So far, we learned about intramolecular forces within ionic solids, metallic solids, and alloys in unit two. Now that we've discussed the different types of intermolecular forces, let's discuss more solids!
Many properties of solids are determined by the strengths and types of intermolecular forces present. Since intermolecular interactions must be completely broken when a substance vaporizes, properties like vapor pressure and boiling point are directly related to the strength of these interactions. The stronger the intermolecular forces, the lower the vapor pressure and the higher the boiling point. Melting points also tend to correlate with interaction strength, though the relationship can be more subtle since interactions are only rearranged, not completely broken.
The structures and properties of solids can be classified according to the forces that hold the atoms together.
- Ionic solids are held together by electrostatic forces between cations and anions (think Coulomb's law).
- Metallic solids are held together by metallic bonds between atoms.
- Covalent network solids are held together by an extended network of covalent bonds (think of diamonds 💎).
- Molecular solids are held together by weak IMFs.
Now that the basics are out of the way, let’s dig into the details!
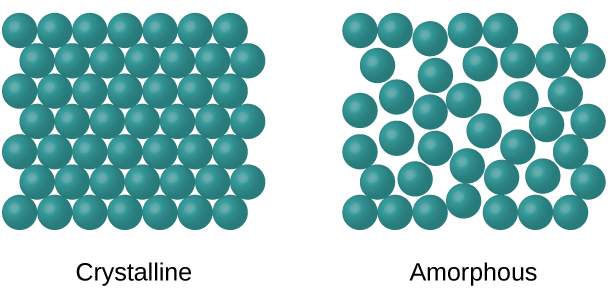
Amorphous Solids
Amorphous solids are those that do not have a long-range, periodic crystal structure. They have considerable disorders in their structures and it is all due to cooling quickly.
These are their own category and will not be studied in-depth in AP Chemistry. However, some examples of amorphous solids include gum, glass, and rubber.
Image Courtesy of OpenTextBC

Crystalline Solids
Crystalline solids are the greater category of solids and include all of the different types of solids that the College Board wants you to know for the AP Chemistry exam: ionic solids, covalent network solids, molecular solids, and metallic solids.
In crystalline solids, particles are arranged in a regularly repeating pattern. Crystalline solids have a definite melting point, and they tend to be more ductile and less brittle than amorphous solids.
Structure of Crystalline Solids
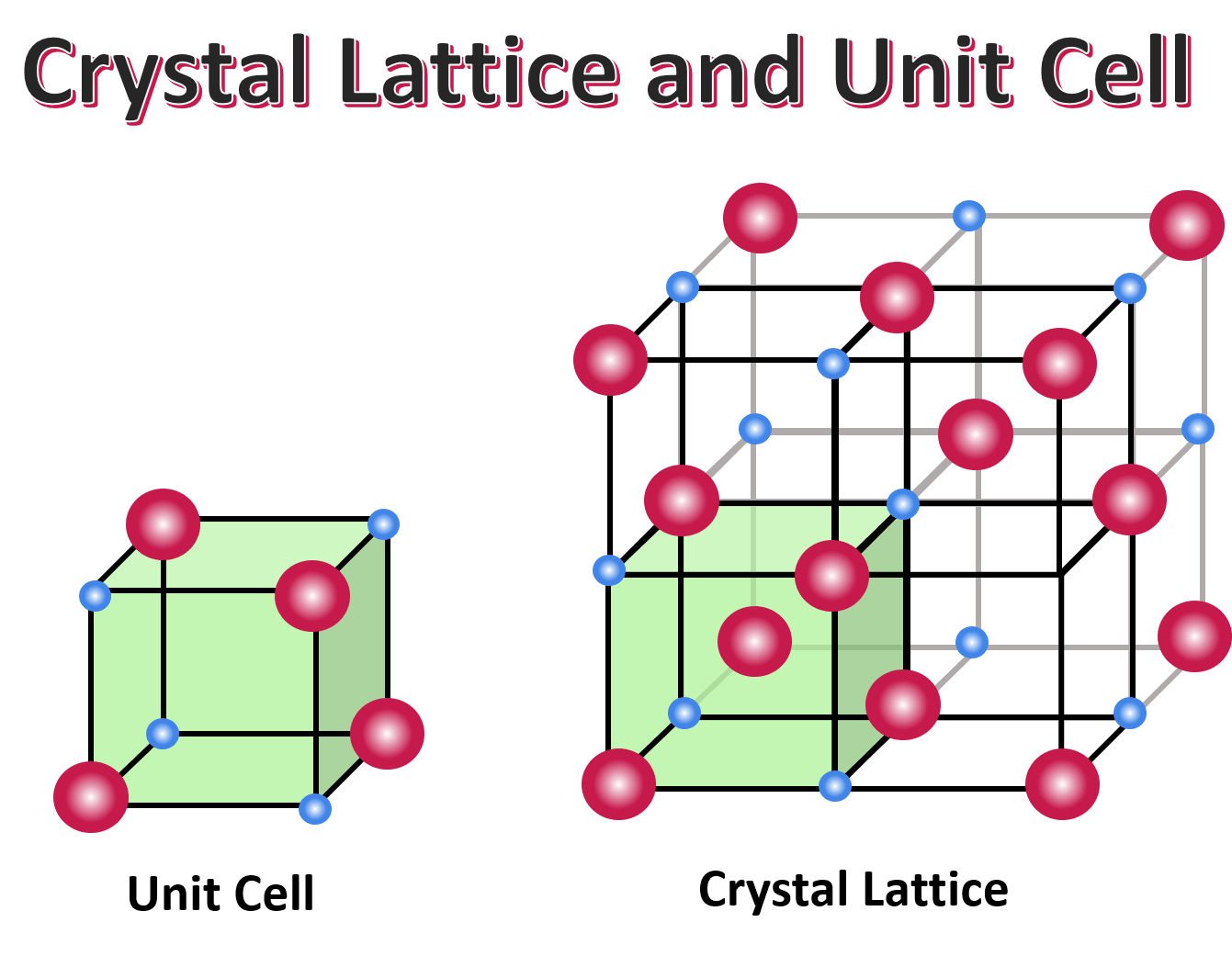
The geometrical pattern of points on which crystalline solids are arranged is called a crystal lattice. The basic building block, or smallest repeating unit, that makes up a crystal lattice is a unit cell.
In other words, the unit cell is the smallest unit that can be used to describe the crystal lattice, and it is repeated throughout the solid to form the crystal lattice.
