X-rays, electrons, and neutrons offer unique advantages for diffraction studies. Each radiation type interacts differently with matter, providing complementary information about atomic structure, composition, and properties of materials.
Understanding the strengths and limitations of these techniques is crucial for selecting the most appropriate method. Factors like penetration depth, resolution, elemental sensitivity, and sample requirements play key roles in experimental design and data interpretation.
Scattering and Penetration
Scattering Cross-Sections and Interaction Mechanisms
- X-rays scatter from electron clouds surrounding atoms
- Electrons interact with both electron clouds and atomic nuclei through electrostatic forces
- Neutrons scatter primarily from atomic nuclei via nuclear forces
- Neutron scattering cross-sections vary irregularly across the periodic table
- X-ray scattering cross-sections increase steadily with atomic number
- Electron scattering cross-sections are generally larger than X-rays or neutrons
- Magnetic neutron scattering occurs due to interaction with unpaired electron spins
Penetration Depth and Sample Thickness
- X-rays penetrate materials to depths of micrometers to millimeters
- Electrons have limited penetration, typically nanometers to micrometers
- Neutrons can penetrate several centimeters in many materials
- X-ray penetration depth decreases with increasing atomic number of the sample
- Electron penetration depth affected by accelerating voltage and sample density
- Neutron penetration allows for bulk sample analysis and in-situ experiments
- Sample thickness must be optimized for each technique to balance signal and absorption
Resolution and Wavelength Considerations
- X-ray wavelengths typically range from 0.5 to 2.5 Å
- Electron wavelengths are much shorter, around 0.02 to 0.05 Å at common accelerating voltages
- Thermal neutron wavelengths are comparable to X-rays, around 1 to 2 Å
- Electron diffraction achieves highest spatial resolution due to short wavelengths
- X-ray diffraction provides good balance of resolution and penetration
- Neutron diffraction offers lower resolution but unique scattering properties
- Wavelength affects the accessible range of reciprocal space in diffraction experiments
Sample and Sensitivity
Sample Preparation and Environmental Considerations
- X-ray samples can be single crystals, powders, or thin films
- Electron diffraction requires very thin samples, often prepared by ion milling or focused ion beam
- Neutron diffraction typically uses larger bulk samples due to weaker scattering
- X-ray experiments often conducted in air or controlled atmospheres
- Electron diffraction performed in vacuum to prevent electron scattering by gas molecules
- Neutron experiments allow for complex sample environments (cryostats, furnaces, pressure cells)
- Sample size requirements: X-rays (μm to mm), electrons (nm to μm), neutrons (mm to cm)
Elemental Sensitivity and Contrast
- X-rays sensitive to elements with higher atomic numbers, struggle with light elements
- Electrons provide good contrast for most elements but are affected by multiple scattering
