Quantum Mechanics: Where is the Electron?
In the early days of chemistry, there were models of the atom all over the place.
The first true model of an "atom" was the Dalton Model, which represented atoms as small indivisible and indestructible balls bouncing off of each other. Of course, as you've learned in unit one, atoms aren't quite so simple.
The next model that came was Thomson's Plum Pudding Model in the late 19th century, shortly after J.J. Thompson discovered the electron. This model represented the atom as a sea of positive charge with specks of negative charge - the electrons, like raisins in a plum pudding.

From this point on, scientists knew two things about the model of the atom: there's a positive part and a negative part. They kept observing that the current model of the atom did not properly explain the observed properties, so more experiments were done and the model kept altering.
From here, Rutherford conducted the gold foil experiment in the early 20th century. He fired a beam of positive alpha particles at a thin sheet of gold foil and measured their deflections. His results were that most of the alpha particles passed straight through the foil, but some deflected at large angles.
Rutherford concluded that the positive charge in an atom is concentrated in a small, dense nucleus at the center of the atom, surrounded by a lot of empty space and a cloud of electrons. The biggest shock at the time was how his gold foil experiment showed that most of the atom was empty space.
⭐ While you don't need to necessarily know all of this history and all of these models for the AP Exam, understanding the foundations of chemistry help to build a foundation for the fundamental question for all of these models, and the question that created the field of quantum mechanics: where the heck do the electrons go?
While we won't exactly answer that exact question in this unit, it will be answered in part when you learn about electron orbitals.
Max Planck and the Ultraviolet Catastrophe
To understand where the electrons are in an atom, we must first understand how light and energy work together to form the basis for the quantum world. As we learned in the last lesson, light can be represented as a wave of electromagnetic radiation permeating space.
One of the most important observations made was that when a piece of metal, or any substance for that matter, was heated up enough, it released light. This makes sense! If you think about someone like a blacksmith, they take glowing hot metal and pound it into shape.
When discussing why exactly this happens, scientists hypothesized about a body that absorbed all light, known as a blackbody. It only absorbed and emitted light; it did not reflect any. However, when discussing light being radiated from a blackbody, it became clear that with this model, a body emits infinite amounts of light and thus infinite amounts of energy, creating the ultraviolet catastrophe. This theory broke laws of conservation of energy and frankly logic. When you put toast in a toaster, it does not burn your toast to smithereens, despite, according to the classical model, a blackbody should emit infinite energy.
The Ultraviolet Catastrophe. Image Courtesy of MSE 5317Max Planck to the Rescue!
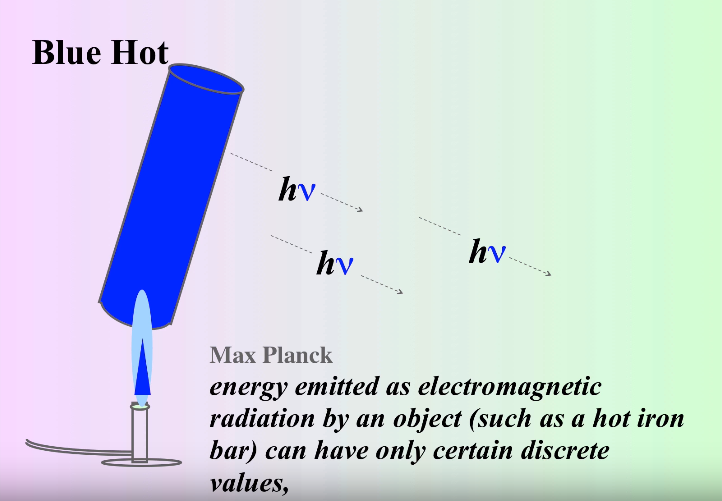
However, shortly after this catastrophe, a physicist named Max Planck solved the puzzle. He theorized that light is emitted in discrete quantities - quanta (hence, quantum mechanics), in which energy is proportional to the frequency of the wave.