Particulate-level representations (drawings showing individual atoms, molecules, or ions) are extremely useful for understanding how the microscopic structure relates to macroscopic properties. When you see these diagrams, pay attention to how the particles are arranged and what forces hold them together - this directly explains the solid's properties!
Image Courtesy of Expii; You do not need to know the types of unit cells, but here is an overview of what this term really means!Basically, if one cube represents a unit cell, then 100 cubes combined into a structure would represent a crystal lattice.
Metallic Solids
Metallic solids are composed of metal atoms held together by metallic bonds.
When metals ionize, they lose a valence electron and become positive ions or cations. Metallic bonding can therefore be represented as an array of cations surrounded by this "sea" of valence electrons. The valence electrons are delocalized and visualized as being free to move throughout the entire metal.
Animation Courtesy of DocSityBecause of the delocalization of valence electrons, metallic substances have very unique properties:
- Good conductors of electricity ⚡
- High melting and boiling points 🌡️
- Shiny appearances 🌟
- Malleability and ductility 🔌
The malleability and ductility of metals are particularly interesting properties. Metals can be hammered into sheets (malleable) or drawn into wires (ductile) due to the ease with which the metal cores can rearrange their structure. When a force is applied, the metal atoms can slide past each other while the delocalized electron "sea" adjusts to maintain metallic bonding throughout the material. This is very different from ionic solids, which shatter when layers shift!
[👉 Want to review more about metallic solids and why they have these properties? Check out our explanations in unit two: Structure of Metals and Alloys.
Alloys
Alloys are materials that possess characteristic metallic properties and are composed of two or more elements. The elements in an alloy can be distributed as either homogeneous or heterogeneous.
- In a substitutional alloy, the atoms of the minority element occupy positions normally occupied by atoms of the majority element.
- In an interstitial alloy, atoms of the minority elements, often smaller nonmetallic atoms, occupy interstitial positions that lie in the "holes" between atoms of the majority element.
The type of alloy affects the material's properties significantly. In interstitial alloys, the interstitial atoms tend to make the lattice more rigid, decreasing malleability and ductility compared to the pure metal. This happens because the small atoms in the holes prevent the metal layers from sliding as easily. For example, steel (iron with carbon atoms in interstitial positions) is harder and less malleable than pure iron, making it more suitable for construction.
