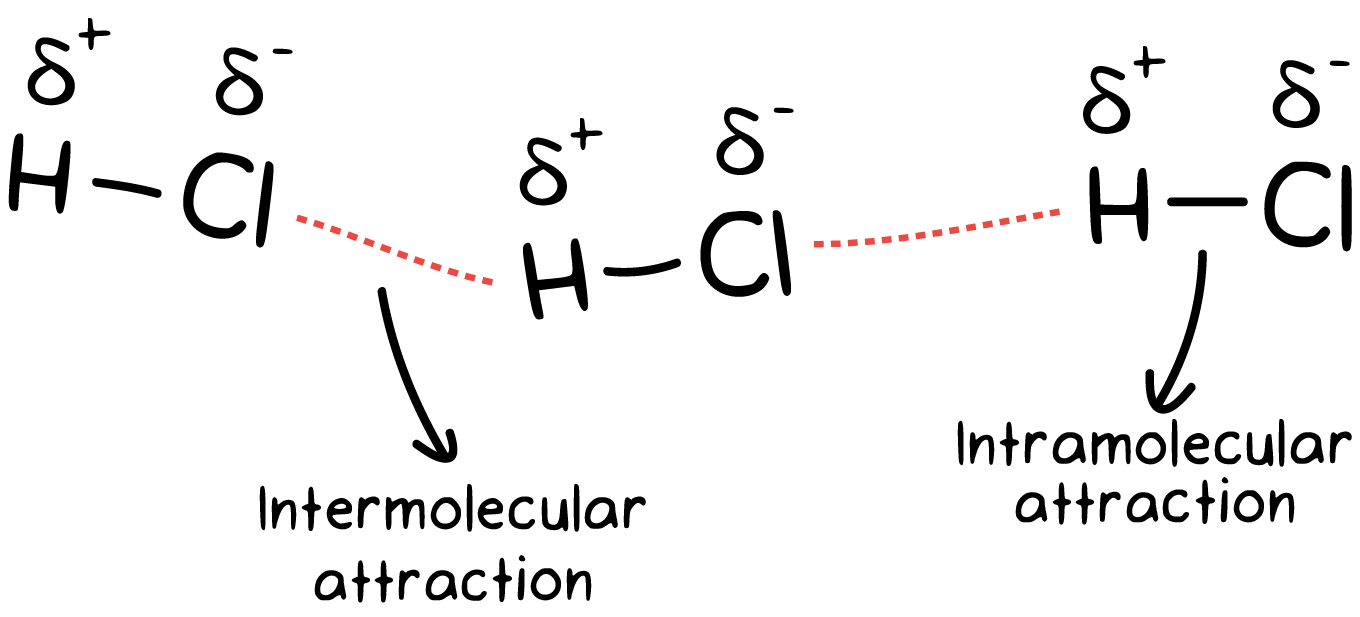
Intermolecular forces (IMFs) are the attractive or repulsive forces between entire molecules due to differences in charge. Many students confuse IMFs with intramolecular forces, which were the center of the last unit. Try to remember the following:
- Intermolecular forces - forces that hold molecules together.- Types: London dispersion forces, dipole-dipole forces, hydrogen bonds, and ion-dipole forces
- Intramolecular forces - forces within a molecule that hold atoms together.- Types: covalent bonds, metallic bonds, ionic bonds
🤔 One way to remember the difference is that "inter" means between, while "intra" means within.
IMFs are much weaker than intramolecular forces. This is due to the simultaneous attraction for electrons that exist between two nuclei within a molecule!
Image Courtesy of Khan AcademyYou can also think of IMFs as weaker because they exist over a larger distance. Remember that Coulomb's law states that the closer the two particles are, the stronger the attraction.
In this study guide, we'll learn about four types of intermolecular forces: London dispersion forces, dipole-dipole forces, hydrogen bonding, and ion-dipole forces.

London Dispersion Forces
London Dispersion Forces (LDFs) are the weakest type of IMFs and occur in all molecular samples. They are the only types of forces to exist between two non-polar molecules and noble gases (solid or liquid form).
