We're back to solutions! Remember that solutions are homogeneous mixtures where the particles are evenly mixed and the solute is uniformly distributed within the solvent. The solute is the substance that is dissolved, while the solvent is the substance that does the dissolving.
We discussed how to calculate the concentration of a solute dissolved in a solvent, but what does this value depend on? Let's go over solubility!
What is Solubility?
To put it simply, solubility is the ability of a substance to dissolve in a solvent to form a homogeneous mixture. A substance that is soluble in a solvent will dissolve completely to form a solution, while a substance that is insoluble in a solvent will not dissolve and will remain in a separate phase.
The solubility of one substance in another depends on:
- The tendency of systems to become more random (by becoming more dispersed in space)
- The relative intermolecular solute-solute energies compared with solute-solvent interactions.
Polar and ionic solutes tend to dissolve in polar solvents, and non-polar solutes tend to dissolve in non-polar solvents (remember “like dissolves like”). This all goes back to intermolecular forces: substances with similar intermolecular forces tend to be miscible or soluble in one another!
👉 Make sure to review intermolecular forces as they will constantly come back in this course!
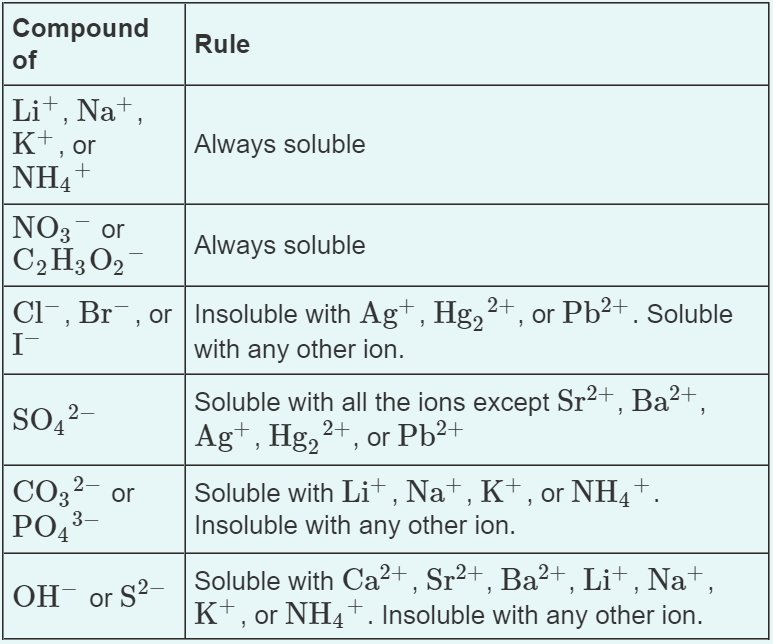
There are a few solubility rules that will be helpful on the AP Exam and we will return back to these when we discuss precipitation reactions in unit four:

You can easily notice that many of the same ions (notably Ag, Hg2 2+, and Pb2+) are often exceptions to these solubility rules. However, do not try to memorize these completely! These, like things like polyatomic ions, are memorized implicitly through usage! That's why by the end of AP Chemistry, most students don't think about charts like these, they simply know them through doing a bajillion problems.**
Saturation of Solutions
Every solution, no matter the solute and solvent, has something called a saturation point. Essentially, this is the point at which no more solute can be dissolved in the solvent.