This can be represented in the equation E = hν, where E is the energy, ν is the frequency and h is Planck's constant.
h = Planck's constant = 6.626 * 10^-34 Js (Joule seconds) - on the AP reference table
This equation helped form the basis for the eventual creation of quantum mechanics.
Quantized Light, Image Courtesy of MisterChemistryThe Photoelectric Effect
However, Planck's discovery was not immediately seen as revolutionary. It was not clear how amazing this discovery was until Albert Einstein uncovered the Photoelectric Effect, yet another mystery in physics.
Experiments showed that electrons were ejected from the surface of certain metals exposed to light of a minimum threshold frequency (and thus, as we've discussed, a minimum energy). Essentially, if you take a high enough frequency of light, you can zap electrons off of the surface of a metal, and this boggled the minds of physicists. Einstein theorized that this was Planck's theory at work.
He theorized that light, in this case, was working like a particle - photons. Mathematically, this can be represented as saying hv = KE of the electron + binding energy of the electron.
Because we know the hν is the energy of a wave of light (or now a photon), we know that if the energy of a photon exceeds the binding energy of an electron, the electron will be zapped away. Then, once we reach the threshold frequency, the kinetic energy of the photon increases as we lower the wavelength (or up the frequency, same thing) of the light.
Image Courtesy of MisterChemistryTo put it in simpler words, the photoelectric effect only occurs if the frequency of light reaches a certain threshold:
- If the frequency is low, the metal absorbs the light. It is not high enough to reach the threshold and therefore does not exhibit the photoelectric effect.
- If the frequency is high enough and reaches the threshold, a specified number of electrons are ejected from the metal.
🎥 Watch AP Chemistry teacher Mónica Gracida teach the photoelectric effect and review the concepts of wavelength, frequency, and energy of a wave.
Speed of Light
There is one more equation to keep in mind besides E=hν and it relates the speed of light to both wavelength and frequency. As we discussed in the last key topic, frequency and wavelength are inversely related: c = λν.
c = speed of light = 2.998 x 10^8 m/s
This equation is usually used in combination with the previous one. Together, they can help you solve a free-response question (FRQ) on the exam. Luckily, they are both listed on the AP Chemistry reference table. If you'd like to solve a previous AP FRQ, check out the next study guide and key topic.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
absorbed photon | A photon taken in by an atom or molecule, increasing the energy of the species by an amount equal to the photon's energy. |
electromagnetic wave | A wave composed of oscillating electric and magnetic fields that travels at the speed of light and carries energy related to its frequency. |
electronic transition | The movement of an electron between different energy levels in an atom or molecule, which occurs when a photon is absorbed or emitted. |
emitted photon | A photon released by an atom or molecule when an electron transitions to a lower energy level, decreasing the energy of the species. |
frequency | The number of wave cycles that pass a point per unit time, represented by the symbol ν, related to wavelength and the speed of light. |
photon | A discrete packet of electromagnetic energy with properties related to the frequency and wavelength of light. |
Planck's constant | The fundamental constant (h) that relates the energy of a photon to its frequency in Planck's equation. |
Planck's equation | The relationship E = hν that describes how the energy of a photon is proportional to its frequency, where h is Planck's constant. |
speed of light | The constant velocity at which electromagnetic radiation travels, represented by the symbol c, equal to approximately 3.00 × 10⁸ m/s. |
wavelength | The distance between successive peaks of an electromagnetic wave, represented by the symbol λ. |
Frequently Asked Questions
What is a photon and how does it relate to light?
