Types of Chemical Bonds
Chemical bonding explains why atoms stick together to form compounds and molecules. The type of bond that forms depends on how atoms interact with each other's electrons: do they transfer them, or share them? That single distinction creates substances with very different properties.

Understanding Chemical Bonds and Electronegativity
Electronegativity is a measure of how strongly an atom attracts electrons in a chemical bond. The difference in electronegativity between two atoms is what determines the type of bond they form.
- A large electronegativity difference (greater than 1.7) typically leads to ionic bonding, where one atom pulls electrons away from the other entirely.
- A small electronegativity difference (less than 1.7) usually results in covalent bonding, where atoms share electrons instead.
Think of it this way: if two atoms have very different "pulling power," the stronger one just takes the electron. If their pulling power is similar, they share.
Characteristics of Ionic and Covalent Bonds
| Property | Ionic Compounds | Covalent Compounds |
|---|---|---|
| Atoms involved | Metal + non-metal (e.g., NaCl) | Non-metal + non-metal (e.g., ) |
| Melting/boiling points | High | Generally lower |
| State at room temperature | Usually solid | Often liquids or gases |
| Electrical conductivity | Conducts when dissolved or molten | Usually does not conduct |
| Bonding mechanism | Electron transfer | Electron sharing |
The high melting points of ionic compounds come from the strong electrostatic forces holding their ions together in every direction. Breaking those attractions takes a lot of energy. Covalent compounds are held together by weaker intermolecular forces (the attractions between molecules, not the bonds within them), so they melt and boil at lower temperatures.
Ionic Bonding
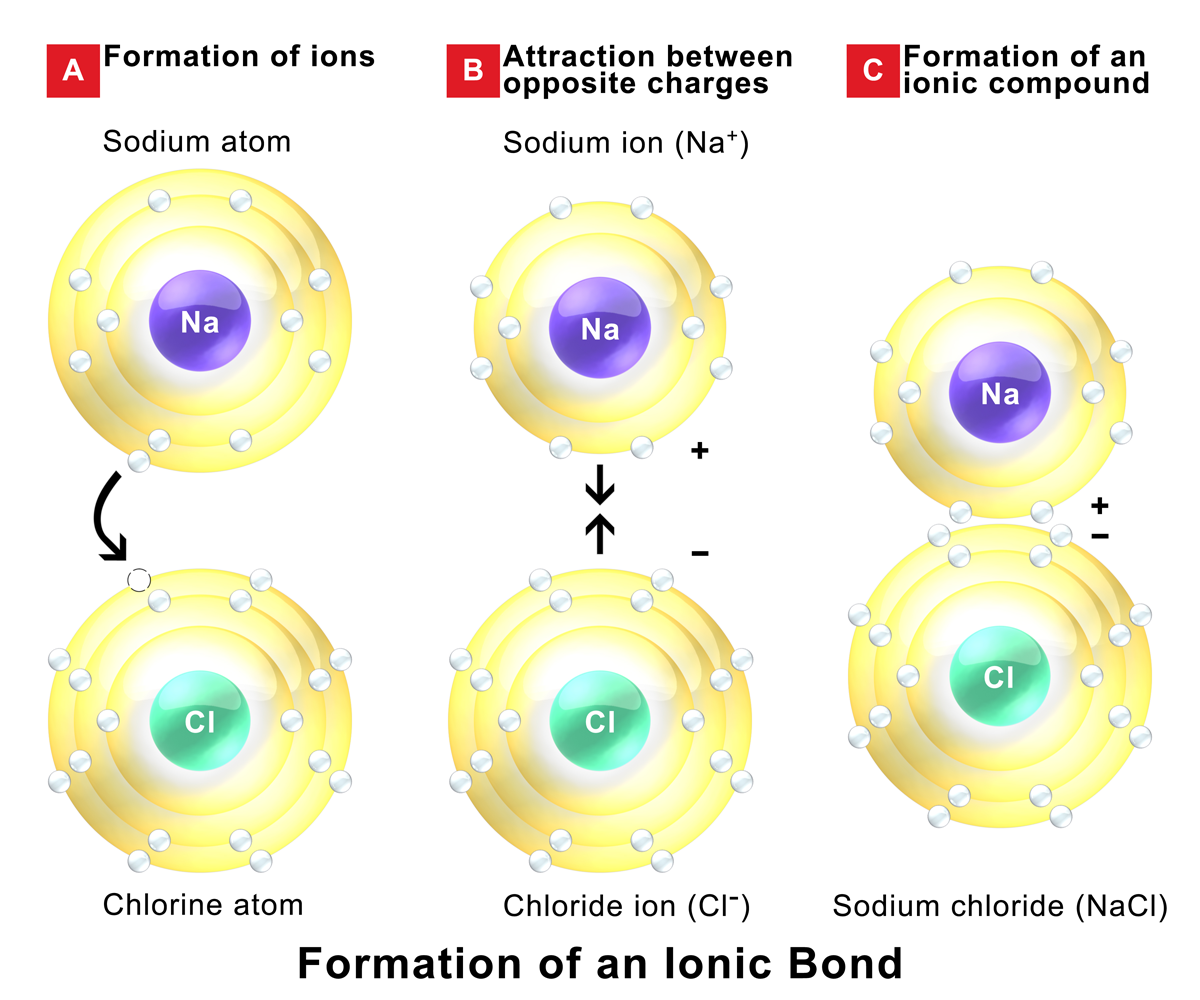
Formation of Ions and Electron Transfer
Atoms form ions when they gain or lose electrons to reach a stable electron configuration, typically matching the nearest noble gas. This stable arrangement is what drives the whole process.
