Remember when we went over matter in unit one? Matter is any physical object that has mass and occupies space. Matter can be classified according to its state (which we'll go over in this study guide), or its composition (the atoms or molecules that make it up).
Gases and solutions are big focuses of this unit, so understanding the content we're going to go over is essential!
Let’s look over some of the characteristic properties of the states of matter:
🧊 Solids
As we learned in the last key topic, solids can either be crystalline or amorphous. Crystalline solids have a structure and 3-D order, while amorphous solids have considerable disorder within their structures.
Regardless of the type of solid, all solids retain their own shape and volume. They don’t expand to fill their container because the particles in a solid are packed very closely together and cannot move. The intermolecular forces are strong enough to keep the particles in place.
Although particles in a solid cannot move, they vibrate back and forth!
In general, solids are virtually incompressible, don’t flow, and the diffusion process within a solid occurs extremely slowly. Compressibility is the degree to which a material can be squeezed, or a measure of volume change when pressure is applied to a substance.

💧Liquids
Liquids assume the shape of a portion of the container it occupies. Liquids do NOT expand to fill their container, they just fill the space that is provided by the container.
Since liquid particles aren't tightly packed together, they have the ability to flow past one another (fluidity). The intermolecular forces are strong enough to hold the molecules closely together, but not strong enough to hold them in place.
Liquids are virtually incompressible like solids, flow readily, and diffusion within a liquid occurs slowly.
The solid and liquid phases of a particular substance typically have similar molar masses since the particles are closely held together.
Surface Tension
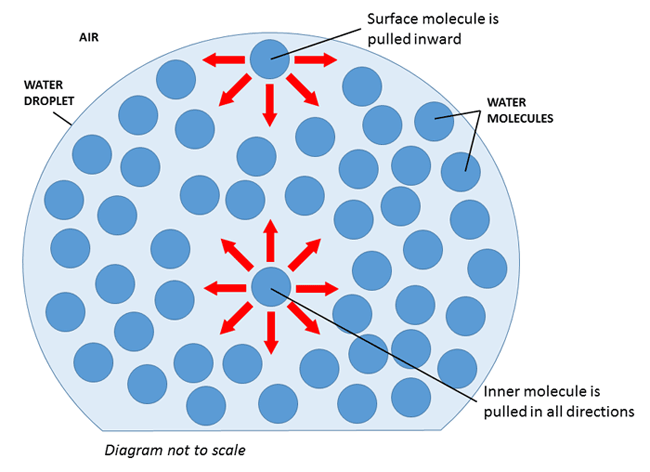
Surface tension refers to the tendency of liquids to minimize their surface area. It results from an imbalance of intermolecular forces when molecules on a surface of a liquid experience a net inward force.
Image Courtesy of PinterestThe interior, or bulk, molecules are attracted to particles in all directions, while surface molecules only experience attraction in lateral and downward directions. This causes surface molecules to be less stable, so liquids attempt to reduce the number of surface molecules they have. They do this by minimizing their surface area!
There are two trends with surface tension that you should note:
- The stronger the intermolecular forces experienced, the higher the surface tension. This is because the surface molecules resist penetration, which increases surface area.
- The higher the temperature, the lower the surface tension. This goes into unit five and the movement of particles, but the higher the temperature, the easier it is to stretch the surface of particles.
Capillary Action
Capillary action is the spontaneous rising of a liquid against gravity. It often happens with polar liquids that have strong IMFs. You could see this when you put a paper towel in contact with a puddle; the water will slowly rise up the towel due to capillary action.
There are two different types of forces involved in capillary action:
- Cohesive Forces: forces between the liquid molecules that hold the liquid together
- Adhesive Forces: forces between liquid molecules and the container
Capillary action occurs because the adhesive forces pull the surface molecules up and the cohesive forces pull the bulk molecules up with it. These two forces work together for it to occur!
On the other hand, the meniscus is due to the competition between these two forces.