Image Courtesy of the University of North FloridaIonic Solids
Ionic solids consist of cations and anions held together in a crystal lattice by electrostatic forces. Since these interactions are quite strong, ionic compounds tend to have a higher melting point.
The key trend to note is that the attractions become stronger as the charges of ions increase and/or the sizes of the ions decrease (Coulomb's law anyone?).
The presence of both attractive and repulsive interactions helps to explain why ionic compounds are brittle. When you apply force to an ionic crystal, it can cause one layer of ions to slide across another layer. This sliding brings ions of the same charge next to each other (cation next to cation, anion next to anion). Since like charges repel, this creates strong repulsive forces that cause the crystal to fracture or shatter. This brittleness is characteristic of ionic solids - they're strong but not flexible!
They only conduct electricity when ions are mobile and can flow. This occurs when ionic solids are melted or put into a solution.
Something new to note about ionic solids is that they possess ions at the lattice points of the solid structure.
Molecular Solids
Molecular solids consist of atoms or molecules held together by IMFs. They possess strong intramolecular forces (covalent bonds), but weak intermolecular forces.
Let's break down the general properties of molecular solids:
- 🌡️ Low melting and boiling points: Because the intermolecular forces holding molecular solids together are weak, molecular solids have relatively low melting and boiling points. The melting point depends on the strength of the IMFs, as well as the efficiency with which the molecules can pack together. Two good examples of molecular solids are ice and sucrose.
- 💎Brittle and hard: Molecular solids tend to be brittle and hard, as they are held together by weak intermolecular forces that are easily broken.
- 🚫🔌Poor conductors of heat and electricity: Molecular solids are poor conductors of heat and electricity, as they do not have a metallic structure with free electrons that can easily flow through the solid. Their valence electrons are rather tightly held within covalent bonds.
Molecular solids can also be composed of very large molecules or polymers. Examples include proteins, DNA, and synthetic polymers like polyethylene. In these large molecules, the properties depend strongly on their three-dimensional shape, which is determined by noncovalent interactions between different parts of the molecule.
Covalent Network Solids
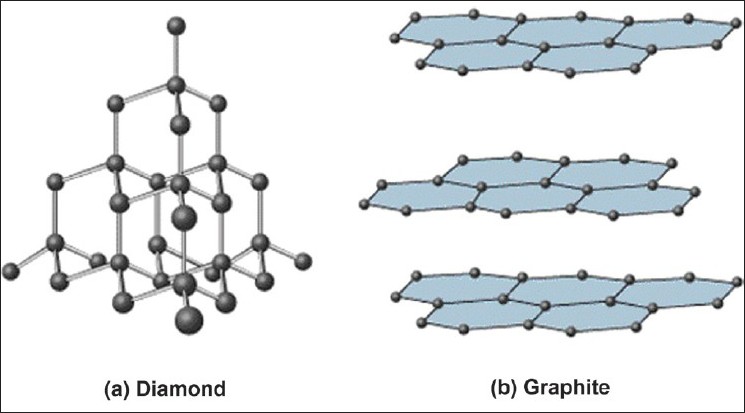
Covalent network solids consist of atoms held together in large networks by covalent bonds. These solids are much harder and have higher melting points than molecular solids. Important examples to keep in mind are graphite and diamond, which are both made up of carbon.
These large networks could be atoms bonded in a 3D network, like diamonds, or 2D layers, like graphite.
Image Courtesy of JidOnline✏️ Graphite
- Layers of Carbon atoms arranged in rings
- sp2 hybridized, pi bonds
- delocalized electrons --> good conductor of heat and electricity
- strong bonds within the layers, but weak bonds between the layers
- soft because the layers can slide past each other (due to weak bonds holding the layers together)
💎 Diamond
- hardest naturally occurring substance
- sp3 hybridized
- insulator
Biomolecules and Polymers
In large biomolecules or polymers, noncovalent interactions play a crucial role in determining their functionality and properties. It's important to understand that these interactions can occur between different molecules or between different regions of the same large biomolecule:
- Intermolecular interactions: Forces between separate molecules (e.g., hydrogen bonds between different protein molecules in a crystal)
- Intramolecular interactions: Forces within a single large molecule between different parts (e.g., hydrogen bonds between different amino acids in the same protein chain)
The shape of these molecules, which is largely dictated by noncovalent interactions like hydrogen bonding, London dispersion forces, and dipole-dipole interactions, determines their biological function. For example:
- Proteins fold into specific 3D shapes due to interactions between amino acid side chains
- DNA maintains its double helix structure through hydrogen bonding between base pairs
- Synthetic polymers like nylon have properties determined by hydrogen bonding between polymer chains
These materials often form molecular solids where the individual polymer chains or biomolecules are held together by intermolecular forces.