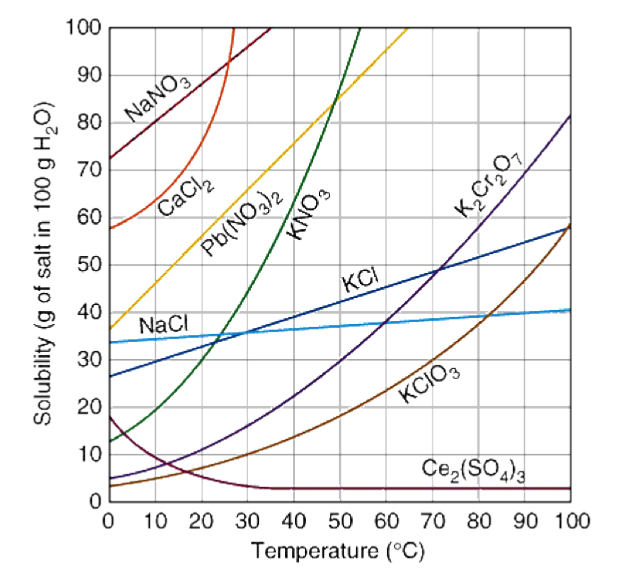
It depends on three things: the temperature, the solvent, and the solute. At a higher temperature, more solute can be dissolved (for the majority of solutes). This can be seen in the following solubility curve:
Image Courtesy of Dynamic ScienceSolubility curves are graphs that show the relationship between the solubility of a substance in a solvent and the temperature of that solvent. These curves are especially helpful when predicting the solubility of a substance at different temperatures.
When learning about a new type of graph, you always want to note the axes:
- The x-axis is the temperature of the solvent in degrees Celcius.
- The y-axis is the mass of the substance that is dissolved in a given amount of solvent.
Take a look at the dark blue curve that represents the solubility of potassium chloride (KCl). At approximately 70 degrees Celcius, there will be almost 50 grams of KCl dissolved in the solution. This value specifically forms a saturated solution.
Practice reading these graphs and ensuring you understand what information can be obtained from them, as well as what relationship is shown.
Saturated Solutions
A saturated solution is a solution in which the maximum amount of solute has been dissolved in a solvent at a specific temperature. You can also think about solubility as the amount of solute needed to form a saturated solution at any particular temperature.
Once you have reached the saturation point, any additional solute (at that temperature) will fall out of the solution and not dissolve. Essentially, with saturated solutions, you are on the solubility curve.
Undersaturated Solutions
Undersaturated solutions are what their name implies - a solution that has not yet reached saturation.
With an undersaturated solution, you can add more solute and it will continue to dissolve (with the temperature remaining constant). Like saturated solutions, an undersaturated solution can be modeled using a solubility curve, representing a point below the solubility curve.
Supersaturated Solutions
Supersaturated solutions are a type of solution where you have more solute than you can dissolve in the solvent. You have basically surpassed the saturation point.
In order to accomplish this, a chemist heats up the solution and then adds enough solute to saturate it at that temperature. The chemist would then slowly cool the solution down to the temperature they desire. This creates a supersaturated solution that when agitated will produce crystals. These crystals are extra solutes coming out of the solution, allowing the supersaturated solution to return to its saturated state.
With this being said, you can identify when a solution will be supersaturated by looking above the solubility curve on a graph.
A supersaturated solution of CH3COONa, GIF Courtesy of GyfcatFactors Affecting Solubility
The solubility of a substance is influenced by several factors, including the temperature of the solvent, the concentration of the solvent, and the presence of other substances.
Polarity
As mentioned before, the polarity of both the solvent and solutes has a huge effect on solubility. This concept can always be thought of as "like dissolves like." Nonpolar substances are more likely to dissolve in nonpolar solvents and polar substances are more likely to dissolve in polar solvents.
Pressure
Pressure only affects the solubility of gases. At higher pressures, more gas can be dissolved in the solvent, while at lower pressures, less gas will dissolve. In other words, as pressure increases, solubility increases as well. This phenomenon explains why soda🥤 becomes flat over time.
This can also be seen using Henry's Law: C = kP, where...
- C stands for the concentration of the dissolved gas in molarity
- k stands for Henry's law constant
- P stands for the partial pressure of the gas above the solution
This equation is just an extra piece of information that your teacher may speak about, and it is not necessary for the AP.
For solids and liquids, pressure does not affect solubility. This is very important and you will have to start paying attention to the phase at which the compounds you are discussing are in.
Temperature
As mentioned before when taking a look at solubility curves, as temperature increases, the solubility of solids and liquids increases as well. This is due to the increased kinetic energy of the solvent molecules at higher temperatures, allowing them to more effectively dissolve the solute. Remember, temperature can be thought of as the average kinetic energy of particles.