A photon is a single packet (quantum) of electromagnetic energy—basically the “particle” of light. Its energy is proportional to the light’s frequency by Planck’s equation, E = hν (h = 6.626×10⁻³⁴ J·s). Wavelength, frequency, and the speed of light are related by c = λν, so higher-frequency (shorter-wavelength) photons carry more energy. In atoms or molecules, absorption of a photon raises the species from a lower (ground) to a higher (excited) energy level by exactly the photon’s energy; emission does the reverse (CED 3.12.A.1, 3.12.A.2). That quantization explains discrete absorption and emission lines and underlies the photoelectric effect. For AP-style work, be ready to use E = hν and c = λν to convert between wavelength, frequency, and photon energy on free-response or multiple-choice questions. For a quick review and practice problems on this topic, see the Topic 3.12 study guide (Fiveable) (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY), the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3), and thousands of practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why do atoms absorb and emit photons?
Atoms absorb or emit photons because their electrons occupy quantized energy levels. When an electron jumps from a lower level to a higher one it must gain a specific amount of energy ΔE—that energy comes from an absorbed photon (ΔE = hv). When an electron falls to a lower level it loses that same ΔE and emits a photon with frequency v (and wavelength λ given by c = λv). Because only certain energy differences exist, only photons with specific energies (frequencies/wavelengths) are absorbed or emitted, producing discrete absorption or emission lines. This is exactly what the CED wants you to explain for 3.12.A: photon energy equals the change in the atom’s energy (E = hv), and wavelength/frequency follow from c = λν. For a clear study walkthrough, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY). For broader unit review and lots of practice problems, check the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3) and the practice bank (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about the relationship between wavelength, frequency, and energy - can someone explain?
Wavelength (λ), frequency (ν), and photon energy (E) are tightly linked by two CED equations: c = λν and E = hν (Topic 3.12, 3.12.A). Because c (3.00×10^8 m/s) is constant, longer λ means smaller ν (they’re inversely related). And since E = hν (h = 6.626×10^-34 J·s), smaller ν means lower photon energy. So: longer wavelength → lower frequency → lower energy; shorter wavelength → higher frequency → higher energy. Quick example: a 500 nm (5.00×10^-7 m) photon has E = hc/λ ≈ (6.626×10^-34 J·s)(3.00×10^8 m/s)/(5.00×10^-7 m) ≈ 3.98×10^-19 J. On the AP, use these equations to relate absorbed/emitted photons to electronic transitions (energy change of the atom equals the photon energy). For a focused study, check the AP Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and practice problems (https://library.fiveable.me/practice/ap-chemistry) to drill calculations and exam-style questions.
What's the difference between absorption and emission of photons?
Absorption: an atom or molecule takes in a photon whose energy exactly equals the gap between two quantized energy levels. The species’ energy increases (electron moves from a lower to a higher level, e.g., ground → excited). Emission: an excited species releases a photon when an electron falls to a lower level; the species’ energy decreases by the photon energy. In both cases Ephoton = hν and c = λν, so higher-energy transitions give higher ν (smaller λ). On spectra, absorption produces dark lines (missing wavelengths) where photons were taken; emission gives bright lines at the wavelengths emitted. Remember: photons aren’t continuous—transitions are quantized, so only photons with energies matching level differences are absorbed or emitted (CED 3.12.A, E = hν). For a quick review, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and test these ideas with practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I use the equation E = hv in problems?
Use E = hν to find the energy of one photon: h is Planck’s constant (6.626×10^−34 J·s) and ν (nu) is frequency in s^−1. Often you’re given wavelength (λ), so combine with c = λν to get E = hc/λ. Steps you’ll use on the AP: 1. Convert λ to meters (1 nm = 1×10^−9 m). 2. Plug into E = hc/λ with c = 3.00×10^8 m/s and h = 6.626×10^−34 J·s. Result is joules per photon. 3. If you need kJ/mol, multiply by Avogadro’s number (6.022×10^23 mol^−1) and divide by 1000. Quick example: for λ = 500 nm, E = (6.626×10^−34)(3.00×10^8)/(5.00×10^−7) ≈ 3.97×10^−19 J per photon → ×6.022×10^23 /1000 ≈ 239 kJ/mol. On the exam, use E = hv to relate absorbed/emitted photon energy to ΔE between quantized levels (CED 3.12.A). For extra practice, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and more problems (https://library.fiveable.me/practice/ap-chemistry).