Comparing Solids
When comparing properties among the different solids, remember this chart:
| Type of Solid | Form of Unit Particles | Forces Between Particles | Properties | Examples |
|---|---|---|---|---|
| Molecular🧊 | Atoms or Molecules | LDFs, dipole-dipole, hydrogen bonding | fairly soft, low melting point, high vapor pressure, bad conductor | Argon, methane, sucrose, dry ice |
| Covalent Network💎 | Atoms connected in a network of covalent bonds | Covalent Bonds | Very hard, very high melting point, extremely low vapor pressure, bad conductor | diamond, quartz |
| Ionic🧂 | Positive and Negative Ions | Electrostatic attractions | Hard and brittle, high melting point, low vapor pressure, bad conductor (conducts when melted/dissolved) | salts (NaCl) |
| Metallic✨ | Atoms | Metallic Bonds | Varying hardness and melting points, low vapor pressure, good conductor, malleable, ductile | metals! Cu, Fe, Al |
Remember: Vapor pressure is inversely related to the strength of intermolecular forces. Molecular solids have the weakest forces, so they have the highest vapor pressures and evaporate most easily!
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
boiling point | The temperature at which a liquid vaporizes, directly related to the strength of intermolecular interactions that must be overcome. |
brittle | The property of a material that causes it to break or shatter easily when subjected to stress, characteristic of ionic solids due to repulsion of like charges. |
covalent network solids | Solids in which atoms are covalently bonded together in continuous three-dimensional or two-dimensional networks, such as diamond and graphite. |
ductile | The property of a material that allows it to be drawn or stretched into thin wires without breaking. |
intermolecular forces | Attractive forces between separate molecules or particles that determine many properties of liquids and solids, including boiling point, melting point, and vapor pressure. |
interstitial alloy | An alloy in which smaller atoms occupy the spaces between larger atoms in the crystal lattice, making the structure more rigid and decreasing malleability and ductility. |
ionic solids | Solids composed of cations and anions held together by strong electrostatic forces, characterized by low vapor pressures, high melting and boiling points, and brittleness. |
macroscopic properties | Observable physical and chemical characteristics of a substance that can be measured at the bulk level, such as melting point, boiling point, and vapor pressure. |
malleable | The property of a material that allows it to be hammered or pressed into thin sheets without breaking. |
melting point | The temperature at which a solid transitions to a liquid, which tends to correlate with the strength of intermolecular interactions. |
metallic solids | Solids composed of metal atoms with delocalized valence electrons that move freely, resulting in good electrical and thermal conductivity, malleability, and ductility. |
molecular solids | Solids composed of distinct molecules held together by relatively weak intermolecular forces, generally having low melting points. |
molten state | The liquid state of an ionic solid in which ions are mobile and able to conduct electricity. |
noncovalent interactions | Weak attractive or repulsive forces between molecules or regions of molecules that do not involve the breaking or formation of covalent bonds. |
particulate-level structure | The arrangement and organization of atoms, ions, or molecules that make up a substance at the atomic and molecular scale. |
polymers | Large molecules composed of repeating units of smaller molecules linked together, whose properties depend on noncovalent interactions and molecular shape. |
valence electrons | Electrons in the outermost shell of an atom that participate in bonding and determine many properties of substances. |
vapor pressure | The pressure exerted by a vapor in equilibrium with its liquid or solid phase at a given temperature. |
Frequently Asked Questions
What's the difference between ionic, covalent, and metallic solids?
