Electrochemical Cell Components and Structure
Electrochemical cells convert chemical energy into electrical energy through spatially separated redox reactions. By splitting oxidation and reduction into two distinct half-cells, the electron transfer is forced through an external circuit, producing usable electrical work. The cell potential and its connection to Gibbs free energy let you predict whether a given redox reaction will proceed spontaneously.
Half-Cells and Electrodes
Each electrochemical cell contains two half-cells, and each half-cell pairs an electrode with an electrolyte solution containing the relevant ionic species.
- The anode is where oxidation occurs. The electrode material (or species in solution) loses electrons, which are released into the external circuit.
- The cathode is where reduction occurs. Electrons arriving from the external circuit are consumed here.
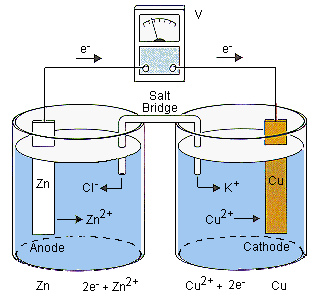
- A classic example is the Daniell cell: a zinc electrode sits in solution (anode), and a copper electrode sits in solution (cathode).
A useful mnemonic: Anode = Away (electrons leave), or "An Ox, Red Cat" (Anode-Oxidation, Reduction-Cathode).
Electrolyte Solutions and Ion Transfer
The electrolyte in each half-cell supplies the ions involved in the half-reaction. But if you just connected two beakers with a wire, charge would build up quickly and the reaction would stop. That's where the salt bridge (or porous membrane) comes in.
- The salt bridge allows ions to migrate between the two half-cells without letting the solutions mix directly.
- It maintains electrical neutrality: as the anode solution gains cations (e.g., dissolving in), anions from the salt bridge flow in to balance the charge. Meanwhile, cations from the salt bridge flow into the cathode solution to replace the being deposited out.
- Common salt bridge electrolytes are or , chosen because and (or ) have similar ionic mobilities and don't participate in the cell reactions.
External Circuit and Current Flow
The external circuit is the wire (plus any load) connecting the two electrodes.
- Electrons flow through the external circuit from anode to cathode.
- By convention, conventional current flows in the opposite direction (cathode to anode in the external circuit), but for electrochemistry you mostly track electron flow.
- The magnitude of the current depends on factors like electrode surface area, electrolyte concentration, and the internal resistance of the cell.
Electrode Potential and Electron Flow
Defining Electrode Potential
Every electrode material, when placed in contact with a solution of its ions, establishes an equilibrium between the metal and its dissolved ions. The electrode potential quantifies the tendency of that half-reaction to proceed as a reduction.
- Electrode potentials cannot be measured in isolation. You always need a reference, and the universal reference is the standard hydrogen electrode (SHE), defined as exactly .
- The SHE consists of a platinum electrode in contact with gas at 1 atm, immersed in a solution with , at 25°C.
- Standard reduction potentials () are measured under standard conditions: 25°C, all solute concentrations at 1 M, and all gases at 1 atm.
Some representative values:
| Half-Reaction | (V) |
|---|---|
A more positive means a stronger tendency to be reduced. A more negative means the species is more easily oxidized.
Determining the Direction of Electron Flow
When two half-cells are connected, the half-cell with the more negative reduction potential acts as the anode (it's easier to oxidize), and the half-cell with the more positive reduction potential acts as the cathode.
- Electrons flow spontaneously from the more negative electrode to the more positive electrode through the external wire.
- In the Daniell cell: zinc () is oxidized, copper () is reduced, and electrons travel from Zn to Cu.
This isn't something you choose arbitrarily. The relative reduction potentials determine which electrode is the anode and which is the cathode.

Cell Potential Calculation
Calculating Standard Cell Potential
The standard cell potential tells you the voltage a cell produces when all species are at standard-state conditions.
Steps to calculate :
- Write the two half-reactions as reductions (the way they appear in standard tables).
- Identify which half-reaction has the more negative . That one will be reversed to become the oxidation (anode).
- Apply the formula:
Note that you use the tabulated reduction potentials directly in this formula. You do not flip the sign of the anode potential before subtracting; the subtraction handles it.
- A positive means the reaction is spontaneous under standard conditions. A negative value means it's non-spontaneous.
Example: Daniell cell (Zn–Cu)
- Cathode: ,
- Anode: ,
The positive value confirms the reaction is spontaneous: zinc dissolves and copper plates out.
Common mistake: Students sometimes double the reduction potential when balancing electrons (e.g., multiplying the Cu half-reaction by 2). Don't do this. Electrode potentials are intensive properties, meaning they don't change when you multiply the half-reaction by a stoichiometric coefficient.
Gibbs Free Energy and Cell Potential
The Connecting Equation
The link between electrochemistry and thermodynamics comes through this relationship:
where:
- is the Gibbs free energy change (in joules)
- is the number of moles of electrons transferred in the balanced cell reaction
- is Faraday's constant,
- is the cell potential (in volts)
Under standard conditions, this becomes .
Relationship Between Cell Potential and Spontaneity
The sign logic here is clean because of the negative sign in the equation:
- : spontaneous (galvanic cell)
- : non-spontaneous (requires external energy, i.e., electrolytic cell)
- : system at equilibrium
Example calculation for the Daniell cell:
The balanced reaction transfers moles of electrons.
The large negative confirms this reaction is strongly spontaneous, which is consistent with the positive cell potential.
Key connection: This equation is what makes electrochemistry so powerful thermodynamically. A simple voltage measurement gives you direct access to , which is otherwise difficult to measure for many reactions. This is also why electrochemical measurements are used to determine equilibrium constants, since .