This trend is different for gases: as temperature increases, the solubility of the gas decreases.
👉This will be discussed in greater depth when we go over unit seven later in this course.
Concentration of the Solvent
The solubility of a substance generally increases as the concentration of the solvent increases. This is due to the increased number of solvent molecules present, which allows for more solute to dissolve.
Presence of Other Substances
The solubility of a substance can be influenced by the presence of other substances, such as electrolytes or solutes. The presence of these substances can affect the solubility of the solute by altering the chemical nature of the solvent or by competing for the same binding sites on the solvent molecules.
Surface Area
The solubility of a substance is generally influenced by the surface area of the solute. A smaller solute particle size will generally increase the solubility of the substance, as it allows for a greater surface area to be exposed to the solvent.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
aqueous solvents | Solvents in which water is the dissolving medium, commonly used in chemistry due to water's polar nature. |
intermolecular interactions | Forces between molecules, such as hydrogen bonding, dipole-dipole forces, and London dispersion forces, that affect the physical and chemical properties of substances. |
ionic compound | Compounds formed by the electrostatic attraction between positively charged cations and negatively charged anions. |
miscible | Capable of being mixed in all proportions without separating into distinct phases. |
molecular compound | Compounds composed of molecules held together by covalent bonds, typically formed between nonmetals. |
nonaqueous solvents | Solvents that do not use water as the dissolving medium, such as organic solvents or liquid ammonia. |
solubility | The maximum amount of a solute that can dissolve in a given amount of solvent at a specific temperature, typically expressed in moles per liter (molarity) or grams per 100 mL of solvent. |
Frequently Asked Questions
What is solubility and how does it actually work?
Solubility is how much of a substance (solute) will dissolve in a solvent at a given temperature. At the particle level, dissolving means solvent molecules overcome the solute’s cohesive forces (like lattice energy in ionic solids or intermolecular forces in molecular compounds) and surround separate particles—solvation (hydration in water). Key idea: “like dissolves like”—polar solvents (high dielectric constant, can H-bond or do ion–dipole interactions) dissolve polar or ionic solutes; nonpolar solvents dissolve nonpolar solutes via London dispersion forces. For ionic salts, solubility depends on the balance between lattice energy (holds ions together) and hydration energy (stabilizes ions in solution); stronger ion–dipole/hydration favors solubility. Other AP-relevant factors: temperature (solubility often changes), common-ion effect and Ksp (solubility product) control equilibrium concentrations, and solvent type (polar protic vs aprotic) affects reactions and solubility. AP exam tasks often ask you to explain these relationships qualitatively and calculate Ksp-based solubilities. For a focused review, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why do some things dissolve in water but others don't?
Short answer: substances dissolve when the new interactions between solute and solvent are comparable to or stronger than the original interactions that hold the solute together—“like dissolves like.” Ionic salts (NaCl) are held together by ionic lattice energy; in water they dissolve if hydration (ion–dipole) energy from water compensates for breaking the lattice. Polar covalent molecules dissolve in polar solvents via dipole–dipole and hydrogen-bonding interactions. Nonpolar molecules dissolve in nonpolar solvents because London dispersion forces match up. Solubility also depends on dielectric constant (water’s high value stabilizes ions), temperature, and equilibria (Ksp, common-ion effect)—all things you’ll see on the AP (Topic 3.10, LO 3.10.A). For a focused review check the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and practice problems for Unit 3 (https://library.fiveable.me/ap-chemistry/unit-3). For lots more practice Qs see Fiveable’s practice page (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between soluble and insoluble compounds?
Soluble means a substance dissolves to form a (homogeneous) solution in a given solvent; insoluble means it doesn’t dissolve appreciably and stays as a separate phase. For ionic solids this comes down to lattice energy vs. solvation (hydration) energy: if hydration (ion–dipole) interactions with water overcome the lattice energy, the salt is soluble. For molecular compounds “like dissolves like”: polar molecules (dipole–dipole, H-bonding) dissolve in polar solvents; nonpolar molecules dissolve in nonpolar solvents via London dispersion. Other AP-relevant points: solvent dielectric constant affects ion separation, temperature and the common-ion effect change solubility, and for sparingly soluble salts you use Ksp to quantify [ions]. This is exactly Topic 3.10 in the CED. For a quick review, check the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and try practice problems (https://library.fiveable.me/practice/ap-chemistry) to solidify the ideas.