Ionic solids: made of alternating cations and anions in a crystal lattice held by strong Coulombic attraction (high lattice energy). Macroscopic: high melting/boiling points, low vapor pressure, brittle (fracture when like-charged layers line up), and only conduct electricity when ions are mobile (molten or dissolved)—see CED 3.2.A.3. Covalent solids (split into covalent network vs molecular): - Covalent network solids (diamond, SiO2) are atoms covalently bonded into 3-D (or layered) networks—very high melting points, hard and rigid; graphite is an exception (layers slide)—CED 3.2.A.4. - Molecular solids are made of discrete molecules held by weak intermolecular forces (van der Waals, H-bonding); low melting points and poor electrical conductivity—CED 3.2.A.5. Metallic solids: metal atoms in a lattice with a “sea” of delocalized valence electrons (metallic bonding). They conduct heat/electricity well, are malleable/ductile, and alloys often remain conductive (3.2.A.6). For a quick AP-aligned refresher, check the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do ionic compounds only conduct electricity when melted or dissolved in water?
Ionic solids (like NaCl) have ions held in a rigid crystal lattice by strong Coulombic attractions (high lattice energy). In the solid state the ions are fixed in place, so they can’t move to carry charge—no electrical conductivity. When you melt the solid or dissolve it in water, those strong ionic interactions are at least partly overcome and the ions become mobile (free Na+ and Cl– in molten salt or in aqueous solution). Mobile charged particles are required for current, so molten or dissolved ionic compounds conduct electricity. This ties directly to CED 3.2.A.3: ionic solids → high melting points, low vapor pressure, and conduct only when ions are mobile. For more on how particulate structure controls macroscopic properties see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about why graphite is soft but diamond is hard when they're both made of carbon.
They’re both carbon, but their bonding and structure are totally different. Diamond is a 3D covalent network (each C is sp3-hybridized and bonded tetrahedrally to 4 others). Those strong covalent bonds run throughout the crystal, so it’s rigid, has a very high melting point, and is hard (CED 3.2.A.4). Graphite is layers of 2D networks (C is sp2), so each atom has strong in-plane covalent bonds but only weak van der Waals forces between layers. Those layers can slide past one another easily, making graphite soft and lubricating. Graphite also has delocalized electrons in the planes, so it conducts electricity; diamond does not. This is a classic particulate-to-macroscopic example you should be able to explain for Topic 3.2 (see the study guide: https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v). For more practice connecting structure to properties, try problems at https://library.fiveable.me/practice/ap-chemistry.
How do intermolecular forces affect melting and boiling points?
Intermolecular forces (IMFs) control how hard it is to separate particles, so stronger IMFs → higher melting and boiling points. Boiling point is especially sensitive: vaporization fully overcomes IMFs, so substances with strong attractions (ion–ion, hydrogen bonding, permanent dipole–dipole) have low vapor pressure and high b.p. Melting is related too but subtler—melting only rearranges interactions, so crystal packing and lattice energy matter. Examples from the CED: ionic and covalent-network solids have very high m.p./b.p. because of strong Coulombic or covalent bonds; molecular solids have low m.p. because only van der Waals (London) or H-bonding hold them together. London dispersion increases with molar mass and surface area, so bigger/less compact molecules have higher b.p. and m.p. Hydrogen bonding gives unusually high b.p. for small molecules (e.g., water). For AP review, focus on linking particulate-level models to macroscopic m.p./b.p. (Topic 3.2); see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What makes metals malleable and ductile compared to other types of solids?
Metals are malleable and ductile because of metallic bonding: metal atoms donate valence electrons to a “sea” of delocalized electrons that bathes positive metal cores. Those mobile electrons produce non-directional bonding that holds the lattice together even when atoms shift. So when you hammer (malleability) or pull (ductility) a metal, layers of metal cores can slide past one another without breaking strong directional bonds or creating large charge repulsions (unlike ionic solids, where sliding brings like charges together and makes them brittle). Alloys can change this—interstitial atoms block sliding and make metals less malleable/ductile. This links directly to CED 3.2.A.6 (metallic solids → good conductors, malleable/ductile). For a quick review tied to the AP unit, check the Topic 3.2 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and use practice problems (https://library.fiveable.me/practice/ap-chemistry) to prep.
Can someone explain why molecular solids have low melting points in simple terms?
