Chemical bonding is what holds atoms together in molecules. Valence bond theory and molecular orbital theory are two complementary frameworks for understanding how electrons interact to form those bonds. Each theory has distinct strengths: valence bond theory gives you an intuitive, localized picture of bonding, while molecular orbital theory provides a more complete description of electronic structure across entire molecules.
Valence Bond Theory
Fundamental Principles and Concepts
Valence bond theory (VBT) explains bonding through the overlap of atomic orbitals on adjacent atoms. When two half-filled orbitals overlap, the electrons pair up and become shared between the two nuclei, forming a localized bond.
Two types of bonds arise from orbital overlap:
- Sigma (σ) bonds form from head-on overlap along the internuclear axis. These are the strongest type of covalent bond.
- Pi (π) bonds form from side-by-side overlap of p orbitals above and below the internuclear axis. They're weaker than σ bonds and only occur alongside an existing σ bond.
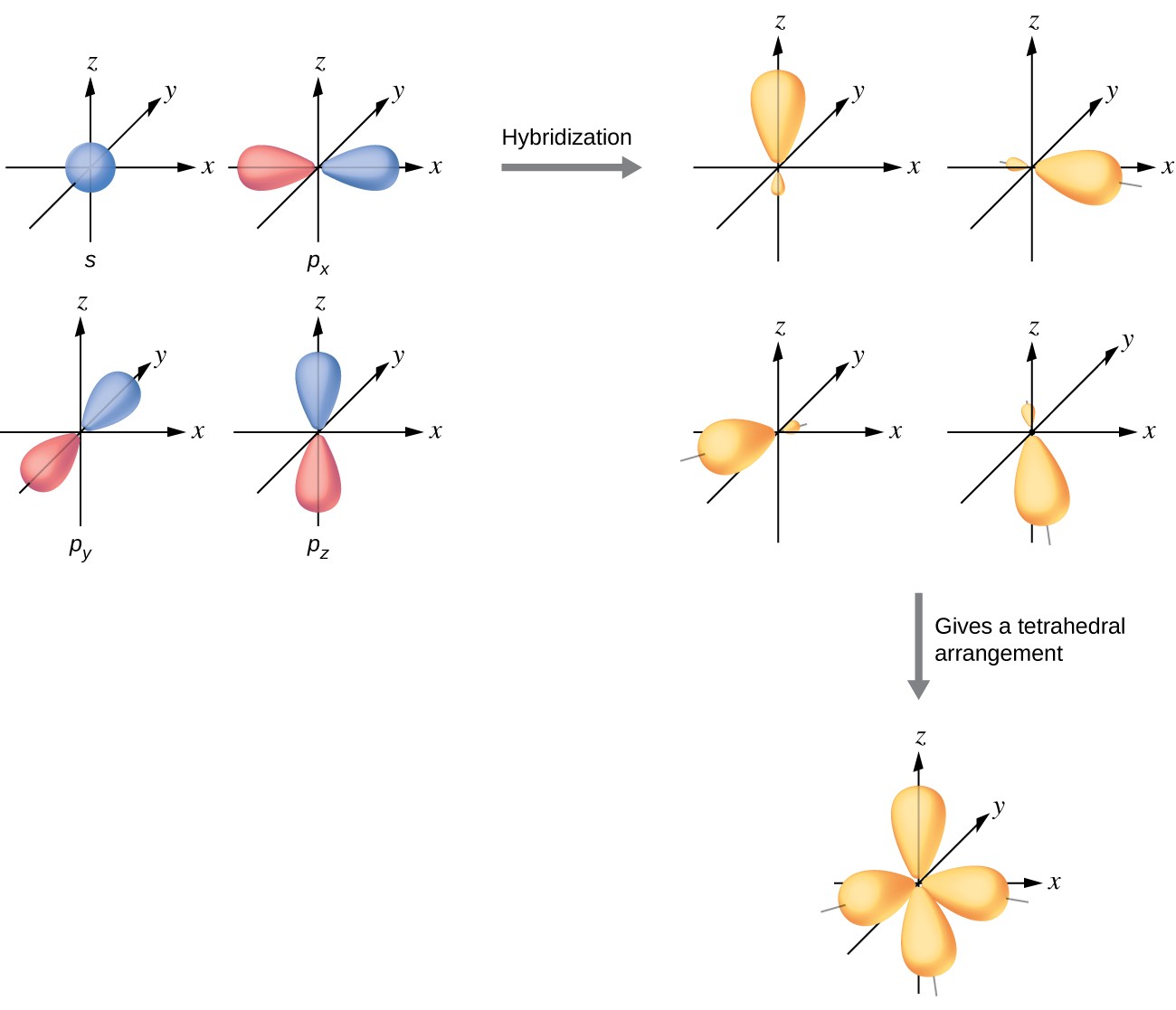
Hybridization is a central concept in VBT. Atomic orbitals on the same atom mix together to produce new hybrid orbitals with specific geometries suited for bonding. The type of hybridization determines molecular shape:
- sp → 2 hybrid orbitals → linear geometry (180°), as in
- **sp}^2\text{BF}_3$$
- **sp}^3\text{CH}_4$$
Applications and Limitations
VBT is straightforward to apply for predicting geometry and bonding in simple molecules. For example, in ethene (), each carbon is hybridized: three orbitals form σ bonds, and the remaining unhybridized p orbital on each carbon overlaps sideways to form the π bond of the double bond. In ethyne (), each carbon is hybridized, leaving two p orbitals per carbon to form two π bonds (a triple bond overall).
