitial alloys incorporate smaller atoms (often nonmetals like C or N) into the gaps between larger metal atoms. Carbon steel is the most common example: small C atoms fit into the interstices of the Fe lattice, dramatically increasing hardness and strength.
Alloys often outperform pure metals. Stainless steel resists corrosion far better than pure iron, and brass is harder than either pure copper or pure zinc.
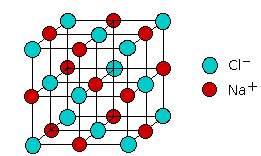
Ionic Solid Properties
Lattice Energy and Crystal Structure
Lattice energy () is the energy required to completely separate one mole of a solid ionic compound into its gaseous ions. It's the single best measure of how strongly an ionic solid is held together.
Two factors dominate lattice energy:
- Ion charge: Higher charges mean stronger Coulombic attraction. (with and ions) has a much larger lattice energy than (with and ions).
- Ionic radius: Smaller ions pack more closely, increasing the electrostatic interaction. has a higher lattice energy than because both ions in LiF are smaller.
Higher lattice energy correlates with higher melting points and greater thermodynamic stability. The crystal structure an ionic solid adopts depends on the radius ratio (), which determines how many anions can geometrically fit around a cation (and vice versa).
Madelung Constant and Electrostatic Interactions
The Born-Landé equation gives a more quantitative treatment of lattice energy:
The Madelung constant () in this equation accounts for the geometry of the crystal. It sums up all the attractive and repulsive Coulombic interactions between every ion pair in the lattice, not just nearest neighbors. Its value depends on the crystal structure type:
- NaCl structure:
- CsCl structure:
- Zinc blende (ZnS) structure:
A larger Madelung constant means the particular geometric arrangement of ions produces stronger net electrostatic stabilization. You don't need to derive yourself, but you should understand that it captures how the specific packing geometry affects overall lattice energy beyond just the nearest-neighbor interaction.

Coordination Number and Packing Efficiency
The coordination number (CN) is the number of nearest-neighbor ions surrounding a given ion in the crystal.
| CN | Geometry | Example Structure |
|---|---|---|
| 4 | Tetrahedral | Zinc blende (ZnS) |
| 6 | Octahedral | Rock salt (NaCl) |
| 8 | Cubic | CsCl |
The radius ratio () predicts which coordination number is favorable. Larger cations relative to anions can accommodate more neighbors, so they tend toward higher coordination numbers. For instance, is large enough relative to to fit 8 anions around it, while the smaller fits only 6.
Packing efficiency describes the fraction of space in the unit cell actually occupied by ions. Higher packing efficiency generally means greater density and stability. The CsCl structure, despite its higher CN, doesn't always win on packing efficiency because the ions involved have different size ratios than those in the NaCl structure. The "best" structure for a given compound is whichever one maximizes lattice energy given the specific ion sizes and charges.
Metallic Solid Properties
Delocalized Electrons and Electrical Conductivity
The "electron sea" model treats a metal as a lattice of cations immersed in a delocalized cloud of valence electrons. This simple picture explains several key metallic properties:
- Electrical conductivity: Delocalized electrons move freely in response to an applied voltage. Metals conduct in the solid state (unlike ionic solids, which need to be melted or dissolved).
- Thermal conductivity: Mobile electrons transfer kinetic energy efficiently, making metals good heat conductors.
- Malleability and ductility: When the lattice is deformed, layers of cations can slide past each other without breaking bonds because the electron sea simply redistributes. In an ionic solid, shifting ions brings like charges together, which causes the crystal to shatter.
For a more rigorous treatment, band theory replaces the electron sea model. In band theory, atomic orbitals combine to form continuous energy bands. The partially filled bands in metals allow electrons to move freely, which is the quantum mechanical basis for conductivity.
Closest Packing and Crystal Structures
Metals tend to adopt structures that maximize the number of nearest neighbors and minimize empty space. The three most common metallic crystal structures are:
- Face-centered cubic (FCC): Atoms at each corner and each face center of the cube. CN = 12. Packing efficiency = 74%. Examples: Cu, Al, Ag, Au.
- Hexagonal close-packed (HCP): Alternating layers in an ABAB stacking pattern. CN = 12. Packing efficiency = 74%. Examples: Mg, Zn, Co, Ti.
- Body-centered cubic (BCC): Atoms at each corner plus one atom in the center of the cube. CN = 8. Packing efficiency = 68%. Examples: Fe (at room temperature), Cr, W, Na.
FCC and HCP are both closest-packed structures, meaning they achieve the theoretical maximum packing efficiency for identical spheres (74%). The only difference between them is the stacking sequence: FCC follows an ABCABC pattern, while HCP follows ABAB.
BCC is less efficiently packed but is still common because other factors (like the electronic structure of the metal) can favor it. Iron at room temperature is BCC, but it transitions to FCC at 912 °C, which is a real-world example of how packing preference can change with conditions.