On the AP Exam, if you are asked to list the types of IMFs being seen in a certain molecule, London dispersion forces will always be an answer. They can easily be forgotten so make sure they always show up in your answer (along with any other forces present)!
A quick note on terminology: While you might hear the term "van der Waals forces," be careful about its usage. Van der Waals forces is a general term for weak intermolecular attractions that includes London dispersion forces. However, you should NOT use these terms interchangeably - London dispersion forces are a specific type of van der Waals force that occurs between temporary dipoles. On the AP exam, always use the specific term "London dispersion forces" rather than the more general "van der Waals forces."
At any given time, one nonpolar molecule might have more electrons on one side than the other side, making it polar. For that instant, the molecule would have a partial negative side and a partial positive side and it creates a temporary dipole.
The molecule with the temporary dipole then induces a dipole on its neighboring molecules, creating a London dispersion force between the partial negative side of one molecule and the partial positive side of another molecule.
In other words, London dispersion forces are a result of Coulombic interactions between temporary and fluctuating dipoles.
Here is a quick visual to sum up this idea of an LDF:
The strength of LDFs increases as the size of a molecule increases. This is because more electrons yield a stronger instantaneous dipole due to a more polarizable electron cloud. Polarizability is the ease at which an electron cloud could be distorted to give a dipole charge distribution. This polarizability is enhanced by the presence of pi bonds, as pi electrons are held less tightly than sigma electrons and can be more easily distorted.
The concept discussed in the last paragraph is a very important piece of information that is typically asked on the AP exam. The more electrons there are, the greater the size of the electron cloud, and the more polarizable it is! This makes it more likely for there to be a temporary dipole.
Dipole-Dipole Interactions
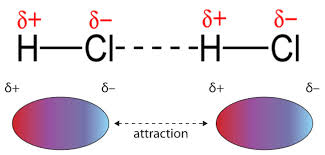
Dipole-dipole attractions occur between the opposite partial charges that exist on opposite ends of a dipole. While LDFs are present due to temporary dipoles, dipole-dipole forces result from permanent dipoles. As a result, dipole-dipole attractions only occur in a sample of polar molecules and are slightly stronger than LDFs.
The dipoles in HCl lead to a positive side and a negative side that are attracted to each other.Image Courtesy of EMedicalPrep As you decrease the distance between the two dipoles, you strengthen the dipole-dipole interaction. This relates back to Coulomb's law. Keep in mind, since these involve polar molecules, the element with the higher electronegativity (in this case Cl-) has a partial negative charge (δ−).
The strength of dipole-dipole interactions depends critically on the orientation of the molecules and the signs of the partial charges. Think of dipoles as having a δ+ end and a δ- end:
- Attractive orientations: When opposite charges align (δ+ to δ-), the force is attractive. The head-to-tail arrangement maximizes this attraction.
- Repulsive orientations: When like charges face each other (δ+ to δ+ or δ- to δ-), the force is repulsive.
- Side-by-side orientations: These create a mix of attractive and repulsive forces, resulting in weaker overall interactions.
You can understand these qualitatively: opposite charges attract, like charges repel. In liquids and gases, molecules rotate freely, so we observe an average of all possible orientations, but the attractive arrangements dominate because they lower the system's energy.
This also means that the more polar the molecules, the greater the dipole-dipole attraction. The stronger the attraction, the higher the melting and boiling points of the sample.
AP Question - LDFs + Dipole-Dipole
The following question is from the 2018 AP Chemistry examination posted online by College Board.
The table above gives the molecular structures and boiling points for the compounds CS2 and COS. In terms of the types and relative strengths of all intermolecular forces in each compound, explain why the boiling point of CS2 (l) is higher than that of COS (l).
Sample Response: CS2 and COS both have London Dispersion Forces, but since COS is a polar molecule, it also exhibits dipole-dipole forces. However, the London Dispersion Forces in CS2 are so strong that they overpower the strength of both the LDFs and the dipole-dipole forces in COS. Therefore, CS2 has a higher boiling point.
🧠 Keep in mind that even though LDFs are the weakest forces, they can overpower dipole-dipole forces. It all depends on the size of the molecule; when there are many London dispersion forces present, they can be stronger.