Remember the typical meniscus you see when you do experiments? The meniscus of water is concave because the adhesive forces are stronger than the cohesive forces, so the water is more strongly attracted to the graduated cylinder than itself
Image Courtesy of BartlebyI bet you've never seen Mercury in a glass container! Well, that creates an upside-down meniscus, which is called a convex meniscus. This occurs because the forces between the mercury molecules are stronger than the forces between the molecules and the glass (cohesive > adhesive).
Viscosity
Viscosity is a measure of a liquid's resistance to flow. You see this with syrup every time you eat pancakes.
The stronger the intermolecular forces present, the higher the viscosity, and the thicker the liquid. The higher the temperature of the liquid, the lower its viscosity. This, again, goes back to unit five, but think of temperature as motion and an increased ability to overcome intermolecular forces.
♨️ Gases
Gases assume the volume and shape of their container. Gas particles move rapidly in straight lines; more about the behavior of gases is covered in the rest of this unit!
The molecules have enough energy to overcome any intermolecular forces that exist, allowing them to move freely. They are compressible, flow readily, and expand to fill the container. Diffusion within a gas occurs quite rapidly.
Density
Density measures how compact a substance is! The formula for Density is D = m/V or density equals mass divided by volume.
It is good to remember that solids are usually the most dense out of the three phases and gases are usually the least dense since they flow freely in open space.
Density Practice Question
A student measured the mass of a sealed 644 mL flash that contained air. The student then flushed the flask with an unknown gas, resealed it, then measured the mass again. The air and the unknown gas were at STP. Calculate the mass of the unknown gas. The density of air at STP is 1.29 g/L.
| Volume of sealed flash | 644 mL |
|---|---|
| Mass of Sealed flask and air | 121.03 g |
| Mass of Sealed Flask and Unknown Gas | 122.60 g |
Before answering the question, let's note that the air is in a sealed flask. This means that it cannot escape into the atmosphere so the most it could do is fill the shape of the container.
Also, don't worry about STP yet, we'll go over that in the next key topic.
- First Step - Find the mass of the air: Since we are given both the density of the air and the volume of the sealed flask, we can find the mass. However, they give us a density in g/L and a volume in mL, so first we have to convert 644 mL into 0.644 L by simply moving the decimal 3 to the left. D = M/V --> 1.29 = M/.644 --> M = 0.831 g
- Second Step - Find the mass of the flask: Since we now know the mass of the air and the mass of both the air and the flask, we can just subtract: 121.03 g - 0.831 g = 120.20 g
- Final Step - Find the mass of the unknown gas: They have given us the mass of both the sealed flask and the unknown gas. Since we know the mass of the sealed flask alone, we can just subtract and get our final answer! 122.60 g - 120.20 g = 2.40 g
Overview
To remember the differences between a solid, liquid, and gas, think about your own experiences in life. For example, liquids, like water, will flow freely whereas a solid, like ice, maintains its shape no matter what container it is in.
🎥 Watch Catherine Liu explain the differences and phase changes between solids, liquids, and gases. She also goes over properties such as fluidity, density, compressibility, surface tension, and viscosity.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
amorphous solid | A solid in which particles do not have a regular or orderly arrangement. |
collision frequency | The number of collisions between reactant particles per unit time. |
crystalline solid | A solid in which particles are arranged in a regular, repeating three-dimensional structure. |
gas | A phase of matter in which particles are in constant motion with minimal intermolecular forces, resulting in no definite volume or shape. |
hydrogen bonding | A strong intermolecular force occurring when hydrogen atoms bonded to highly electronegative atoms (N, O, F) are attracted to the negative end of a dipole in another molecule or region. |
interparticle interactions | Forces between particles in a system that affect the energy changes during physical and chemical processes. |
liquid | A phase of matter in which particles are in close contact and in continual motion and collision with one another. |
molar volume | The volume occupied by one mole of a substance; typically similar between solid and liquid phases because particles are in close contact. |
particulate model | A representation of matter showing individual atoms, molecules, or ions and their interactions to describe chemical processes at the molecular level. |
polarity | The distribution of electric charge in a molecule, determining its ability to interact with polar and nonpolar substances. |
Frequently Asked Questions
What's the difference between crystalline and amorphous solids?
