Redox Reactions
Oxidation and Reduction Processes
Redox reactions are paired processes: one species loses electrons (oxidation) while another gains them (reduction). You can't have one without the other. Splitting these into half-reactions is the standard approach for balancing complex redox equations and for analyzing electrochemical cells.
- Oxidation: loss of electrons, increasing the oxidation state. Occurs at the anode.
- Reduction: gain of electrons, decreasing the oxidation state. Occurs at the cathode.
A useful mnemonic: OIL RIG (Oxidation Is Loss, Reduction Is Gain). Half-reactions let you track electron flow explicitly, which becomes essential once you start working with electrode potentials.
Oxidation States and Electron Transfer
Oxidation states track how electron density is distributed in a compound. They're assigned based on electronegativity differences and bonding rules (e.g., oxygen is typically , fluorine is always ). For most elements, oxidation states range from about to , though transition metals can access a wide range depending on their d-electron configuration.
The core principle: electrons move from the reducing agent (the species being oxidized) to the oxidizing agent (the species being reduced). The total electrons lost must equal the total electrons gained.
Redox reactions show up everywhere:
- Combustion: hydrocarbons oxidized by
- Corrosion: iron oxidized to in the presence of water and oxygen
- Biological systems: electron transport chains in photosynthesis and cellular respiration
Electrochemical Cells
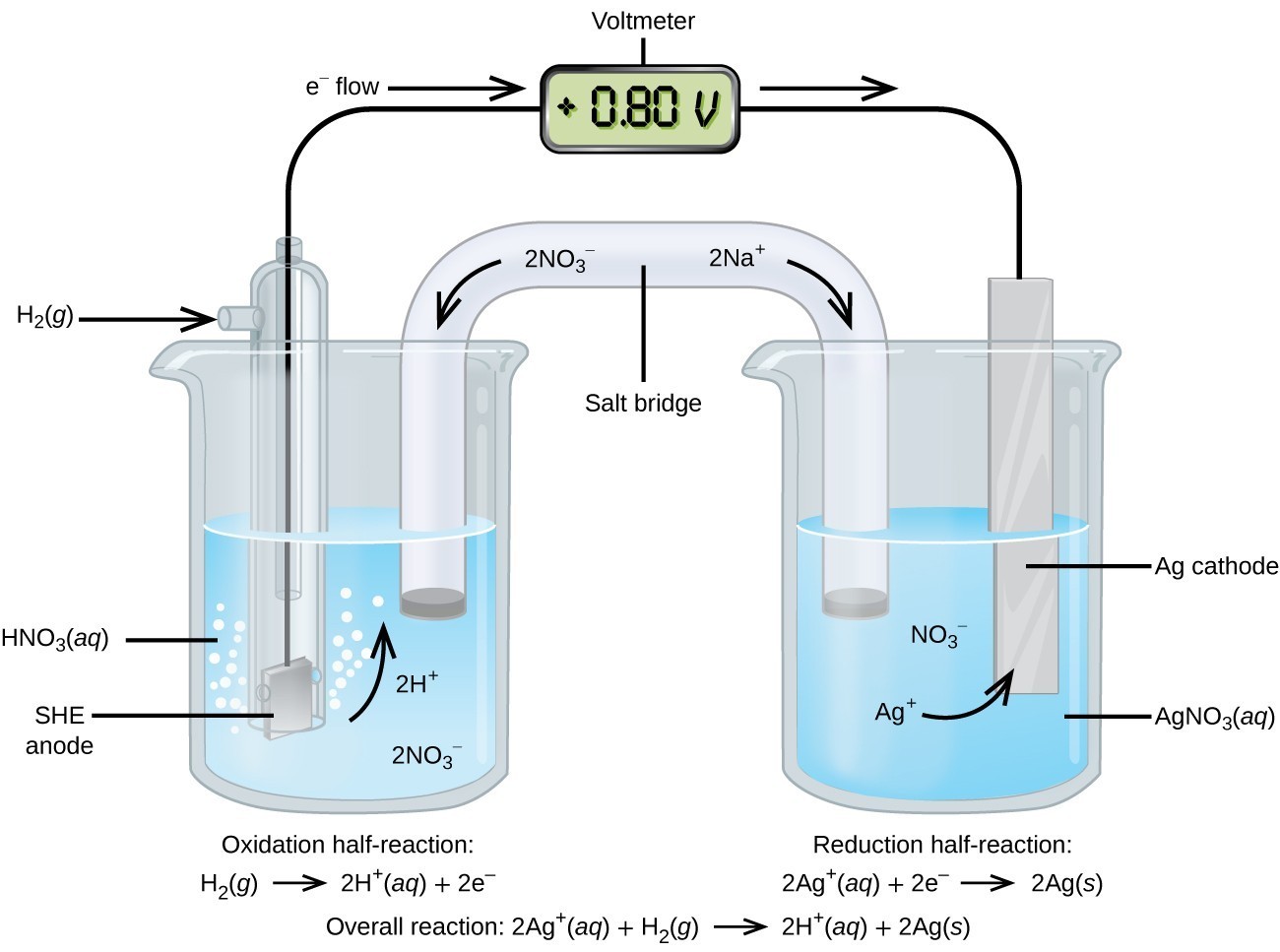
Components and Structure
An electrochemical cell converts chemical energy to electrical energy (or the reverse). Every cell has the same basic architecture:
- Two half-cells, each containing an electrode immersed in an electrolyte solution.