You will not be expected to predict when the LDFs are stronger than the dipole-dipole forces present, but you will rather be given information that tells you this. In this question, you are given the boiling points to tell you that the intermolecular forces present in CS2 are stronger than those in COS.
Dipole-Induced Dipole Interactions
Dipole-induced dipole interactions are a distinct type of intermolecular force that occurs between polar and nonpolar molecules. When a molecule with a permanent dipole approaches a nonpolar molecule, the electric field of the dipole distorts the electron cloud of the nonpolar molecule, inducing a temporary dipole in it.
Key characteristics of dipole-induced dipole forces:
- They are always attractive (unlike dipole-dipole, which can be repulsive)
- Their strength depends on:
- The magnitude of the permanent dipole moment
- The polarizability of the nonpolar molecule
- The distance between molecules
- They are generally weaker than dipole-dipole interactions but stronger than London dispersion forces between small molecules
For example, when oxygen gas (O₂, nonpolar) dissolves slightly in water (H₂O, polar), the water's permanent dipole induces temporary dipoles in the O₂ molecules. The partial charges on water orient the electron cloud in oxygen, creating a weak attraction. This explains why even nonpolar substances have some (though limited) solubility in polar solvents.
Hydrogen Bonding
Hydrogen bonding is really everyone's favorite! It's the easiest to identify and the strongest in pure substances, so they really stick in your mind throughout the whole year.
Hydrogen bonding (which is NOT a bond) is actually an unusually strong dipole-dipole attraction and only occurs when hydrogen is directly bonded to F, O, or N in a molecule. It occurs between these molecules because of their high electronegativity difference and small sizes, which leads to really really strong attractions.
Remember "Hydrogen bonding is FON," or fun, and that it only occurs in polar molecules!
The hydrogen bonded to the F, O, or N is partially positively charged and is attracted to the neighboring unshared electrons on the F, O, or N.
Since these attractions are super strong, the boiling points of molecules with hydrogen bonds are very high! An example of a molecule that has hydrogen bonding is water, which is part of why it takes so long for water to boil. The amount of energy needed to break the intermolecular forces between water molecules when boiling is relatively high.
Importantly, hydrogen bonding can occur not just between different molecules but also between different parts of the same molecule (intramolecular hydrogen bonding). This is particularly common in large biomolecules:
- In proteins, hydrogen bonds between different parts of the polypeptide chain help create and stabilize secondary structures like α-helices and β-sheets
- In DNA, intramolecular hydrogen bonding can occur in single-stranded regions, forming hairpin loops and other structures
- In carbohydrates, OH groups on different carbons can hydrogen bond within the same molecule
These intramolecular hydrogen bonds are crucial for the three-dimensional shapes of biomolecules, which determine their biological functions.
Let's review the misconception once more before moving on:
On the AP Exam, you may be asked to draw a molecule with the proper orientation and create hydrogen bonding. Make sure you don't confuse the difference between intermolecular and intramolecular. Remember, intermolecular is in between two molecules. Fiveable's here to help!
🎥 Watch Jacob Jeffries review polarity and discuss dipole-dipole forces, hydrogen bonding, and London dispersion forces. He reviews their significance to physical properties, along with applications in biomolecules.
Ion-Dipole Forces
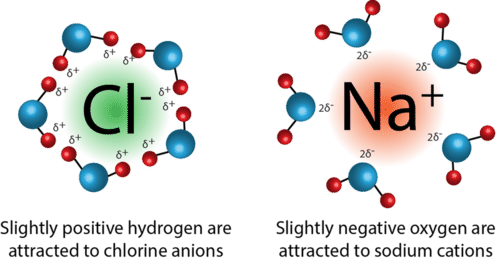
Ion-dipole attractions only occur in a mixture of an ionic compound and polar molecules. These attractions occur when ions (cations or anions) are attracted to the positive or negative end of a dipole.
Overall, this IMF is stronger than dipole-dipole and H-Bonding.
Image Courtesy of ck12.orgAn example of ion-dipole forces is when NaCl is dissolved in a sample of water. Once the NaCl dissolves, it separates into its positive sodium cation and the respective negative chloride anion. From here, the positive end of the water molecule (hydrogen) is attracted to the negative chloride anion, while the negative end of the water molecule (oxygen) is attracted to the positive sodium cation.