Crystalline solids have particles (atoms, ions, or molecules) arranged in a regular, repeating 3-D lattice (think ionic lattices, covalent-network solids, or metals). That long-range order gives definite melting points, predictable packing efficiencies, and directional properties. Amorphous solids lack that long-range order—the particles are disordered like a very slow liquid frozen in place (glass, many plastics). In both types particle motion is limited (no overall translation), but crystalline solids show repeating symmetry at the particulate level while amorphous solids do not. On the AP exam you should be able to represent these differences with particulate models and connect structure to properties (melting behavior, packing, and how intermolecular/interparticle forces matter). For a quick refresher, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do particles move differently in solids, liquids, and gases?
In solids the particles are tightly packed and can only vibrate about fixed positions—they don’t translate past one another. Crystalline solids have an ordered 3-D lattice; amorphous solids lack long-range order, but in both the motion is very limited (linked to strong interparticle forces and packing). In liquids particles are still in close contact but can move and translate past one another; they constantly collide and flow, so a liquid has a definite volume but no fixed shape. In gases particles are far apart, move constantly in straight lines between collisions, and have large mean free paths; intermolecular forces are negligible, so gases have neither definite shape nor definite volume (collision frequency and spacing depend on T, P, and V). These particulate descriptions are exactly what AP wants you to be able to represent (CED 3.3.A). Review this topic in the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do solids and liquids have similar molar volumes but gases don't?
Solids and liquids have similar molar volumes because in both phases the particles stay in close contact—packing is what sets their volume. For example, liquid water is about 18 mL per mole, and most solids/liquids are tens of mL per mole (same order of magnitude) because intermolecular/interparticle forces hold molecules or ions near one another (packing efficiency, lattice structures, hydrogen bonding, dispersion). Gases are totally different: at STP one mole of any ideal gas occupies ~22.4 L (22,400 mL), so the average spacing between particles is thousands of times larger. Kinetic molecular theory explains this—gas particles move freely with negligible persistent attractions, so volume is set by temperature and pressure (and gases are highly compressible), not by packing. This difference is exactly what the CED emphasizes for Topic 3.3. For a quick review, see the Topic 3.3 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about particle arrangement - can someone explain how particles are organized in each phase?
Think of each phase at the particle level: - Solid: particles are packed closely and don’t translate past each other. In crystalline solids they sit in a regular 3-D lattice (ionic lattice, covalent network, metallic), while amorphous solids lack long-range order. Motion is mostly vibration; strong interparticle forces and packing determine lattice energy and low compressibility. - Liquid: particles are still in close contact but move and slide past one another—continual collisions and translation give a definite volume but no fixed shape. Strength/type of intermolecular forces (polarity, hydrogen bonding, London dispersion) and temperature set viscosity, surface tension, and similar molar volume to the solid. - Gas: particles are far apart, move constantly, and collisions are infrequent. Forces between particles are negligible; volume and shape are not fixed. Mean free path, diffusion, and pressure depend on T, V, and P (Kinetic Molecular Theory). For more AP-aligned review, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and extra practice problems (https://library.fiveable.me/practice/ap-chemistry).
What forces affect how particles move in liquids?
In liquids, particle motion is controlled by a balance between kinetic energy (temperature) and intermolecular forces. Higher T gives particles more kinetic energy so they move, collide, and diffuse faster; lower T slows them and increases order. Intermolecular forces keep particles close and determine how freely they move: London dispersion (present in all molecules), dipole–dipole attractions (in polar molecules), and hydrogen bonding (strong, in H–O, H–N, H–F) all reduce mobility, raise viscosity and surface tension, and lower vapor pressure. These forces also explain why liquids have similar molar volumes to solids (close contact) but no fixed shape. On the AP exam you should be able to draw particulate models and link polarity/H-bonding to properties like viscosity, diffusion, and vapor pressure (Topic 3.3 in the CED). For a quick review, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and more unit resources (https://library.fiveable.me/ap-chemistry/unit-3). Practice problems are at (https://library.fiveable.me/practice/ap-chemistry).
