What Is Light?
Scientists began to understand light and the way light works by analyzing the light that was emitted or absorbed by substances.
The light we see with our eyes, visible light, is one type of electromagnetic radiation; because electromagnetic radiation carries through space, it is also known as radiant energy.
Light is carried in the form of photons, a type of quantum particle that acts as a "force-carrying particle" for electromagnetic energy. For example, when you turn on a flashlight, trillions of photons shoot out, creating a beam of light that illuminates a dark area. The same concept is applied in lasers and in many incredibly important measurements involving concentrations of solutions, as we'll see later in this unit when discussing the Beer-Lambert law.
😲Fun fact: The word LASER is actually an acronym for Light Amplification by Stimulated Emission of Radiation
We'll get a deeper understanding of what light really is and its role in the quantum world in the next study guide and key concept!

Properties of Light
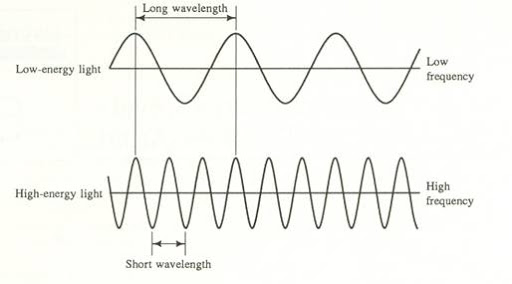
When we discuss light, we typically refer to its wavelength and its frequency. These two terms can help us describe how light will act. Light is an interesting quantum idea because of the fact that it acts both as a particle (the photon) and as a wave. This is called particle-wave duality. This goes back to what we discussed in unit one and electrons also exist as both particles and waves.
When thinking of a wave, it is useful to visualize it as a sine wave, oscillating back and forth periodically.
- Amplitude refers to the vertical height of the wave from the midline. The amplitude of a wave determines the light's intensity or brightness. The greater the amplitude, the brighter the light.
- Wavelength (λ) refers to the length of one period of the wave. This can be described either as the peak-to-peak distance, or the zero-to-zero distance. It is typically measured in nanometers (nm), but it can also be measured in meters or micrometers. The wavelength of a wave determines the color of light.
- Frequency(ν) describes the number of waves that pass a fixed place in a given amount of time and is measured in cycles per second (s^-1) or Hertz (Hz). The hertz is defined as one cycle per second. The frequency of a wave is directly proportional to the speed at which the wave is traveling. Basically, the faster the wave, the greater the frequency.
Frequency and wavelength have an inverse relationship. Essentially, a high wavelength implies a small frequency and vice versa. This relationship can be expressed through the equation c = λν, where c refers to the speed of light. We'll come back to this formula in the next section!
Image Courtesy of Florida State College🌈 The Electromagnetic Spectrum
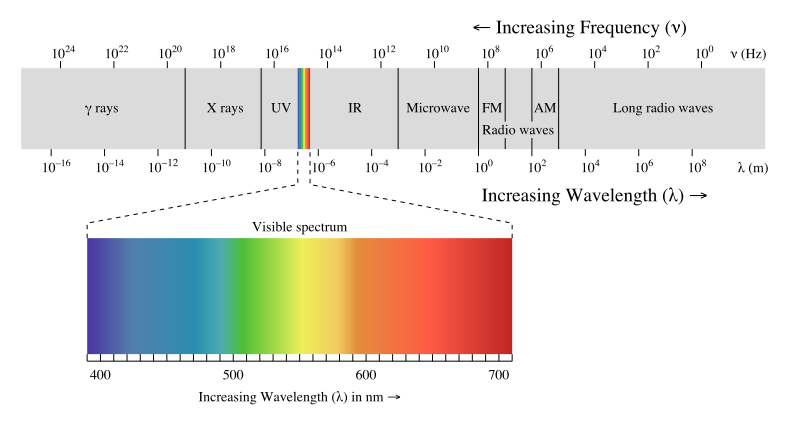
Visible light, which is the light we can see, is only a small category of light. The electromagnetic spectrum includes all wavelengths of electromagnetic radiation, ranging from very short gamma rays to very long radio waves. A key trend to note is that electromagnetic radiation can be characterized by its wavelength; the shorter the wavelength, the higher the frequency.