Let's break it down further: when we take a look at a water molecule, oxygen has a partial negative charge. This is due to its high electronegativity, and it more strongly attracts electrons toward it. This unequal distribution of electrons results in partial positive and negative charges, or a dipole. The charges produced in this dipole can then attract the ions produced by the dissolution of NaCl.
Ion-Ion Attractions
Ion-Ion attractions occur in a sample of ionic compounds. This is the strongest type of attraction because there aren’t partial charges, but instead full charges on the ions.
These attractions hold the ions together in a solid crystal lattice, giving rise to the high melting and boiling points of ionic compounds. Think about the formation of NaCl, where the sodium atom donates one of its electrons to the chloride atom, forming a sodium cation and chloride anion. The positive sodium ion is attracted to the negative chloride anion through ion-ion attractions.
These forces can be confused with ion-dipole interactions, but try to think about each example given. Ion-dipole interactions are present when NaCl dissolves in water, but ion-ion interactions are present within a solid sample of NaCl itself.
👉 Not sure what a crystal lattice is? Check out our study guide about the structure of ionic solids.
Noncovalent Interactions in Biomolecules
In large biomolecules like proteins and DNA, noncovalent interactions play a crucial role in determining their three-dimensional structure and function. These interactions can occur between different molecules or between different parts of the same large molecule.
For example, in proteins:
- Hydrogen bonds form between the backbone atoms and between side chains, helping to create alpha helices and beta sheets
- Ionic interactions occur between charged amino acid side chains (like lysine and aspartate)
- London dispersion forces exist between nonpolar amino acid side chains, creating hydrophobic pockets
- Dipole-dipole interactions occur between polar side chains
In DNA, hydrogen bonding between complementary base pairs (A-T and G-C) holds the two strands together, while stacking interactions between the aromatic bases involve London dispersion forces. The overall shape and stability of these biomolecules depend on the cumulative effect of many weak noncovalent interactions working together.
These interactions are particularly important because they're strong enough to maintain structure but weak enough to be broken and reformed when needed for biological processes like DNA replication or protein folding.
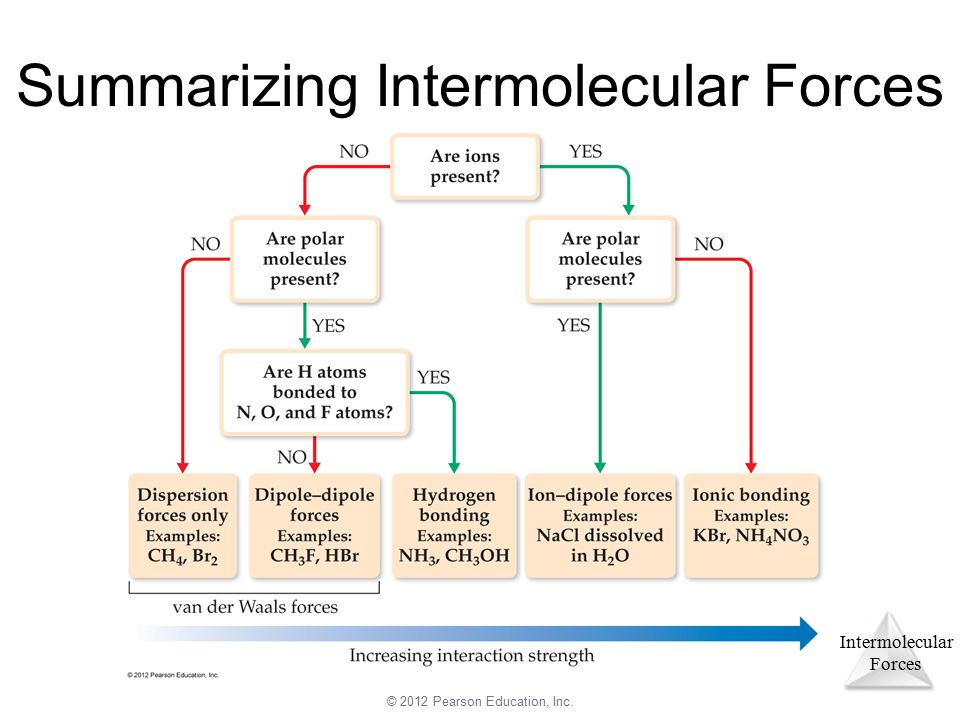
Summary of IMFs
You may be wondering how we can determine the dominant IMF - well it’s quite easy with this simple diagram:
Image Courtesy of Pearson Education🎥 Watch AP Chemistry teacher Wes Winter discuss the importance of intermolecular forces and how the strength of these forces affects the bulk scale properties.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
biomolecule | A large organic molecule such as a protein, nucleic acid, carbohydrate, or lipid that is essential to living organisms. |
contact area | The surface area between molecules that affects the strength of London dispersion forces. |
dipole moment | A measure of the separation of positive and negative charge in a polar molecule. |
dipole-dipole interactions | Intermolecular forces between polar molecules resulting from the attraction between their permanent dipoles. |