What happens to an atom when it absorbs a photon?
When an atom absorbs one photon, the atom’s energy increases by exactly the photon’s energy (E = hν). If that energy matches the difference between two quantized electronic levels, an electron jumps from a lower (usually ground) level to a higher excited level. If the photon energy is larger than any available bound-state gap, the electron can be ejected (photoionization) and the atom becomes an ion. Later the excited electron often relaxes back down, emitting photons with energies equal to the level differences (atomic emission lines). Use c = λν and E = hν (h ≈ 6.63×10⁻³⁴ J·s) to connect wavelength, frequency, and energy. This is exactly what AP Learning Objective 3.12.A covers—know that absorption raises the species’ energy by the photon’s energy and that spectra come from quantized transitions. For a quick review, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain electronic transitions in simple terms?
Think of an atom like a building with floors (energy levels). An electron normally sits on the lowest floor (ground state). When it absorbs a photon, that photon’s energy (E = hν) exactly equals the energy gap between its current floor and a higher one, so the electron jumps up (excited state). When an electron falls back down, it emits a photon whose energy equals the drop in energy. Because E = hν and c = λν, bigger energy gaps → higher frequency → shorter wavelength (bluer light); smaller gaps → longer wavelength (redder light). These jumps are quantized—only specific photon energies match allowed transitions—which gives discrete absorption and emission lines (what the AP CED wants you to explain, LO 3.12.A). For more practice and visual examples, check the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does the energy of a photon equal hv and what does each variable mean?
E = hν comes from the fact that light is quantized: electromagnetic energy is carried in packets (photons), and each packet’s energy is proportional to its frequency. Planck introduced the constant h (6.626×10^-34 J·s) to fit observed blackbody and photoelectric data, so E (energy of one photon) = h·ν (frequency). Physically, when an atom absorbs or emits a photon, the species’ energy changes by ΔE = E(photon)—so ΔE = h·ν (CED 3.12.A.1). You can also use c = λν (CED 3.12.A.2) to write E = hc/λ if you’re given wavelength instead of frequency. So: - E = photon energy (joules) - h = Planck’s constant (6.626×10^-34 J·s) - ν (nu) = frequency of the wave (s^-1 or Hz) - λ = wavelength (m) and c = speed of light (3.00×10^8 m/s). For more AP-aligned review and practice on the photoelectric effect and photon energy, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3). Practice problems available at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how photon energy relates to electronic transitions - help?
Think of an electronic transition as an electron jumping between quantized energy levels. If an atom or molecule absorbs a photon, the species’ energy increases by exactly the photon energy; if it emits a photon, its energy decreases by that same amount (CED 3.12.A.1). Quantitatively: ΔE = Efinal − Einitial = ±Ephoton and Ephoton = hν, with c = λν. So combine them: ΔE = h(c/λ). That means only photons with energy equal to the level gap (or the matching wavelength) can cause that transition—which is why spectra are lines, not a continuous smear. For a given transition, calculate ΔE from level energies or use ΔE = hc/λ to find the wavelength you’d see in absorption or emission. On the AP exam you may be asked to use E = hν and c = λν to connect transitions to specific wavelengths (Topic 3.12, Learning Objective 3.12.A). For a clear review and practice problems, check the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and the AP Chem practice bank (https://library.fiveable.me/practice/ap-chemistry).
What's the connection between the color of light and photon energy?