Spectroscopy is the study of the interaction of radiant energy and matter. The electromagnetic spectrum diagram is shown below to use as a reference:
Image Courtesy of Khan AcademyThere are a few things to notice about the electromagnetic (EM) spectrum, so let's break it down:
- It is broken up into sections based on wavelength from ten-thousandths of nanometers long to hundreds of meters long. To put this in perspective, a nanometer is one billionth of a meter or 10^-9 meters.
- If we start on the left of the EM spectrum, we can see gamma (γ) rays have the shortest wavelength and thus the highest frequency. Because of their high frequencies, they can ionize radiation and are very dangerous. This means that they have enough energy to ionize atoms or molecules by removing their electrons and can easily pass through matter. This makes them very dangerous to humans and we are only exposed to them in small amounts in medical and industrial settings.
- Next on the EM spectrum is X-rays, something you're already familiar with! X-rays have a longer wavelength and lower frequency when compared to gamma rays. Their properties allow them to pass through substances that block visible light, which allows us to use them to image internal bones and organs!
- Next, we got ultraviolet radiation or UV rays. Since their frequency is still relatively high (although low compared to gamma and x-rays), they are very harmful to us in high doses. UV rays are the reason why we have to wear sunscreen when exposed to sunlight for a prolonged period of time, as excessive exposure to UV light increases the risk of skin cancer.
- Taking a look at the EM spectrum, we can see that next is the visible light spectrum, which is the EM radiation that we can see. The photoreceptors in our eyes are only receptive to light within this spectrum, ranging from 400 nm in wavelength to 700 nm. Note that purple💜 has the shortest wavelength while red❤️ has the longest wavelength.- UV rays and visible light are both associated with transitions in energy levels at an atomic level.
- After the visible light spectrum, we have infrared radiation, represented as IR on the EM spectrum above. All of the heat that we feel is infrared radiation since warm objects (even human bodies) emit light at this wavelength! IR is also associated with transitions in molecular vibrational levels.
- Moving towards the opposite end of the spectrum, we are now at microwaves. Microwaves are associated with transitions in molecular rotational levels. They are used in satellite systems and can cook food (hence our microwaves in the kitchen!).- How do microwave ovens warm our food? When you microwave food, the microwaves pass through the container and into the food. The microwaves are absorbed by the food, causing the molecules to vibrate and produce heat.
- Last but not least, we have radio waves. Radio waves have the longest wavelength and thus the lowest frequency. Because of this, radio waves are not really dangerous to us. They are actually used to transmit signals responsible for radio, our cell signal, and television!
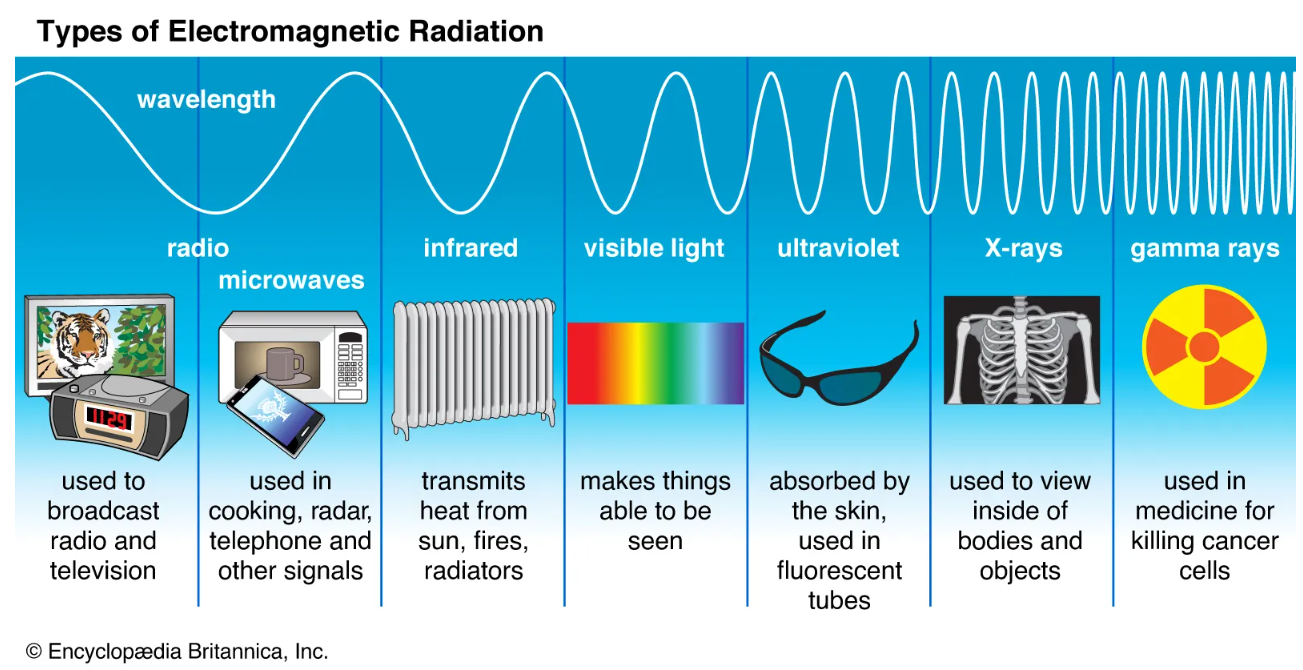
Here is another image that quickly summarizes the practical use of these different rays of light:
Image Courtesy of BritannicaVocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
electromagnetic spectrum | The range of all types of electromagnetic radiation, organized by wavelength and frequency, from radio waves to gamma rays. |