dipole-induced dipole interactions | Attractive forces between a polar molecule and a nonpolar molecule, where the polar molecule induces a temporary dipole in the nonpolar molecule. |
electron cloud | The region surrounding an atom or molecule where electrons are distributed. |
electronegative atom | An atom with a strong tendency to attract electrons in a covalent bond, such as nitrogen, oxygen, or fluorine. |
hydrogen bonding | A strong intermolecular force occurring when hydrogen atoms bonded to highly electronegative atoms (N, O, F) are attracted to the negative end of a dipole in another molecule or region. |
intermolecular forces | Attractive forces between separate molecules or particles that determine many properties of liquids and solids, including boiling point, melting point, and vapor pressure. |
ion-dipole interactions | Attractive forces between an ion and a polar molecule, where the charged ion interacts with the partial charges on the polar molecule. |
London dispersion forces | Intermolecular forces resulting from Coulombic interactions between temporary, fluctuating dipoles in molecules. |
noncovalent interactions | Weak attractive or repulsive forces between molecules or regions of molecules that do not involve the breaking or formation of covalent bonds. |
nonpolar molecule | A molecule with no net dipole moment due to symmetrical charge distribution. |
partial charge | A fractional electric charge on an atom within a molecule due to unequal sharing of electrons in a covalent bond. |
pi bonding | Covalent bonding formed by the sideways overlap of p orbitals, which enhances polarizability. |
polar molecule | A molecule with an uneven distribution of charge, resulting in a net dipole moment. |
polarizability | The ability of a molecule's electron cloud to be distorted, creating a temporary dipole in response to an external electric field. |
temporary dipole | A brief, fluctuating separation of charge in a molecule caused by uneven electron distribution at any given moment. |
van der Waals forces | A broad category of weak intermolecular forces including London dispersion forces and dipole-dipole interactions. |
Frequently Asked Questions
What is the difference between intermolecular forces and intramolecular forces?
Intramolecular forces hold atoms together inside a molecule—think covalent bonds (shared electrons) or ionic bonds—and determine molecular identity and bond energies. Intermolecular forces are the weaker, noncovalent attractions between separate molecules or particles: London dispersion (temporary fluctuating dipoles), dipole–dipole, dipole–induced dipole, ion–dipole, and hydrogen bonding (H bonded to N, O, or F interacting with a lone pair). Intramolecular >> intermolecular in strength; breaking intramolecular bonds changes chemical identity, while overcoming intermolecular forces changes phase or solubility. For AP Chem, you should connect molecular structure (size, electron count, pi bonds, polarity, geometry) to relative intermolecular strengths (dispersion increases with polarizability and contact area; H-bonding and ion–dipole are stronger) as called for in Topic 3.1 (CED 3.1.A). For a concise study guide and practice questions tied to this topic, see Fiveable’s Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3). For more practice, check Fiveable’s AP Chem practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do London dispersion forces actually work and why do bigger molecules have stronger ones?