Molecular solids have low melting points because the units in the solid are whole, neutral molecules held together by relatively weak intermolecular forces (London dispersion, dipole–dipole, and sometimes hydrogen bonding), not strong ionic or covalent bonds. Inside each molecule the atoms are held by strong covalent bonds, but to melt you only need enough energy to overcome or reorganize the weak attractions between molecules—that takes much less energy than breaking ionic lattice or a covalent network. So melting points correlate with intermolecular force strength: stronger IMFs → higher mp; weaker IMFs → lower mp (CED 3.2.A.1, 3.2.A.5). Molecular solids also don’t conduct electricity because electrons are localized in bonds/lone pairs. For a quick AP-aligned review, see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try related practice problems (https://library.fiveable.me/practice/ap-chemistry). This idea shows up on the exam when you explain macroscopic properties from particulate structure (LO 3.2.A).
Why are ionic solids brittle and what happens when you try to bend them?
Ionic solids are brittle because their crystal lattice is made of alternating positive and negative ions held by strong Coulombic attraction. If you try to bend or shear the crystal, entire planes of ions can shift so that like-charged ions (e.g., Na+ next to Na+ or Cl− next to Cl−) become adjacent. Those like-charge neighbors repel each other strongly, and the lattice fractures instead of flowing. So when you bend an ionic solid it doesn’t deform plastically—it cleaves or shatters along planes where repulsion becomes too large. This contrasts with metals, which are malleable because a “sea” of electrons lets metal cations slide past one another without catastrophic repulsion (CED 3.2.A.3 and 3.2.A.6). For more on particulate-level models and AP exam alignment for Topic 3.2, see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the deal with covalent network solids and how are they different from regular covalent compounds?
Covalent network solids are NOT just big collections of molecules—they’re atoms linked by covalent bonds into one huge 2-D or 3-D lattice (diamond, graphite, SiO2, SiC). Because the strong covalent bonds extend through the whole solid, network solids have very high melting points, are rigid/hard (3-D diamond), and usually don’t conduct electricity (except graphite, where layers slide and delocalized electrons conduct). By contrast “regular” covalent compounds (molecular solids) are made of discrete molecules held together by relatively weak intermolecular forces (London, dipole, H-bond). Those molecular solids have much lower melting points and don’t conduct electricity because electrons stay localized in bonds or lone pairs (CED 3.2.A.4–3.2.A.5). On the AP exam you should be able to connect particulate structure (bonding) to macroscopic properties like melting point, hardness, and conductivity—review Topic 3.2 (study guide: https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I remember which type of solid conducts electricity and which ones don't?
Quick memory trick: ask “are the charge carriers mobile?” If yes → it conducts. - Metallic solids: yes in the solid state—they have a sea of delocalized valence electrons that move (3.2.A.6, electron-sea model). - Ionic solids: no as solids (ions locked in lattice) but yes when molten or dissolved because ions become mobile (3.2.A.3). - Molecular solids: generally do not conduct—electrons are locked in covalent bonds or lone pairs (3.2.A.5). - Covalent network solids: usually don’t conduct (diamond no), except some allotropes like graphite conduct because of delocalized electrons in layers (3.2.A.4). So: metals = conduct; ionic = conduct only when ions can move (molten/aqueous); molecular/network = usually don’t (watch graphite). For a quick AP-aligned review see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why do alloys become less malleable when you add interstitial atoms?
Metals are malleable because metal atoms (positive cores) can slide past each other while the “sea” of delocalized valence electrons keeps bonding intact. When small interstitial atoms (like C in steel) sit in the gaps of the metal lattice, they crowd the spaces between metal atoms. Those extra atoms distort the regular lattice and block the easy movement of metal layers and dislocations, so the metal cores can’t rearrange as readily. The result: the lattice becomes more rigid and malleability and ductility drop, even though the sea of electrons (and electrical conductivity) usually remains (CED 3.2.A.6). This is exactly what the AP CED wants you to link: particulate structure → particle interactions → macroscopic property changes. For a quick refresh, see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
What are some real world examples of each type of solid that I can use on the test?