Can someone explain "like dissolves like" in simple terms because I'm totally confused?
Think of "like dissolves like" as matching kinds of stickiness: solvents dissolve solutes that interact with them strongly. If both have similar intermolecular forces they mix; if not, they don’t. - Polar solvents (water, dielectric constant ≈80) have permanent dipoles, hydrogen bonding, and make strong ion–dipole or dipole–dipole interactions. That’s why ionic salts (NaCl) and polar molecules usually dissolve well in water—hydration energy overcomes lattice energy. - Nonpolar solvents (hexane) have only London dispersion forces, so they dissolve nonpolar molecules (oils, hydrocarbons) because dispersion–dispersion interactions are compatible. - Polar aprotic vs polar protic matters for ions/complexation (e.g., solubility of salts with polar aprotic solvents). - If attraction between solute and solvent < solute–solute + solvent–solvent attractions, the solute won’t dissolve (think energy balance: lattice vs hydration/solvation). This idea is exactly what AP LO 3.10.A tests—focus on identifying intermolecular interactions (ion–dipole, H-bond, dipole–dipole, dispersion) when predicting solubility. For extra practice, check the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do intermolecular forces affect whether something will dissolve or not?
Solubility comes down to how well the solute’s intermolecular forces (IMFs) can be replaced by solute–solvent interactions. “Like dissolves like”: polar solutes (ionics, polar molecules) dissolve in polar solvents because strong ion-dipole, dipole-dipole, or H-bonding with the solvent compensate for breaking solute IMFs (and lattice energy for ionic solids). Nonpolar solutes rely on London dispersion forces and dissolve best in nonpolar solvents. For ionic compounds in water, compare lattice energy (energy to separate ions) vs. hydration energy (ion-dipole stabilization); if hydration > lattice, it’s soluble. Solubility also depends on solvent properties (dielectric constant, protic vs aprotic), temperature, and common-ion effects (Ksp constraints on ionic solubility). On the AP exam you should be able to explain these using terms like ion-dipole, hydration, lattice energy, Ksp, and give reasoning for miscibility/insolubility. For a focused review, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does salt dissolve in water but oil doesn't?
Salt (NaCl) dissolves in water because NaCl is ionic and water is polar. Water molecules have strong dipoles that orient around Na+ and Cl– and form ion–dipole interactions; the hydration energy gained when water solvates the ions overcomes the lattice energy holding the solid ionic crystal together, so the ions separate and dissolve. Oil is nonpolar (mostly long hydrocarbon chains) and interacts by weak London dispersion forces. Water’s hydrogen-bonding and dipole–dipole interactions don’t stabilize nonpolar oil molecules, so oil and water are immiscible—“like dissolves like” (polar solvents dissolve polar/ionic solutes; nonpolar solvents dissolve nonpolar solutes). Dielectric constant and solvent polarity matter: water’s high dielectric constant also reduces electrostatic attractions between ions, helping dissolution. This topic is in the AP CED under solubility and ion–dipole/hydration concepts (Topic 3.10). For a focused review, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What does miscible mean and how is it different from soluble?
Miscible means two liquids mix in all proportions to form a single homogeneous phase (e.g., ethanol and water). Soluble is broader: a substance (solid, liquid, or gas) that dissolves in a solvent up to some limit (e.g., NaCl is soluble in water; O2 is slightly soluble). Key difference: miscible implies complete mixing with no limit for liquid–liquid pairs; soluble can be limited (a solubility value) and applies to more types of solutes/solvents. AP connection: use intermolecular interactions to predict miscibility/solubility—“like dissolves like.” Strong ion–dipole or hydrogen-bonding favors solubility in water; similar dipole/polarity between two liquids favors miscibility. For exam questions, link qualitative predictions to forces (ion-dipole, dipole-dipole, H-bonding, London dispersion) and mention dielectric constant when comparing aqueous vs nonaqueous solvents. Review Topic 3.10 on Fiveable (study guide: https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and practice related problems at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why polar substances dissolve in polar solvents - can someone break this down?