electronic energy levels | Discrete energy states that electrons can occupy in an atom or molecule. |
electronic transition | The movement of an electron between different energy levels in an atom or molecule, which occurs when a photon is absorbed or emitted. |
infrared radiation | Electromagnetic radiation with wavelengths between microwave and visible light, associated with molecular vibrational transitions. |
microwave radiation | Electromagnetic radiation with longer wavelengths and lower frequencies than infrared, associated with molecular rotational transitions. |
molecular rotational levels | Discrete energy states associated with the rotation of a molecule around its axis. |
molecular vibrational levels | Discrete energy states associated with the vibration of atoms within a molecule. |
photon absorption | The process by which matter takes in energy from electromagnetic radiation. |
photon emission | The process by which matter releases energy in the form of electromagnetic radiation. |
ultraviolet/visible radiation | Electromagnetic radiation with shorter wavelengths and higher frequencies than infrared, associated with electronic transitions. |
Frequently Asked Questions
What is the electromagnetic spectrum and how does it relate to spectroscopy?
The electromagnetic spectrum is the full range of light (electromagnetic) waves arranged by wavelength or frequency—from long-wavelength radio and microwaves through infrared (IR), visible, ultraviolet (UV), to X-rays and gamma rays. Photon energy increases as wavelength decreases (E = hc/λ), so different regions can cause different quantized transitions in molecules. Spectroscopy is how we use those photons to probe matter: when molecules absorb or emit light in a specific region, they undergo specific transitions: - Microwaves → rotational level transitions (rotational spectroscopy) - Infrared → vibrational mode transitions (IR spectroscopy) - UV–visible → electronic energy level transitions (UV-Vis spectroscopy) On the AP exam you should be able to link a spectral region to the type of motion and explain absorption vs. emission, selection rules/transition dipole moments, and why higher-energy photons produce electronic changes. For a focused review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do different types of radiation cause different molecular changes?
Different types of radiation cause different molecular changes because photons have different energies (E = hf). Molecules have quantized energy levels for rotation, vibration, and electronic states—each gap needs a specific photon energy to be excited. Microwaves have low energy and match rotational level spacings, infrared matches vibrational mode spacings, and UV–visible has much higher energy that can promote electrons between electronic energy levels (CED 3.11.A.1). Whether a transition actually happens also depends on selection rules and the transition dipole moment (some transitions are allowed and strong, others are forbidden or weak). So it’s both a matter of matching photon energy to the quantized gap and whether the molecular symmetry/selection rules permit that transition. This is exactly what the AP CED asks you to explain for Topic 3.11.A. For a quick review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ). More unit review and practice problems are at the Unit 3 page (https://library.fiveable.me/ap-chemistry/unit-3) and Fiveable’s practice set (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between microwave, infrared, and UV-visible spectroscopy?