Why don't gas particles have definite shape or volume?
Gas particles don’t have a definite shape or volume because, at the particulate level, they move constantly and are far apart so intermolecular forces are negligible. According to the kinetic molecular model (CED Topic 3.3 / keywords: kinetic molecular theory, mean free path), gas molecules translate rapidly in all directions, colliding only occasionally. Because the average spacing between particles is large and their motion isn’t restricted by neighbors, a gas expands to fill whatever container it’s in (no fixed shape) and its particles can change how much space they occupy with pressure or temperature changes (no fixed volume). Contrast that with solids/liquids where particles stay in close contact so shape/volume are defined by packing and intermolecular forces (polarity, hydrogen bonding, London dispersion). For more AP-aligned review on particulate models and gas behavior, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC). You can also practice related problems at Fiveable’s practice page (https://library.fiveable.me/practice/ap-chemistry).
How does temperature affect particle collision frequency in gases?
Raising the temperature increases the average kinetic energy of gas particles, so they move faster. Faster particles hit each other more often, so collision frequency goes up. At constant volume and number of moles, collision frequency is roughly proportional to the average speed, which scales with the square root of temperature (vavg ∝ √T), so doubling T doesn’t double collisions—it increases them by about √2. Also note: collision energy increases too (more energetic impacts), which matters for reaction rates (Unit 5/Kinetics) and for gases’ behavior. If pressure or volume change with temperature, collision frequency will also be affected by particle density (more particles per volume → more collisions). For AP review, tie this to kinetic molecular theory and mean free path in Topic 3.3 and 3.5 (see the Topic 3.3 study guide: https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC). For extra practice, Fiveable has many AP-style problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between how particles move in solids versus liquids?
In solids the particles are held tightly in place by interparticle forces (ionic, covalent network, metallic, or dispersion/H-bonding). In both crystalline and amorphous solids individual particles have only limited motion—they vibrate about fixed positions and don’t undergo overall translation past one another. In liquids the particles are still in close contact (so molar volumes are similar to solids) but have enough kinetic energy to move, collide, and slide/translate around each other; their arrangement is disordered and constantly changing, and movement depends on temperature and intermolecular forces (polarity, H-bonding, London dispersions). AP questions expect you to use a particulate-level model to show these differences (solid = restricted vibrations, liquid = continual translation/collisions). For a quick review see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC), the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do particles in solids stay in place but liquid particles can move around?
Particles in solids stay in place because strong interparticle forces and tight packing lock each particle into a fixed position (crystalline or amorphous). The particles still vibrate, but they don’t undergo overall translation relative to neighbors. In liquids the particles are also in close contact (similar molar volume) but intermolecular forces are weaker or overcome more easily by thermal motion, so particles can move past and slide around each other—you get continual translation and collisions. Temperature (kinetic energy) and the type/strength of forces (polarity, hydrogen bonding, London dispersion) determine how much motion occurs: raise T or weaken attractions and a solid can melt to a liquid. For AP-style particulate models, draw tightly fixed dots for solids vs. closely spaced, mobile dots for liquids (Topic 3.3). More review: see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and extra practice (https://library.fiveable.me/practice/ap-chemistry).
How do intermolecular forces like hydrogen bonding affect the liquid phase?
Hydrogen bonding (a strong type of dipole–dipole interaction) pulls molecules closer and makes them stickier in the liquid phase. On a particle level this means molecules spend more time near each other and need more energy to escape into the gas phase. Observable effects: higher boiling points and lower vapor pressures compared with similar-mass molecules that lack H-bonds; greater viscosity (flows more slowly); higher surface tension; and often more ordered local structure (e.g., transient H-bond networks in liquid water). On the AP exam, describe these changes with particulate (dot/ball) models and link them to intermolecular-force strength (keywords: hydrogen bonding, polarity, vapor pressure, viscosity, surface tension). For a quick review, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and try practice problems (https://library.fiveable.me/practice/ap-chemistry) to apply these ideas.