Ionic solids—e.g., table salt (NaCl) and calcium carbonate (CaCO3). High melting points, brittle, conduct only when molten/dissolved (CED 3.2.A.3). Covalent network solids—e.g., diamond and graphite (elemental), quartz (SiO2), silicon carbide (SiC). Very high melting points; rigid 3D networks or layered structures (CED 3.2.A.4). Molecular solids—e.g., dry ice (CO2), iodine (I2), sucrose (table sugar), solid H2O (ice). Low melting points, held by van der Waals/hydrogen bonding; nonconductive (CED 3.2.A.5). Metallic solids—e.g., copper (wiring), iron (steel), aluminum foil; alloys like steel or brass. Good electrical/thermal conductors, malleable/ductile due to electron sea (CED 3.2.A.6). Polymers/biomolecular solids—e.g., polyethylene (plastic), proteins (keratin in hair); properties set by noncovalent interactions and chain shape (CED 3.2.A.7). Use these concrete examples on the exam to link macroscopic properties to particle-level interactions. For review, see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and grab practice questions at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how particle diagrams show the connection between structure and properties.
Think of a particle diagram as a zoomed-in map that links structure → interactions → macroscopic properties (that’s exactly what CED 3.2.A asks you to explain). For example: - Ionic lattice: alternating +/− ions tightly packed—strong Coulombic attraction → high melting/boiling points, low vapor pressure, brittle (like-charge repulsion when layers slide), conducts only when ions move (molten/dissolved). - Covalent network: atoms covalently bonded into 3D or layered networks → very high melting points and hardness (diamond) or soft/slippery layers (graphite). - Molecular solid: discrete molecules held by weak van der Waals/H-bonds → low melting points, poor electrical conductivity. - Metallic: metal cations in an electron sea → good electrical/thermal conductivity, malleable/ductile. When you draw a particle diagram, show multiple species, bond types, and mobility—then ask: how strong are interactions? Are charge carriers mobile? Can layers slide? Those observations let you predict melting point, conductivity, ductility, vapor pressure, etc. For quick review and examples, check the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why do metals have a "sea of electrons" and what does that actually mean?
Metals have a "sea of electrons" because their valence electrons are not held tightly to individual atoms—instead they become delocalized across the whole metal lattice. Practically that means positively charged metal ions sit in a regular crystal lattice while the valence electrons move freely among them. This “electron sea” is the metallic bond: electrostatic attraction between the mobile electrons and the metal cations holds the solid together (CED 3.2.A.6 keywords: metallic bonding, free valence electrons). Why it matters: mobile electrons let metals conduct electricity and heat easily, and because the cation cores can slide past one another while the electron cloud still holds them together, metals are malleable and ductile. For AP Chem, connect this particulate model to macroscopic properties (Topic 3.2; see the Topic 3.2 study guide on Fiveable: https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v). For extra practice, try relevant solids questions at https://library.fiveable.me/practice/ap-chemistry.
How do noncovalent interactions affect the shape and function of large biomolecules like proteins?
Noncovalent interactions—hydrogen bonding, ionic (electrostatic) attractions, van der Waals (London dispersion) forces, and hydrophobic effects—fold proteins into specific 3D shapes (secondary, tertiary, quaternary). These weak, reversible forces between side chains and backbone atoms set the particulate-level structure that determines macroscopic function: active sites, binding pockets, stability, and whether subunits assemble. Change or disruption of these interactions (pH shifts, salt, heat, or solvents) alters shape and often destroys function (denaturation). On the AP exam, connect microscopic interactions to macroscopic properties (CED 3.2.A and EK 3.2.A.7): show how specific interactions stabilize helices/sheets, hold tertiary folds, or mediate subunit interfaces. For practice drawing particulate-level models and linking interactions to properties, see the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and hundreds of practice questions (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between vapor pressure and intermolecular forces in solids?
Vapor pressure is a measure of how readily particles escape from a phase into the gas. Stronger intermolecular (or interparticle) forces make it harder for particles to leave a solid, so stronger forces → lower vapor pressure and higher boiling/melting points (CED 3.2.A.1). That’s why ionic and covalent-network solids (very strong Coulombic or covalent bonds) have essentially negligible vapor pressures at room temperature, while molecular solids with weak van der Waals or H-bonding have higher vapor pressures and can sublime or vaporize more easily (CED 3.2.A.3–3.2.A.5). For AP exam framing: link vapor pressure and boiling point to interaction strength and use particulate models to justify macroscopic trends (3.2.A). Want a quick review or practice problems on this topic? Check the Topic 3.2 study guide (https://library.fiveable.me/ap-chemistry/unit-3/properties-solids/study-guide/0lW4bHW7ksIahDb0zW9v) and try practice questions at (https://library.fiveable.me/practice/ap-chemistry).