Short answer: they probe different quantized motions because photon energy depends on wavelength. - Microwave spectroscopy: probes molecular rotational level changes (lowest energy). Typical wavelengths are ~1 mm–1 m (GHz). Good for identifying rotational constants and small polar molecules; transitions require a changing dipole (rotational selection rules). - Infrared (IR) spectroscopy: probes molecular vibrational modes (mid energy). Mid-IR ≈ 2.5–25 µm (4000–400 cm⁻¹). IR tells you functional groups (stretching, bending); active vibrations require a change in dipole moment. Overtones and combination bands are weaker. - Ultraviolet–visible (UV-vis) spectroscopy: probes electronic transitions (highest energy here). Wavelengths ≈ 200–700 nm. UV-vis shows π→π*, n→π* transitions and gives info on conjugation and electronic structure; intensity depends on transition dipole and allowed/forbidden rules. On the AP exam this is Topic 3.11 (LO 3.11.A). For review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How does infrared spectroscopy actually work to identify molecules?
Infrared (IR) spectroscopy identifies molecules by measuring which IR wavelengths they absorb—those absorptions correspond to quantized vibrational transitions of bonds (CED 3.11.A.1.b). A bond only absorbs IR if the vibration changes the dipole moment (selection rule, transition dipole moment). In practice you get an absorption spectrum (intensity vs. wavenumber, cm⁻¹): sharp, strong peaks (like C=O ≈ 1700 cm⁻¹) or broad features (O–H ~3200–3600 cm⁻¹) tell you which functional groups are present. The “fingerprint region” (≈1500–400 cm⁻¹) is complex but unique to a molecule, so matching spectra can confirm identity. On the AP exam you should be able to link spectral regions to molecular motion (rotational, vibrational, electronic) and interpret key peaks. For a quick review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about molecular transitions - what does that even mean?
A “molecular transition” just means a molecule changes from one quantized energy state to another by absorbing or emitting a photon. Different kinds of transitions correspond to different motions and show up in different spectral regions (CED 3.11.A.1): rotational transitions—microwave; vibrational transitions—infrared; electronic transitions—UV/visible. The photon energy must match the energy gap (ΔE = hν), so only specific wavelengths are absorbed or emitted. Selection rules and the transition dipole moment determine whether a transition is allowed (and how strong the absorption is). You’ll also see overtones (weaker higher-energy vibrational lines) and Raman scattering (a different light-shift mechanism). For AP exam practice, focus on linking spectrum region → type of transition and why (energy scale and molecular motion). For a clear topic review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and more practice questions at (https://library.fiveable.me/practice/ap-chemistry).
Why do molecules absorb different wavelengths of light?
Molecules absorb different wavelengths because different kinds of energy are needed to move them between quantized energy levels. Photon energy = hc/λ, so short wavelengths (high energy) can drive big jumps; long wavelengths (low energy) drive small ones. In AP terms: microwaves match rotational transitions, infrared matches vibrational modes, and UV–visible matches electronic excitations (CED 3.11.A.1). Whether a transition actually absorbs light also depends on selection rules and the transition dipole moment—if the molecular motion changes the dipole (or meets symmetry rules), absorption is allowed and you see a band; if not, it’s weak or absent (Raman can catch some forbidden transitions). Practically, that’s why IR spectra show vibrational peaks (functional groups) while UV–vis shows color from electronic transitions. For more on spectral regions, examples, and AP-style practice, check the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and try practice sets (https://library.fiveable.me/practice/ap-chemistry).
What happens when a molecule absorbs microwave radiation vs infrared radiation?