London dispersion forces come from tiny, momentary imbalances in an electron cloud that create instantaneous dipoles. Those temporary dipoles induce opposite dipoles in nearby molecules and the resulting Coulombic attractions (partial + next to partial −) pull molecules together. Bigger molecules have stronger dispersion forces because they have more electrons and a larger, more easily distorted electron cloud (greater polarizability), and they can touch across a larger contact area—both increase the instantaneous dipole strength. Extended pi systems (more delocalized electrons) also boost polarizability, so long, flat, or branched molecules show stronger LDF. On the AP exam this is Topic 3.1: explain how structure (size, electron count, contact area, pi bonds) controls dispersion strength (CED 3.1.A.1). For a focused review see the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and for extra practice try the AP problem set collection (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about dipole-dipole forces - can someone explain them in simple terms?
Dipole-dipole forces are just the attraction between polar molecules—think of each molecule as a tiny bar magnet with a partially positive end (δ+) and a partially negative end (δ–). When two polar molecules get near each other, the δ+ end of one lines up near the δ– end of the other and they attract. These forces add on top of London dispersion forces, so polar molecules of similar size usually stick together more than nonpolar ones. Strength depends on the size of the dipoles (bigger partial charges → stronger attraction) and on orientation—the alignment matters. Dipole-dipole < ion-dipole and generally > dipole-induced-dipole for comparable molecules. This idea is in AP Topic 3.1 (CED 3.1.A.2b–c); it helps explain things like higher boiling points for polar liquids. For a quick review, check Fiveable’s Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What makes hydrogen bonding so much stronger than other intermolecular forces?
Hydrogen bonding is stronger than ordinary dipole–dipole or dispersion forces for two main reasons. First, H bonded to a very electronegative atom (N, O, or F) creates a very large partial positive charge on H and a large partial negative on the N/O/F, so the Coulombic attraction between molecules is large (CED 3.1.A.4; keywords: electronegative atoms, partial charges). Second, hydrogen is just one proton with a tiny electron cloud, so the H can approach the lone pair on a neighboring N/O/F unusually closely—shorter distance means a much stronger interaction (Coulomb’s law, orientation dependence). That short distance also gives hydrogen bonds some partial covalent/directional character, making them stronger and more orientation-dependent than regular dipole–dipole interactions. You’ll see this show up in much higher boiling points and stronger association (important for AP-style reasoning questions). For a quick review, check the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do nonpolar molecules still have intermolecular forces if they don't have dipoles?
Even nonpolar molecules have intermolecular forces because their electron clouds aren’t frozen—they fluctuate. At any instant the electrons can be unevenly distributed, creating a temporary (instantaneous) dipole in one molecule that induces a dipole in a neighbor. The resulting Coulombic attraction is called a London dispersion force (fluctuating/temporary dipoles). Dispersion strength increases with polarizability (bigger, more electrons, larger electron clouds or lots of π-bonding) and with contact area between molecules, so large nonpolar molecules can have sizable attractions. On the AP exam you should be able to explain dispersion forces in these CED terms (temporary dipoles, polarizability, contact area) and compare them to dipole-dipole or dipole-induced dipole interactions. Want a quick review or practice? Check the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and hit the practice problems page (https://library.fiveable.me/practice/ap-chemistry) for exam-style questions.
What's the difference between van der Waals forces and London dispersion forces?
Short answer: van der Waals forces is a broad label for the weak, noncovalent attractions between molecules (it commonly refers to London dispersion, dipole–dipole, and dipole–induced dipole interactions). London dispersion forces are one specific type of van der Waals force: they arise from temporary, fluctuating dipoles (instantaneous electron-cloud distortions) and are present in every molecule. Key differences to remember (CED-aligned): - Scope: “van der Waals” = umbrella term; “London dispersion” = one mechanism under that umbrella. The CED explicitly warns not to use them synonymously. - Origin: dispersion = Coulombic attraction between instantaneous dipoles; dipole–dipole and dipole–induced are due to permanent or induced dipoles. - Strength trends: dispersion increases with polarizability (more electrons, bigger electron cloud, more pi bonds) and contact area, and for large molecules it can be the dominant intermolecular force. For more AP-aligned review, see the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
How do I know which intermolecular force is strongest between two different molecules?