I don't understand particle packing - how does it influence solid structure?
Think of particle packing as how particles (atoms, ions, molecules) fit together in a solid—that packing controls density, structure type (crystalline vs amorphous), and many macroscopic properties. Close-packed arrangements (FCC, HCP) have high packing efficiency and high coordination numbers, so crystals built that way are denser and often have higher melting points and better metallic conductivity. Less-efficient packings (simple cubic) leave more empty space, lowering density and changing mechanical strength. For ionic solids, packing must also accommodate charge and ionic sizes (lattice type + coordination affect lattice energy), so packing + interparticle forces (ionic, metallic, covalent, H-bonding, London forces) determine whether a solid is a stiff covalent network, a brittle ionic lattice, or a malleable metal. On the AP exam you should be able to represent these differences at the particulate level (CED LO 3.3.A). For a focused review, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC), the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens to particle spacing when you change from liquid to gas?
When a substance goes from liquid to gas, the average spacing between particles increases a lot. In a liquid the particles are in close contact and continually move/ collide, so molar volumes of liquids and solids are similar. In a gas, particles are far apart, move independently, and the mean free path (average distance between collisions) becomes much larger. That’s why gases have no definite shape or volume and are highly compressible—spacing (and collision frequency) depends strongly on temperature, pressure, and volume. On the AP exam you should represent this with a particulate model: show closely packed, touching particles for liquids and widely separated, fast-moving particles for gases (LO 3.3.A; see Topic 3.3 in the CED). For a quick review, check the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why are particles in liquids always colliding with each other?
Particles in a liquid are always colliding because they have thermal kinetic energy and are packed close together. At a given temperature each particle moves randomly (kinetic molecular theory), so with neighbors only a few angstroms away those random motions make frequent impacts. Unlike gases, the mean free path in a liquid is tiny because intermolecular forces hold particles in close contact, so collisions are continuous and transfer momentum and energy between neighbors. Those constant collisions explain liquid properties you study in Topic 3.3—diffusion, viscosity, surface tension, and why liquids have a definite volume but no fixed shape. On the AP exam you should be able to describe this particulate-level picture (LO 3.3.A) using terms like intermolecular forces, kinetic energy, and mean free path. For a short review, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
How does pressure affect particle behavior in the gas phase?
Pressure is just force from particle collisions per area, so changing pressure changes how often and how close gas particles collide. At constant temperature, increasing pressure (usually by decreasing volume) forces particles closer together: mean free path drops, collision frequency rises (roughly proportional to pressure), and collisions with the container increase. Under ideal-gas/kinetic-molecular-theory assumptions the speed distribution (and average kinetic energy) stays the same if T is constant, so pressure changes don’t change molecular speeds—they change how often particles hit things. At very high pressure or low temperature, particles are close enough that intermolecular forces matter, the gas compresses more (deviates from ideal behavior), and you can get condensation to liquid/solid. These ideas link to keywords in the CED (kinetic molecular theory, mean free path, compressibility, intermolecular forces). For a focused review, check the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and practice problems (https://library.fiveable.me/practice/ap-chemistry)—both are useful for AP-style questions.
Can someone explain why gases expand to fill their container but solids and liquids don't?
Think of the three phases at the PARTICLE level (that’s exactly what the CED wants you to be able to represent). In solids particles are tightly packed and held in place by strong interparticle forces (ionic/covalent/metallic or network/dispersion, etc.), so they don’t translate past each other—definite shape and volume. In liquids the particles are still in close contact (similar molar volume to solids) but have enough kinetic energy to move/slide and collide; surface tension and viscosity matter, but the attractive forces still keep a definite volume. In gases, particles have much higher kinetic energy, much larger average spacing (long mean free path), and intermolecular forces are negligible, so they move freely and uniformly fill whatever container volume they can access—no definite shape or volume (kinetic molecular theory). For more AP-aligned visuals and practice, see the Topic 3.3 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solids-liquids-gases/study-guide/uNMmoAyjJQkL63ysUrJC) and Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3).