Color = photon energy. Use c = λν and E = hν (so E = hc/λ). Shorter wavelength (violet, ~400 nm) means higher frequency and higher energy; longer wavelength (red, ~700 nm) means lower energy. Numerically: E(400 nm) ≈ (6.63×10⁻³⁴·3.00×10⁸)/(4.00×10⁻⁷) ≈ 5.0×10⁻¹⁹ J (≈3.1 eV). E(700 nm) ≈ 2.8×10⁻¹⁹ J (≈1.8 eV). In atoms/molecules, a photon’s energy must equal the energy gap ΔE between quantized electronic levels: if Ephoton = ΔE the species absorbs (or emits) that wavelength. That’s exactly what the CED wants you to explain for 3.12.A (use E = hν and c = λν on the exam to calculate or compare photon energies). For more review and worked examples, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I calculate the energy of a photon if I know its wavelength?
Use c = λν and E = hν. Combine them to get the standard formula: E = hc / λ Where h = 6.626×10^−34 J·s and c = 3.00×10^8 m/s. Make sure λ is in meters. Quick steps: 1. Convert wavelength to meters (e.g., 500 nm = 500×10^−9 m). 2. Plug into E = (6.626×10^−34 J·s)(3.00×10^8 m/s) / λ. 3. Solve for E in joules per photon. If you want eV, divide by 1.602×10^−19 J/eV. Example: λ = 500 nm E = (6.626e−34)(3.00e8) / (500e−9) = 3.97e−19 J ≈ 2.48 eV. On the AP exam you should show the two equations (c = λν and E = hν), the algebra to combine them, units conversion, and the final units—that’s what graders look for. For more review on the photoelectric effect and photon energy, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do different elements emit different colors of light when heated?
Different elements emit different colors because their electrons occupy different sets of quantized energy levels. When an atom is heated, electrons jump to higher energy levels (excited states) and then fall back down; each electronic transition releases a photon with energy E = hν. Because E = hν and c = λν, different transition energies give different wavelengths (colors). Each element has a unique spacing of energy levels (due to nuclear charge and electron arrangement), so the allowed transition energies—and therefore the emission lines—are characteristic of that element (atomic emission spectrum). This is exactly what Topic 3.12 asks you to explain (LO 3.12.A using E = hν and c = λν). For extra review and practice on photons and the photoelectric effect, see the Topic 3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and more unit review (https://library.fiveable.me/ap-chemistry/unit-3). If you want more practice problems, check the AP practice bank (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when we say an electron "jumps" energy levels?
Saying an electron “jumps” energy levels means it changes from one quantized energy state to another—not a tiny planet hopping, but a quantum transition of its allowed energy (ground state → excited state or vice versa). That transition happens only when the atom absorbs or emits a photon whose energy exactly equals the energy difference between the two levels: ΔE = Efinal − Einitial = hv (Planck’s equation) and c = λν links wavelength to frequency. If the atom absorbs a photon, the electron moves to a higher (less negative) energy level; if it emits a photon, it falls to a lower level and releases hv. In modern terms the electron’s wavefunction changes instantly (probabilistically), so “jump” describes the discrete change, not a path through space. This is exactly what CED 3.12.A covers—relate transitions to photon energy (E = hv). For more review, check the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
How do absorption and emission spectra work and why are they important?
Absorption spectra happen when atoms or molecules absorb photons whose energy exactly matches the gap between quantized electronic levels. Emission spectra occur when excited species drop to lower levels and emit photons with E = hν (h = 6.63×10⁻³⁴ J·s), and c = λν relates wavelength to frequency (c ≈ 3.00×10⁸ m/s). Because energy levels are discrete, hydrogen-like atoms give sharp line spectra (specific λ’s), not a continuous rainbow. On the AP exam you should be able to link a photon’s wavelength to an electronic transition using E = hν and explain that absorption raises energy (excitation) and emission lowers it (relaxation)—CED 3.12.A.1–2. Spectra are important: they identify elements (astronomy, lab analysis), reveal energy-level structure (Bohr/model testing), and underlie techniques like flame tests and spectroscopy. For a focused review, see the Topic 3.12 study guide (https://library.fiveable.me/ap-chemistry/unit-3/photoelectric-effect/study-guide/aSateoQF56rKcT1xgLeY) and practice problems (https://library.fiveable.me/practice/ap-chemistry).