Start by ranking the types of forces present between the two species—the strongest relevant force usually dominates. Quick checklist (in order): 1. Ion present? If yes, ion–dipole (or ion–ion) beats everything else. 2. Can either molecule hydrogen-bond? If one has H covalently bonded to N, O, or F and the other has a lone pair on N/O/F, hydrogen bonding is next-strongest. 3. Are both molecules polar? If so, dipole–dipole interactions add on top of dispersion. Larger dipole magnitude → stronger dipole–dipole. 4. If one is polar and the other nonpolar, dipole-induced dipole matters (stronger when polarizability is high). 5. Otherwise, London dispersion forces dominate—but remember dispersion gets much stronger with larger size, more electrons, greater contact area, or lots of pi bonding (so big nonpolar molecules can have the strongest net IMF). Also consider orientation dependence (dipole interactions depend on alignment) and polarizability (bigger electron clouds → stronger dispersion). For AP Topic 3.1 problems use this hierarchy and cite structure: look for ions → H-bond donors/acceptors → polarity → size. For a review, see Fiveable’s Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
Can you explain what polarizability means and why it matters for intermolecular forces?
Polarizability is how easily an atom or molecule’s electron cloud can be distorted to form a temporary (instantaneous) dipole. Bigger species with more electrons and larger, more diffuse electron clouds are more polarizable; pi bonds also make clouds easier to distort. That matters because London dispersion forces come from Coulombic attractions between these temporary dipoles. So, for molecules of similar shape, more polarizable = stronger dispersion forces = higher boiling point, greater surface contact, and stronger dipole-induced interactions with polar partners (CED 3.1.A.1–A.2). On the AP exam, use this idea to explain trends: e.g., I2 (large, polarizable) has stronger dispersion than Cl2; larger nonpolar molecules mix less with small polar ones because dipole-induced dipole strength depends on polarizability (CED language). For targeted review and practice problems on these ideas, see the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and more practice questions (https://library.fiveable.me/practice/ap-chemistry).
What are ion-dipole forces and when do they occur?
Ion-dipole forces are electrostatic attractions between a full ion (like Na+ or Cl–) and the partial charge on a polar molecule (like H2O). They occur whenever an ionic species interacts with polar molecules—for example, when salt dissolves in water the Na+ is attracted to the oxygen end (δ–) of H2O and Cl– to the hydrogen ends (δ+). These interactions are orientation-dependent (how the dipole lines up with the ion) and are typically stronger than dipole–dipole forces because a full ionic charge interacts with a partial charge. Ion–dipole forces help explain solubility of ionic compounds in polar solvents and appear in CED Essential Knowledge 3.1.A.2.c and 3.1.A.3. For a quick review, see the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01), and practice problems are at (https://library.fiveable.me/practice/ap-chemistry).
Why does contact area between molecules affect the strength of dispersion forces?
Dispersion (London) forces come from instantaneous fluctuations in an electron cloud that create temporary dipoles; those dipoles induce opposite dipoles in nearby molecules and attract each other (CED: London dispersion, fluctuating dipoles, polarizability). Contact area matters because larger surface contact lets more of those instantaneous charge fluctuations overlap—more opposite partial charges can line up across the two molecules at once. That increases the total Coulombic attraction summed over the contact region, so flattened or elongated molecules (larger contact area) have stronger dispersion forces than compact, spherical ones of the same mass. Polarizability and electron-cloud size also matter: bigger, more polarizable clouds amplify those fluctuations (CED: polarizability increases with electron number and pi bonds). This idea shows up on AP problems comparing boiling points or solubilities—review Topic 3.1 in the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
I don't understand dipole-induced dipole interactions - how does a polar molecule affect a nonpolar one?
A polar molecule has a permanent dipole (partial + and −). When it comes near a nonpolar molecule, its electric field pulls the nonpolar molecule’s electrons a bit toward the positive or negative end, temporarily shifting that electron cloud. That temporary shift creates an induced dipole in the nonpolar molecule with the opposite sign near the polar end, so the two attract. These dipole-induced dipole interactions are always attractive and add on top of London dispersion forces. Their strength increases if the polar molecule has a larger dipole moment and if the nonpolar molecule is more polarizable (bigger, more electrons, or more diffuse electron cloud). This idea is in Topic 3.1 (CED 3.1.A.2.a)—review the Intermolecular Forces study guide on Fiveable for examples and diagrams (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01). For more practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
How do intermolecular forces work in proteins and other big biomolecules?