- Neutrons offer unique sensitivity to light elements, especially hydrogen and its isotopes
- X-ray scattering factors increase monotonically with atomic number
- Neutron scattering lengths vary non-monotonically, allowing differentiation of nearby elements
- Isotope sensitivity of neutrons enables contrast variation experiments
- Electron diffraction sensitive to valence electron distribution, useful for bonding studies
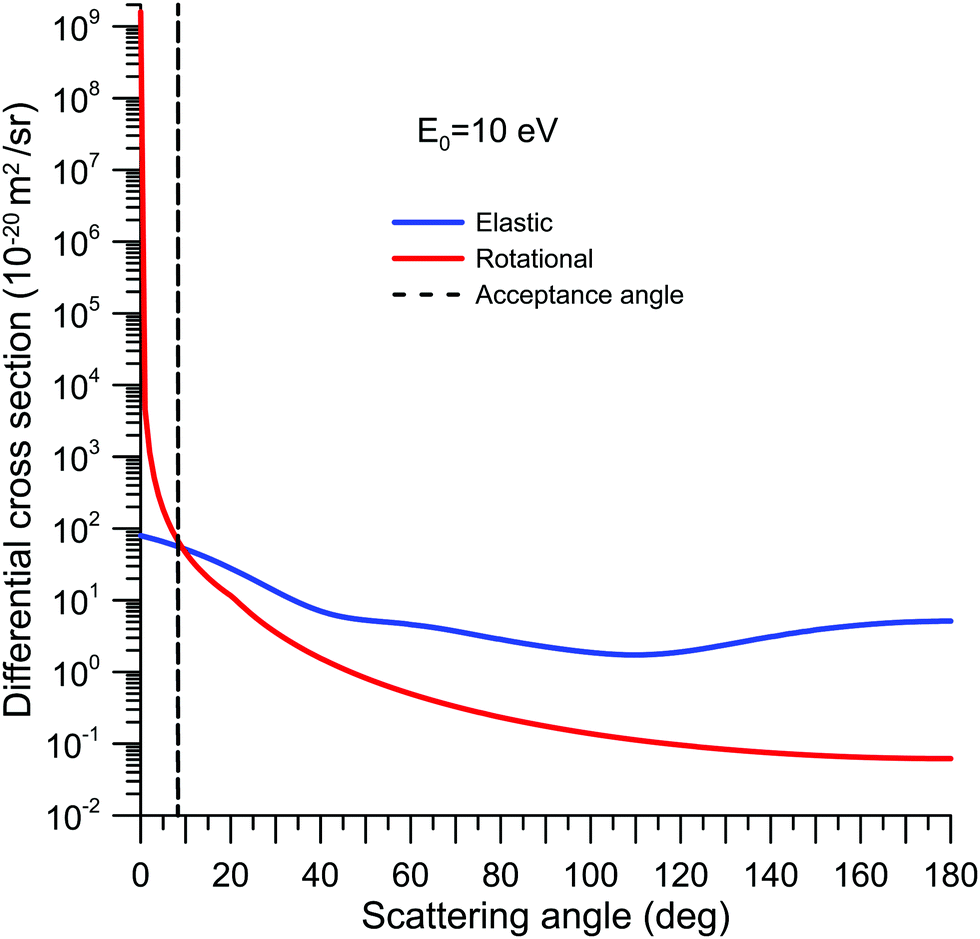
Radiation Damage and Non-Destructive Analysis
- X-rays can cause radiation damage in sensitive materials (proteins, organic compounds)
- Electrons frequently cause sample damage, limiting exposure times
- Neutrons generally non-destructive, allowing for repeated measurements or long exposures
- X-ray damage mechanisms include radiolysis and heating
- Electron beam damage includes knock-on damage, radiolysis, and charging effects
- Neutron activation may occur, requiring cooling periods for some materials
- Cryogenic techniques often used to mitigate radiation damage in biological samples
Advanced Analysis Techniques
Magnetic Structure Determination
- Neutron diffraction uniquely suited for determining magnetic structures
- Neutrons interact with unpaired electron spins via dipole-dipole coupling
- X-rays can probe magnetic structures through resonant scattering techniques
- Electron diffraction limited in magnetic structure analysis due to strong electrostatic interactions
- Neutron polarization analysis separates nuclear and magnetic scattering contributions
- Magnetic form factors differ between X-ray and neutron techniques
- Combined X-ray and neutron studies provide comprehensive magnetic structure information
Complementary Techniques and Multi-Modal Analysis
- X-ray absorption spectroscopy (XAS) probes local electronic structure
- Electron energy loss spectroscopy (EELS) provides elemental and chemical state information
- Inelastic neutron scattering measures phonon and magnon dispersions
- Pair distribution function (PDF) analysis applicable to all three radiation types
- Small-angle scattering techniques (SAXS, SANS) investigate nanoscale structures
- Tomography methods developed for 3D imaging with X-rays, electrons, and neutrons
- Combining techniques yields comprehensive structural and dynamical information
- X-ray/neutron diffraction for average structure
- Electron microscopy for local structure and defects
- Spectroscopic methods for electronic properties
Time-Resolved and In-Situ Experiments
- Synchrotron X-ray sources enable ultra-fast time-resolved studies (femtosecond to nanosecond)
- Pulsed neutron sources allow for microsecond to millisecond time resolution
- Dynamic transmission electron microscopy (DTEM) achieves nanosecond time resolution
- In-situ X-ray diffraction used for studying phase transitions and chemical reactions
- Environmental transmission electron microscopy (ETEM) observes samples under gas or liquid
- Neutron diffraction suitable for in-situ studies under extreme conditions (high pressure, temperature)
- Time-resolved techniques reveal structural dynamics and intermediate states in various processes
- Catalytic reactions
- Phase transformations
- Protein folding