- Cations are positively charged ions. They form when atoms lose electrons. For example, sodium (Na) has 11 electrons but loses one to become , which now has the same electron configuration as neon (10 electrons).
- Anions are negatively charged ions. They form when atoms gain electrons. Chlorine (Cl) gains one electron to become , giving it the same electron configuration as argon (18 electrons).
Electron transfer goes from a metal to a non-metal. The number of electrons transferred depends on how many valence electrons each atom needs to lose or gain. Sodium has 1 valence electron it can give up; chlorine needs 1 to complete its outer shell. That's why they bond in a 1:1 ratio to form NaCl.
For a different example, consider magnesium (Mg) bonding with chlorine. Magnesium has 2 valence electrons to give up, but each chlorine atom can only accept 1. So magnesium needs two chlorine atoms, and the formula is .
Ionic Compound Properties and Structure
Once ions form, the positive and negative charges attract each other and lock into a repeating pattern called a crystalline lattice structure. This isn't just one pair of ions stuck together. It's a huge, orderly 3D arrangement of alternating positive and negative ions.
- Ionic compounds always form in whole-number ratios so the total positive and negative charges balance out. The compound is electrically neutral overall.
- A formula unit represents the simplest whole-number ratio of ions. For sodium chloride, the formula unit is NaCl (one for every one ).
- Many ionic compounds dissolve well in water because water molecules are polar and can pull individual ions away from the lattice. This is also why ionic compounds conduct electricity when dissolved or melted: the ions are free to move and carry charge. In solid form, the ions are locked in place and can't conduct.
Covalent Bonding

Electron Sharing and Lewis Dot Structures
In covalent bonding, atoms share electrons rather than transferring them. This typically happens between two non-metal atoms that have similar electronegativities.
Lewis dot structures are diagrams that help you visualize how valence electrons are arranged in a molecule. Here's how to draw one:
- Count the total valence electrons for all atoms in the molecule. For water (): oxygen brings 6, each hydrogen brings 1, so the total is 8.
- Place the least electronegative atom in the center (hydrogen is never the central atom).
- Draw single bonds (lines) between the central atom and each surrounding atom. Each line represents 2 shared electrons.
- Distribute remaining electrons as lone pairs on the surrounding atoms first, then the central atom, so each atom satisfies the octet rule (or the duet rule for hydrogen).
- If the central atom doesn't have enough electrons for an octet, convert lone pairs on surrounding atoms into double or triple bonds.
Lewis structures are useful because they help you predict how atoms connect, what shape a molecule takes, and whether the molecule is polar.
Octet Rule and Bond Types
The octet rule states that atoms tend to form bonds until they have eight valence electrons in their outer shell. Hydrogen is an exception since it only needs two (sometimes called the "duet rule").
Atoms can share different numbers of electron pairs:
- Single bond: one shared pair (one line). Example: ()
- Double bond: two shared pairs (two lines). Example: ()
- Triple bond: three shared pairs (three lines). Example: ()
As you go from single to double to triple bonds, the bond gets stronger and shorter. More shared electrons means a tighter hold between the atoms.
Some elements in period 3 and beyond (like phosphorus and sulfur) can hold more than eight valence electrons because they have access to empty d orbitals. For a physical science course, just know these exceptions exist.
Molecular Properties and Polarity
Even within covalent compounds, not all bonds share electrons equally. The electronegativity difference still matters:
- Nonpolar covalent bonds form when the electronegativity difference is very small (less than about 0.5). Electrons are shared roughly equally (e.g., , ).
- Polar covalent bonds form when the difference is between about 0.5 and 1.7. One atom pulls the electrons closer, creating a partial negative end () and a partial positive end ().
Whether the overall molecule is polar depends on both the bond polarity and the molecule's shape. For instance, has two polar bonds, but they point in opposite directions and cancel out, making the molecule nonpolar overall. Water () has a bent shape, so its polar bonds don't cancel, and the whole molecule is polar.
Molecular polarity affects real-world properties. Polar molecules like water dissolve other polar substances well and tend to have higher boiling points than nonpolar molecules of similar size. Water also exhibits hydrogen bonding, a particularly strong type of intermolecular force that occurs when hydrogen is bonded to a highly electronegative atom like oxygen, nitrogen, or fluorine. Hydrogen bonding is why water has an unusually high boiling point (100°C) for such a small molecule.