Where VBT falls short:
- It can't easily explain molecules with unpaired electrons, since it focuses on paired, localized bonds.
- It doesn't directly predict magnetic properties (paramagnetism vs. diamagnetism).
- It handles resonance awkwardly, requiring multiple equivalent structures rather than a single unified picture.
Molecular Orbital Theory

Fundamental Principles and Concepts
Molecular orbital theory (MOT) takes a different approach. Instead of keeping electrons localized between two atoms, it combines atomic orbitals mathematically through linear combination of atomic orbitals (LCAO) to produce molecular orbitals that spread across the entire molecule.
When two atomic orbitals combine, they produce two molecular orbitals:
- A bonding molecular orbital (lower energy), where electron density concentrates between the nuclei, stabilizing the molecule.
- An antibonding molecular orbital (higher energy, marked with an asterisk like or ), where electron density is pushed away from the region between nuclei.
Electrons fill molecular orbitals following the same rules as atomic orbitals:
- Aufbau principle: fill lowest-energy orbitals first.
- Pauli exclusion principle: each orbital holds at most 2 electrons with opposite spins.
- Hund's rule: when orbitals of equal energy are available, electrons fill them singly before pairing.
Bond order quantifies bond strength and is calculated as:
A higher bond order means a stronger, shorter bond. A bond order of zero means no stable bond forms.
Applications and Predictions
MOT is especially powerful for diatomic molecules. You can construct molecular orbital diagrams for species like , , , and and read off their bond order, stability, and magnetic behavior directly.
A classic example: VBT predicts should have all electrons paired (diamagnetic), but experiments show is paramagnetic. MOT correctly predicts this because the MO diagram for places two electrons in degenerate antibonding orbitals, and by Hund's rule, they remain unpaired.
MOT also introduces the concept of frontier molecular orbitals:
- HOMO (highest occupied molecular orbital): the highest-energy orbital that contains electrons.
- LUMO (lowest unoccupied molecular orbital): the lowest-energy empty orbital.
The HOMO-LUMO gap governs chemical reactivity and spectroscopic properties. Molecules absorb light when electrons transition from occupied to unoccupied molecular orbitals, and the energy of that transition corresponds to the HOMO-LUMO energy difference.
Valence Bond vs. Molecular Orbital Theory
Key Differences and Comparisons
| Feature | Valence Bond Theory | Molecular Orbital Theory |
|---|---|---|
| Electron description | Localized between bonded atoms | Delocalized across the molecule |
| Key concept | Hybridization and orbital overlap | LCAO → bonding/antibonding MOs |
| Geometry prediction | Directly through hybridization | Not the primary focus |
| Magnetic properties | Cannot predict | Predicts para-/diamagnetism |
| Resonance | Requires multiple structures | Naturally handled by delocalization |
| Ease of use | More intuitive for simple molecules | Requires more quantum mechanical background |
Strengths and Weaknesses
VBT gives you a clear, visual picture of where bonds are and what shape a molecule takes. It's the go-to for quickly determining molecular geometry and understanding localized σ and π bonds. But it breaks down for molecules where electrons aren't neatly localized between two atoms.
MOT provides a more complete and accurate electronic description. It handles delocalized electrons, unpaired electrons, and magnetic properties naturally. The tradeoff is that it requires more mathematical sophistication and is harder to visualize for large molecules.
In practice, chemists use both theories together. VBT is typically used to describe molecular geometry and localized bonding, while MOT is brought in when you need to explain electronic properties, magnetism, or reactivity involving delocalized electrons.
Electronic Structure of Molecules
Molecular Orbital Analysis
To determine the electronic structure of a molecule using MOT:
- Identify the atomic orbitals available on each atom.
- Combine them to form the molecular orbital energy-level diagram.
- Fill electrons into the MOs from lowest to highest energy, following the aufbau principle, Hund's rule, and the Pauli exclusion principle.
- Calculate the bond order using the formula above.
The bond order tells you about stability and bond length: higher bond order means greater stability and shorter bond length. For instance, has a bond order of 3 (triple bond), making it extremely stable with a short bond length of 1.10 Å.
Photoelectron spectroscopy (PES) provides experimental evidence for MOT. The ionization energies observed in PES spectra correspond to the energies of occupied molecular orbitals, giving direct confirmation of the MO energy-level ordering.
Valence Bond Interpretation
VBT remains valuable for understanding localized bonding patterns. The tetrahedral geometry of follows directly from hybridization: carbon's 2s and three 2p orbitals mix to form four equivalent hybrid orbitals pointing toward the corners of a tetrahedron, each overlapping with a hydrogen 1s orbital.
For molecules like benzene (), VBT uses resonance structures to approximate electron delocalization. The two equivalent Kekulé structures suggest that the real electronic structure is an average of both, with equal bond lengths throughout the ring. This is where VBT reaches its limits and MOT's delocalized picture becomes more natural: MOT describes benzene's π electrons as occupying molecular orbitals that span the entire ring, rather than alternating single and double bonds.