Proteins and big biomolecules are held together mostly by noncovalent intermolecular forces described in the CED. Locally, hydrogen bonds (N–H and O–H to lone pairs on N/O) and dipole–dipole interactions stabilize secondary structures (α-helices, β-sheets). Oppositely charged side chains form ionic (ion–dipole/ion-pair) attractions. Over large surfaces, London dispersion forces become very important: bigger electron clouds and greater contact area (and pi systems in aromatic side chains) increase polarizability and net dispersion attraction. These forces act between different molecules and between distant regions of the same molecule to fold and maintain tertiary/quaternary structure. Orientation matters for dipoles and H-bonds; dispersive forces are less orientation-dependent but scale with size. For AP review, focus on how polarizability, contact area, dipoles, and H-bond donors/acceptors determine relative strengths (see Topic 3.1 CED). For a targeted study, check the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01), the unit overview (https://library.fiveable.me/ap-chemistry/unit-3), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the deal with hydrogen bonding only happening with N, O, and F atoms?
Hydrogen bonding only happens when H is covalently bonded to a very electronegative, small atom (N, O, or F) because two things must coincide: a large partial positive on H and a lone pair on a strongly electronegative acceptor. N, O, and F pull electron density away from H, making Hδ+ very exposed; their small size concentrates negative charge in lone pairs so the H···A distance is short and the Coulombic attraction is strong (see CED 3.1.A.4 and keywords: partial charges, electronegativity). Bigger or less electronegative atoms (like Cl, S) don’t create as large a Hδ+ or as tight an interaction, so you get only weaker dipole or dispersion forces. Consequences: H-bonding raises boiling points and solubility (important for comparing molecules on AP free-response/multiple-choice for Topic 3.1). For a focused review, see the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do you predict which substance will have a higher boiling point based on intermolecular forces?
Boiling point increases when intermolecular forces (IMFs) are stronger—more energy is needed to separate molecules. To predict which substance boils higher, compare IMFs using this checklist: 1. Type of IMF (strongest → weakest for neutral species): ion–dipole/ionic > hydrogen bonding (H bonded to N, O, F) > dipole–dipole > London dispersion. 2. For dispersion forces, bigger = stronger: higher molar mass, larger electron cloud, more pi-bonding, and greater contact area increase polarizability and dispersion strength. 3. If two molecules have the same IMFs, the one with greater molar mass or more surface area usually has the higher bp. 4. Polar molecules have dipole–dipole in addition to dispersion, so they often boil higher than similar-size nonpolar molecules. 5. Ionic compounds (or solutions with ions) have very high bps because of ion–dipole/ionic attractions. Use AP vocabulary: London dispersion, polarizability, dipole–dipole, ion–dipole, hydrogen bonding. For more examples and practice, see the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why are intermolecular forces important in real life and what do they actually do?
Intermolecular forces (IMFs) are what make molecules stick to each other—they control macroscopic properties you care about. Stronger IMFs (hydrogen bonding, ion-dipole, dipole-dipole, and dispersion) raise boiling/melting points, increase viscosity and surface tension, and reduce vapor pressure. They determine solubility (like dissolving salt vs. oil), chromatographic separations, and how salts dissociate (ion-dipole). At the molecular level noncovalent IMFs control protein folding, enzyme–substrate binding, DNA base pairing, and drug-receptor affinity—so biological function and drug design depend on them. Key trends: dispersion increases with polarizability and contact area; dipole interactions depend on dipole magnitude and orientation; hydrogen bonds require H bonded to N, O, or F. On the AP exam Topic 3.1 is part of Unit 3 (18–22% weighting), so be fluent with these concepts and how structure affects IMF strength. For a focused review, check the Topic 3.1 study guide (https://library.fiveable.me/ap-chemistry/unit-3/intermolecular-forces/study-guide/aq9pSoNJoW70eCdben01) and practice problems (https://library.fiveable.me/practice/ap-chemistry).