- An external circuit connecting the electrodes, through which electrons flow.
- A salt bridge (or porous barrier) connecting the two solutions internally.
The anode is where oxidation occurs. It releases electrons into the external circuit. The cathode is where reduction occurs. It accepts electrons from the external circuit.
Watch the sign conventions carefully, because they flip between cell types:
| Galvanic Cell | Electrolytic Cell | |
|---|---|---|
| Anode | Negative () | Positive () |
| Cathode | Positive () | Negative () |
The salt bridge maintains electrical neutrality by allowing ions to migrate between the half-cells. Without it, charge would build up in each compartment and current would stop almost immediately. It typically contains a concentrated electrolyte like or in a gel.
Types of Electrochemical Cells
Galvanic (voltaic) cells produce electricity from spontaneous redox reactions. The reaction has a positive , so electrons flow from anode to cathode through the external circuit on their own. Batteries and fuel cells are practical examples.
Electrolytic cells use an external power source to drive non-spontaneous redox reactions (negative ). Applications include electroplating, electrolytic refining of metals (e.g., copper purification), and industrial production of chemicals like and from brine.
The key distinction: galvanic cells produce electrical work; electrolytic cells consume it.
Standard Reduction Potentials

Standard Hydrogen Electrode and Reference
Since you can't measure the absolute potential of a single half-cell, all reduction potentials are measured relative to the standard hydrogen electrode (SHE). The SHE is defined as having .
The SHE setup:
- A platinum electrode in contact with solution
- gas bubbled over the electrode at 1 atm
- The half-reaction:
A standard reduction potential () quantifies how strongly a species tends to gain electrons under standard conditions (1 M concentrations, 1 atm partial pressures, 25°C). More positive means a stronger tendency to be reduced, meaning the species is a stronger oxidizing agent.
For example, has (very strong oxidizing agent), while has (very weak oxidizing agent, meaning metal is a powerful reducing agent).
Electrochemical Series and Predictions
The electrochemical series ranks half-reactions from most negative (top, least tendency to be reduced) to most positive (bottom, greatest tendency to be reduced). This ranking lets you make several predictions:
- Spontaneity: A species lower in the series (more positive ) will oxidize a species higher in the series.
- Oxidizing agent strength: Increases going down the series (toward more positive ).
- Reducing agent strength: Increases going up the series (toward more negative ).
- Metal displacement: A metal with a more negative will displace a metal ion with a more positive from solution. For instance, () displaces () from solution.
To calculate the standard cell potential:
A positive means the reaction is spontaneous under standard conditions. Note that you do not multiply values by stoichiometric coefficients. Reduction potentials are intensive properties.
Cell Potential and Nernst Equation
Cell Potential and Gibbs Free Energy
The cell potential () is directly related to the thermodynamic favorability of the reaction through Gibbs free energy:
where:
- = number of moles of electrons transferred in the balanced equation
- = Faraday's constant ()
- = standard cell potential in volts
The signs work out neatly: a positive gives a negative , confirming the reaction is spontaneous. This equation also connects electrochemistry to the equilibrium constant through , giving you:
So a large positive corresponds to a very large equilibrium constant, meaning the reaction strongly favors products.
Nernst Equation and Non-Standard Conditions
Real cells rarely operate at standard conditions. The Nernst equation adjusts the cell potential for actual concentrations (or pressures):
where is the reaction quotient. At 25°C, this simplifies to a commonly used form:
How to apply the Nernst equation:
- Write the balanced overall cell reaction.
- Identify (electrons transferred) and calculate .
- Write the expression for using actual concentrations/pressures.
- Plug into the equation and solve.
At equilibrium, and , which is how you can extract equilibrium constants from electrochemical data.
Concentration cells are a direct application of the Nernst equation. Both half-cells have the same electrode and the same redox couple, but at different concentrations. Since (identical half-reactions), the entire driving force comes from the concentration difference. Electrons flow from the dilute side (anode) to the concentrated side (cathode), and the cell operates until concentrations equalize.