“Like dissolves like” means substances with similar intermolecular interactions mix because the solvent can replace the interactions that hold the solute together. For polar solutes (or ions) that have dipoles or charges, polar solvents (like water) have permanent dipoles and can form strong dipole–dipole, hydrogen-bonding, or ion–dipole interactions with the solute. Those solvent–solute interactions release solvation (or hydration) energy that compensates for breaking solute–solute attractions (and, for ionic solids, the lattice energy). If the net energy change is favorable and entropy increases enough, the solute dissolves. On the AP Chem exam this is Topic 3.10 (LO 3.10.A): use terms like dipole–dipole, hydrogen bonding, ion–dipole, London dispersion, lattice energy, and solvation/hydration energy in your explanations. For a quick refresher, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I predict if an ionic compound will be soluble in water?
Predict solubility by comparing two energies: lattice energy (how strongly ions stick in the solid) vs hydration (ion–dipole) energy from water. If hydration energy ≥ lattice energy, the ionic solid dissolves; if lattice energy is much larger, it stays insoluble. Use these practical rules on the AP: 1) memorized solubility rules (e.g., all nitrates, alkali salts, and NH4+ are soluble; most Ag+, Pb2+, and Pb2+/Hg2+/Ba2+ salts often insoluble), 2) consider ion charge/size (high charge and small radius → large lattice energy → lower solubility), and 3) solvent polarity and dielectric constant (water ε ≈ 80 stabilizes ions via strong ion–dipole interactions; nonpolar solvents don’t). Also remember common-ion effect and Ksp: adding a common ion lowers solubility (important for equilibrium problems on the exam). For a compact AP-aligned review and examples, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between molecular polarity and solubility?
Polarity guides solubility by “like dissolves like.” Polar solvents (water) stabilize polar molecules and ions via dipole–dipole, hydrogen bonding, or ion–dipole interactions; nonpolar solvents stabilize nonpolar molecules via London dispersion forces. So ionic salts (NaCl) dissolve well in water because strong ion–dipole hydration overcomes lattice energy, while they’re insoluble in hexane. Small polar molecules with H-bond donors/acceptors (ethanol) mix with water; large nonpolar areas reduce water solubility. Polar protic solvents (water, alcohols) can H-bond; polar aprotic solvents (acetone, DMSO) solvate cations differently and affect solubility/reactivity. Dielectric constant also matters: higher dielectric constant lowers electrostatic attractions, helping ions separate. This fits AP LO 3.10.A (relate intermolecular interactions to solubility). For a concise review and examples, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and more practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do nonpolar substances dissolve better in nonpolar solvents?
Nonpolar substances dissolve better in nonpolar solvents because “like dissolves like”—substances with similar intermolecular interactions mix. Nonpolar molecules interact mainly through London dispersion forces (instantaneous induced dipoles). When a nonpolar solute enters a nonpolar solvent, you replace solute–solute and solvent–solvent dispersion interactions with similarly sized solute–solvent dispersion interactions, so the enthalpy change isn’t strongly unfavorable. Polar solvents (high dielectric constant, with dipole–dipole and H-bonding) can’t form those favorable interactions with nonpolar solute molecules, and breaking strong solvent–solvent H-bonds or ion-dipole networks costs energy, so nonpolar solutes remain insoluble. Entropy also usually increases as solute mixes into a like solvent, helping solubility. This is exactly what AP CED LO 3.10.A expects you to explain (use terms like London dispersion, dipole–dipole, hydrogen bonding, dielectric constant). For a quick review, see the Topic 3.10 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do hydrogen bonds affect solubility in water?
