Reaction Rate and Its Significance
Definition and Units of Reaction Rate
The reaction rate measures how fast the concentration of a reactant decreases (or a product increases) over time. For a general reaction where reactant A forms product B, the rate is defined as:
The negative sign accounts for the fact that reactant concentration decreases over time, keeping the rate a positive quantity. Units are typically molarity per second (M/s) or molarity per minute (M/min).
One thing to watch for: the rate of disappearance of a reactant and the rate of appearance of a product aren't always equal. You need to account for stoichiometric coefficients. For a reaction , the relationship is:
Importance of Reaction Rates in Chemical Kinetics
Chemical kinetics is the branch of physical chemistry focused on reaction rates and the factors that control them. Understanding rates matters across many fields:
- Industrial chemistry: Optimizing production processes (e.g., tuning conditions in the Haber process for ammonia synthesis)
- Biochemistry: Enzyme kinetics and metabolic pathways depend on precisely regulated rates
- Materials science: Controlling reaction rates during synthesis determines the properties of the final material
Thermodynamics tells you whether a reaction can happen. Kinetics tells you how fast it actually does.
Deriving Rate Laws
Rate Law Equation and Reaction Orders
A rate law is an equation that connects the reaction rate to the concentrations of the reactants:
- is the rate constant, which depends on temperature but not on concentration
- and are reactant concentrations
- and are the reaction orders with respect to and
The overall reaction order is the sum . A critical point: reaction orders are not determined from the balanced equation. They must be found experimentally. The stoichiometric coefficients and the reaction orders only match for elementary (single-step) reactions.
Experimental Determination of Reaction Orders and Rate Constant
The most common approach is the method of initial rates:
- Run the reaction multiple times, each time measuring the initial rate.
- Between trials, change the concentration of one reactant while holding everything else constant.
- Compare how the rate changes relative to the concentration change.
- If doubling doubles the rate, the reaction is first-order in (). If doubling quadruples the rate, it's second-order (). If the rate doesn't change, it's zero-order ().
- Once all orders are known, plug any trial's data into the rate law to solve for .
For example, suppose you have two trials where doubles while stays the same, and the rate quadruples. Then , because .
Types of Reaction Orders
- Zero-order (): Rate is independent of reactant concentration. The reaction proceeds at a constant rate until the reactant is used up. Example: decomposition of on a hot platinum surface.
- First-order (): Rate is directly proportional to the concentration of one reactant. Double the concentration, double the rate. Example: radioactive decay.
- Second-order (): Rate is proportional to either the square of one reactant's concentration, or the product of two reactants' concentrations. Example: dimerization of .
Factors Influencing Reaction Rates
Concentration and Rate Law
Increasing reactant concentration generally increases the rate because there are more molecules available to collide per unit volume. The rate law quantifies exactly how much the rate changes for a given concentration change, which is why knowing the reaction orders matters.
Temperature and the Arrhenius Equation
Higher temperature means molecules move faster on average. This leads to both more frequent collisions and a greater fraction of collisions with enough energy to overcome the activation energy barrier. Even a modest temperature increase (say, 10 K) can significantly boost the rate.
The Arrhenius equation captures this temperature dependence:
- is the pre-exponential factor (related to collision frequency and molecular orientation)
- is the activation energy (the minimum energy needed for a reactive collision)
- is the gas constant ()
- is the absolute temperature in Kelvin
A useful linearized form for comparing two temperatures is:
This lets you calculate from rate constants measured at two different temperatures, or predict how changes when you heat or cool the system.
Catalysts and Activation Energy
Catalysts speed up reactions without being consumed. They do this by providing an alternative reaction pathway that has a lower activation energy. The thermodynamics of the reaction (, equilibrium constant) don't change; only the rate does.
Two main categories:
- Homogeneous catalysts exist in the same phase as the reactants (e.g., acid catalysis in aqueous solution)
- Heterogeneous catalysts are in a different phase, typically a solid surface where gaseous or liquid reactants adsorb and react (e.g., platinum in a catalytic converter)

Other Factors: Surface Area and Pressure
- Surface area: In heterogeneous reactions, increasing the surface area of a solid reactant exposes more sites where reactions can occur. Grinding a solid into a fine powder dramatically increases the rate compared to using a single chunk.
- Pressure: For gaseous reactions, increasing pressure effectively increases the concentration of gas-phase reactants (by compressing them into a smaller volume), which raises the collision frequency and the rate.
Integrated Rate Laws and Applications
Equations for Zero, First, and Second-Order Reactions
While the differential rate law tells you how rate depends on concentration at a given instant, integrated rate laws tell you how concentration changes over time. These are derived by integrating the differential form.
| Order | Differential Form | Integrated Form | Linear Plot (y vs. x) | Slope |
|---|---|---|---|---|
| Zero | vs. | |||
| First | vs. | |||
| Second | vs. |
To determine the reaction order from experimental data, plot the concentration-time data in each of these three ways. Whichever plot gives a straight line tells you the order.
Half-Life Calculations
The half-life () is the time it takes for the reactant concentration to drop to half its initial value.
- Zero-order: — depends on initial concentration; shorter half-life as the reaction progresses
- First-order: — independent of initial concentration. This is why radioactive decay has a constant half-life regardless of how much material remains.
- Second-order: — depends on initial concentration; each successive half-life is longer than the previous one
Applications of Integrated Rate Laws
Integrated rate laws are the practical tools you use to extract kinetic parameters from experimental data:
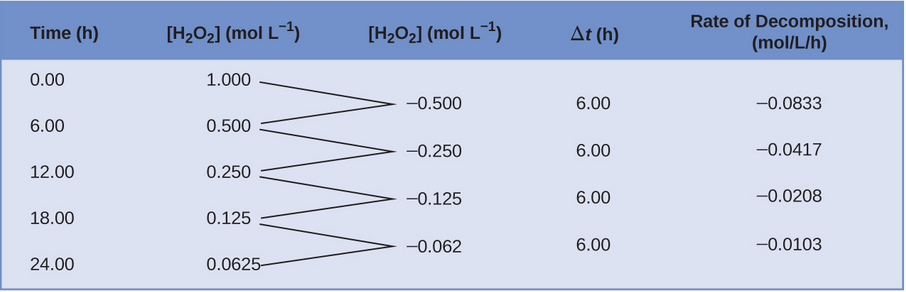
- Decomposition of hydrogen peroxide (): First-order kinetics. Plotting vs. time gives a straight line.
- Hydrolysis of esters: Often pseudo-first-order when water is the solvent (its concentration barely changes), simplifying the analysis.
- Dimerization of cyclopentadiene: Second-order. Plotting vs. time yields a straight line.
By fitting data to these integrated forms, you can determine both the reaction order and the rate constant for a given reaction.