When a molecule absorbs microwave vs infrared light it excites different quantized motions because the photon energies are different. - Microwave photons (low energy, ~10^9–10^11 Hz) excite rotational transitions—the molecule changes its rotational quantum state. These transitions require the molecule to have a changing dipole as it rotates (selection rules) and give information about molecular bond lengths and moments of inertia. - Infrared photons (higher energy, ~10^13–10^14 Hz) excite vibrational modes—bonds stretch, bend, or wag. IR absorption requires a vibrational mode that changes the dipole moment (transition dipole moment). Overtones and combination bands can appear at higher energies, but the primary IR peaks correspond to fundamental vibrational frequencies. On the AP CED this is 3.11.A.1(a–b): microwaves → rotational, IR → vibrational. For more review and examples (including which modes are IR-active vs inactive), see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ). Practice related problems at (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain electronic transitions in simple terms?
Electronic transitions: electrons in atoms or molecules live in quantized energy levels. If a photon has exactly the right energy (ΔE = hν), an electron can absorb it and jump from a lower to a higher electronic level—that’s absorption. When the electron falls back down it emits a photon (emission). Ultraviolet/visible light corresponds to these electronic transitions (CED 3.11.A.1.c). Higher-energy photons (shorter wavelength, higher frequency) can reach bigger ΔE jumps. Keep these quick points in mind for AP Chem: - Photon energy E = hν; bigger ΔE needs higher-frequency (UV) light. - Transitions follow selection rules (not every jump is allowed; transition dipole moment matters). - Spectra: absorption spectra show wavelengths removed from white light; emission spectra show discrete lines at emitted wavelengths. If you want short review + examples, check the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ). For more practice problems, go to Fiveable’s practice page (https://library.fiveable.me/practice/ap-chemistry).
How do I remember which type of radiation goes with which molecular motion?
Think of least-to-most energy: rotation → vibration → electronic. Match that to long → medium → short wavelength: - Rotational transitions: lowest energy, longest wavelength—microwaves (molecular rotation levels). - Vibrational transitions: medium energy/wavelength—infrared (molecular vibrational modes). - Electronic transitions: highest energy, shortest wavelength—ultraviolet/visible (electronic energy levels). Memory tricks: - Mnemonic: R-V-E (Rotation, Vibration, Electronic)—read it as “Rabbits Visit Europe.” - Or link energy to motion size: rotation = whole-molecule, vibration = bonds moving, electronic = electrons jumping—bigger motion = lower energy. - Also remember selection rules: IR needs a changing dipole; microwaves depend on rotational energy levels; UV/Vis involves electron promotion. For AP exam coverage, the CED expects you to explain these region-to-transition links (3.11.A.1 a–c). Review the Topic 3.11 study guide on Fiveable for concise examples (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and practice problems (https://library.fiveable.me/practice/ap-chemistry) to cement it.
What's the difference between rotational, vibrational, and electronic energy levels?
Rotational, vibrational, and electronic energy levels are just different quantized ways molecules store energy—and each absorbs different parts of the EM spectrum (CED 3.11.A.1). - Rotational levels: lowest-energy, small spacing. They’re transitions between rotational states of whole molecules and absorb microwaves. Think of a molecule spinning faster or slower; selection rules let ΔJ = ±1 for a dipole-active rotor. - Vibrational levels: mid-energy, bigger spacing than rotational. Vibrational modes (stretching, bending) absorb infrared photons. You can also get overtones and combination bands; IR active vibrations require a changing dipole moment. - Electronic levels: highest-energy changes in which electrons occupy different molecular orbitals; these absorb UV–visible light. Electronic transitions often couple to vibrational/rotational structure (fine structure) and require transition dipole moments. For AP prep, know the spectrum-region ↔ transition mapping (microwave → rotational; IR → vibrational; UV/Vis → electronic) and be ready to explain why on the exam (CED 3.11.A). For a focused review check the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and use practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does UV-visible light affect electrons but infrared doesn't?