Hydrogen bonding strongly increases a molecule’s solubility in water because water can form H-bonds (donor or acceptor) with solute molecules—think alcohols, amines, and sugars. In AP terms: “like dissolves like”—substances with similar intermolecular interactions (here hydrogen bonding and dipole interactions) are more miscible (CED 3.10.A.1). H-bond capable solutes get stabilized by hydration (ion-dipole or H-bonding), lowering free energy and favoring dissolution. By contrast, a nonpolar solvent can’t H-bond, so H-bonding solutes are usually insoluble there (polar protic vs. nonpolar/aprotic behavior). Note: strong H-bonding within a solute (intramolecular H-bond) can actually reduce water solubility because those sites aren’t available to water. For more AP-aligned examples, practice problems, and a quick study guide on Topic 3.10, check Fiveable’s solubility study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and their AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
What happens at the molecular level when something dissolves?
When something dissolves, three molecular steps happen: (1) you break solute–solute attractions (like ionic lattice energy or molecular intermolecular forces), (2) you break some solvent–solvent interactions, and (3) you form new solute–solvent interactions (solvation or hydration). Whether dissolution is favorable depends on the balance of those energies—e.g., hydration (ion–dipole) energy must overcome lattice energy for ionic salts to dissolve. “Like dissolves like” summarizes this: polar solvents (high dielectric, can H-bond or have strong dipole) stabilize ions and polar molecules via ion–dipole, dipole–dipole, or H-bonding; nonpolar solvents stabilize nonpolar solutes via London dispersion. Electrolytes dissociate into ions and conduct; solubility can be reduced by the common-ion effect or limited by Ksp. On the AP exam, you should use these terms (lattice energy, ion-dipole, hydration, dielectric constant, solvation) to explain why a substance is soluble or not. For extra practice and examples, check the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1), the Unit 3 overview (https://library.fiveable.me/ap-chemistry/unit-3), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about solubility rules - do I need to memorize them all for the AP exam?
Short answer: you don’t need to blindly memorize every weird exception, but you should know the common solubility “rules” and, more importantly, the underlying reasons (lattice energy vs. hydration energy, ion–dipole interactions, dielectric constant). AP graders expect you to explain solubility using CED concepts like ion–dipole interactions, hydration energy, common-ion effect, and Ksp (Topic 3.10.A). Practical checklist to memorize: - Always soluble: nitrates (NO3−), alkali metal salts, ammonium (NH4+). - Usually soluble: acetates, most chlorides/bromides/iodides (except Ag+, Pb2+, Hg2 2+), most sulfates (except Ba2+, Pb2+, Ca2+, Sr2+). - Usually insoluble: carbonates, phosphates, sulfides, hydroxides (except strong-base cations). Study smart: practice applying ideas (lattice vs. hydration, polar vs. nonpolar solvents, common-ion) to justify answers rather than rote lists. For targeted review, use the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and drill problems from the AP practice bank (https://library.fiveable.me/practice/ap-chemistry). Unit 3 is 18–22% of the exam, so solid practice beats pure memorization.
Why can't we just mix any two liquids together and have them dissolve?
You can’t just mix any two liquids and have them dissolve because solubility/miscibility depends on the intermolecular interactions between the liquids—“like dissolves like.” Polar solvents (water) stabilize polar solutes via dipole–dipole, hydrogen bonding, or ion–dipole (for ions) interactions and have high dielectric constants; nonpolar solvents (hexane) rely on London dispersion forces. If the solvent can’t compensate for the solute’s self-interactions (or an ionic solid’s lattice energy), the mixture won’t dissolve or be miscible. Example: oil (nonpolar) and water (polar) separate; ethanol (small polar, H-bonding) mixes with water. For ionic compounds, compare lattice energy vs hydration (solvation) energy to predict solubility. This is exactly Topic 3.10 in Unit 3 of the CED (keywords: ion-dipole, hydrogen bonding, dielectric constant). For a focused review, see the Topic 3.10 study guide (https://library.fiveable.me/ap-chemistry/unit-3/solubility/study-guide/XmVywOyhrzQscHR0OYP1) and practice problems at (https://library.fiveable.me/practice/ap-chemistry).