Because photon energy depends on frequency (E = hν), different EM regions match different quantized energy gaps in molecules. UV–visible photons (≈400–700 nm for visible; shorter for UV) have enough energy (a few eV) to promote electrons between electronic energy levels—those are the electronic transitions tested in Topic 3.11 (UV/vis → electronic). Infrared photons have much lower energy and instead match vibrational energy level spacings (bond stretches and bends). So IR is absorbed by changing vibrational states, not by ejecting or exciting electrons to higher electronic orbitals. Selection rules and transition dipole moments also matter: only transitions with the right symmetry and nonzero dipole coupling absorb light. AP-relevant keywords: photon energy, wavelength/frequency, electronic transitions (UV/vis), vibrational transitions (IR), quantized energy levels. For a quick refresher, check the Topic 3.11 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do scientists use spectroscopy to figure out what's in a sample?
Scientists shine light on a sample and record which wavelengths are absorbed or emitted—that pattern is a fingerprint for what’s inside. Different spectral regions probe different transitions: microwaves → rotational levels, infrared → molecular vibrations (functional groups), and UV–visible → electronic transitions (conjugation, metal ions) (CED 3.11.A.1). An absorption peak at a specific frequency means a quantized transition matched the photon energy (E = hν); selection rules and the transition dipole moment determine whether a transition appears. UV–vis can also give concentration using Beer–Lambert law. Comparing measured spectra to known reference spectra or using characteristic IR bands lets you identify molecules; high-resolution spectra can even show isotopes or bond environments. For AP prep, know which region corresponds to which transition and how spectra are used on MC/FR items. For a focused review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ), the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3), and practice questions (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how photon energy relates to molecular transitions - help?
Think of a photon as a packet of energy exactly equal to the gap between two quantized molecular energy levels. E(photon) = h·ν = hc/λ, so shorter λ (higher frequency) = more energy. Different regions of the EM spectrum match different gaps in molecules: microwaves (lowest energy) excite rotational level changes, infrared excites vibrational modes, and UV–visible (highest) promotes electronic transitions (CED 3.11.A.1). A transition only happens if the photon energy equals the level spacing and the transition is allowed (nonzero transition dipole moment / selection rules). So if you want to break an electronic bond or move an electron, you need UV/visible photons; to change bond stretch you need IR; to change overall rotation you need microwaves. This idea shows up on the exam as identifying which radiation causes which transition or using E = hc/λ numerically. For practice and review of spectra and example problems, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and more practice questions (https://library.fiveable.me/practice/ap-chemistry).
What happens to a molecule when it absorbs a photon of light?
When a molecule absorbs a photon, the photon’s energy is taken up in a quantized transition—the molecule jumps from a lower to a higher energy level. Which kind of motion changes depends on the photon energy: microwave photons change rotational levels, infrared photons excite vibrational modes, and UV–visible photons promote electrons to higher electronic states (CED 3.11.A.1). After absorption the molecule is in an excited state and can relax by: emitting a photon (fluorescence or phosphorescence), losing energy nonradiatively as heat (vibrational relaxation), or undergoing a chemical change (bond breaking) if the photon energy is large enough. Whether a transition is allowed depends on selection rules and the transition dipole moment, which determine the absorption spectrum you measure. For AP review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to know about different regions of the electromagnetic spectrum in chemistry?
You need to know different EM regions because each region probes different allowed, quantized transitions in molecules—so the wavelength tells you what kind of motion or energy change you’re measuring. Per the CED: microwaves excite rotational levels, infrared excites vibrational modes, and UV–visible promotes electronic transitions. Photon energy (E = hν) increases from microwave → IR → visible/UV, so only photons with the right energy and obeying selection rules and transition dipole moments will be absorbed or emitted. That’s why IR spectra tell you functional groups (vibrations), microwave/rotational spectra reveal molecular geometry and moments of inertia, and UV–Vis gives conjugation/ electronic structure. On the AP exam Topic 3.11 you may be asked to link a spectral region to the type of transition, interpret absorption vs emission, or explain why a transition is allowed. For targeted review, see the Topic 3.11 study guide (https://library.fiveable.me/ap-chemistry/unit-3/spectroscopy-electromagnetic-spectrum/study-guide/Swp8nLjZFev1h1Fu9sqJ) and practice questions (https://library.fiveable.me/practice/ap-chemistry).