Pressure is a fundamental concept in fluid dynamics, quantifying force per unit area. It governs how fluids behave at rest and in motion, and it shows up in nearly every problem you'll encounter in this course.
Hydrostatic pressure, caused by a fluid's weight, increases linearly with depth. The hydrostatic pressure equation, , is the key tool for calculating pressure at various depths. This concept is vital for designing fluid systems and analyzing submerged structures.
Definition of pressure
Pressure quantifies the force applied perpendicular to a surface per unit area. It's a scalar quantity, which means it has magnitude but no direction. At any given point in a fluid, pressure acts equally in all directions. This is a subtle but important distinction: even though force is a vector, pressure itself is not.
Pressure as force per unit area
The defining equation for pressure is:
where is pressure, is the force acting perpendicular to the surface, and is the area over which that force is distributed. The force can come from a solid object pushing on a fluid, from the weight of a liquid column, or from gas molecules colliding with a container wall.
A quick example: if a 500 N force is spread over 0.25 m², the pressure is . Notice how the same force over a smaller area produces higher pressure. That's why a sharp knife cuts better than a dull one.
Units of pressure measurement
The SI unit for pressure is the pascal (Pa), defined as one newton per square meter:
Other common units include:
- Pounds per square inch (psi): common in U.S. engineering applications
- Atmospheres (atm): convenient for referencing standard atmospheric conditions
- Bar: widely used in meteorology and industrial settings ()
A useful conversion to memorize: .
Hydrostatic pressure
Hydrostatic pressure is the pressure exerted by a fluid at rest due to the weight of the fluid above a given point. Any time you're dealing with a stationary fluid in a tank, pipe, reservoir, or hydraulic device, hydrostatic pressure is at play.
Pressure at a depth in a fluid
In a fluid at rest, the only thing generating pressure at a given depth is the weight of the fluid column sitting above that point. The deeper you go, the more fluid is stacked above you, and the greater the pressure. This increase is perfectly linear for an incompressible fluid with constant density.
Pressure vs depth relationship
The relationship between hydrostatic pressure and depth is:
where:
- = hydrostatic pressure (Pa)
- = fluid density (kg/m³)
- = acceleration due to gravity (9.81 m/s²)
- = depth below the free surface (m)
The linear dependence on means that doubling the depth doubles the pressure. The rate of increase depends on the fluid: water () produces about 9,810 Pa of pressure per meter of depth, while mercury () produces roughly 13.6 times more per meter.
Hydrostatic pressure equation
The equation rests on two key assumptions:
- The fluid is at rest (no flow).
- The fluid density is constant throughout the column (incompressible fluid).
If either assumption breaks down, you'll need a different approach. For gases, where density changes with pressure, the simple linear equation doesn't hold over large height differences. But for liquids under normal conditions, is extremely reliable.
Pressure in different fluid states
The behavior of pressure in a fluid depends on whether that fluid is a liquid or a gas. The core principle that pressure transmits in all directions applies to both, but the details differ significantly.
Pressure in liquids
Liquids are generally treated as incompressible, meaning their density stays constant regardless of the pressure applied (under normal conditions). This is what makes work so cleanly. Pressure in a liquid is transmitted equally in all directions (Pascal's law) and increases linearly with depth.
For example, at 10 m depth in freshwater: . So every 10 meters of water adds roughly one atmosphere of pressure.
Pressure in gases
Gases are compressible, so their density changes as pressure changes. Pressure still transmits equally in all directions, but the relationship between pressure and depth (or height) is no longer linear. Instead, it follows an exponential decay with altitude.
The behavior of an ideal gas is governed by:
where is pressure, is volume, is the number of moles, is the universal gas constant, and is temperature. Because gas density depends on pressure and temperature, you can't simply use for a tall gas column without accounting for how varies.
Incompressible vs compressible fluids
- Incompressible fluids (most liquids): density is essentially constant. Analysis is simpler, and applies directly.
- Compressible fluids (gases): density varies with pressure and temperature. Analysis requires thermodynamic relationships.
In practice, even gases can be treated as incompressible over small height differences where the density change is negligible.
Atmospheric pressure
Atmospheric pressure is the pressure exerted by the weight of the Earth's atmosphere on a surface. It affects everything from weather patterns to how your pressure gauges read.

Definition of atmospheric pressure
Atmospheric pressure at any location equals the weight of the entire air column above that point, divided by the area it acts on. At sea level, this air column extends roughly 100 km upward, though most of the atmosphere's mass is concentrated in the lowest 10-15 km.
Standard atmospheric pressure
Standard atmospheric pressure is the defined reference value at sea level under normal conditions:
This value serves as the baseline for gauge pressure measurements and for comparing pressures across different applications.
Atmospheric pressure vs altitude
Atmospheric pressure decreases with increasing altitude because there's less air above you. The relationship is approximately exponential: pressure drops off more steeply at lower altitudes (where the air is denser) and more gradually at higher altitudes.
A useful benchmark: at about 5,500 m (18,000 ft), atmospheric pressure is roughly half its sea-level value. This is why aircraft cabins are pressurized and why cooking times change at high elevations.
Gauge pressure vs absolute pressure
Pressure measurements come in two flavors depending on the reference point, and mixing them up is a common source of errors in calculations.
Definition of gauge pressure
Gauge pressure is measured relative to the local atmospheric pressure. A gauge pressure of zero means the system is at atmospheric pressure. Most everyday pressure instruments (tire gauges, blood pressure cuffs) read gauge pressure.
Gauge pressure can be positive (above atmospheric) or negative (below atmospheric, sometimes called vacuum pressure).
Definition of absolute pressure
Absolute pressure is measured relative to a perfect vacuum (zero pressure). It's always a positive number. Absolute pressure is what you need for thermodynamic equations like the ideal gas law.
Relationship between gauge and absolute pressure
The conversion is straightforward:
For example, if a tire gauge reads 200 kPa and atmospheric pressure is 101.3 kPa, the absolute pressure inside the tire is .
When solving problems, always check whether the given pressure is gauge or absolute. If a problem says "the pressure is 150 kPa" without specifying, look for context clues or ask.
Pascal's law
Pascal's law is one of the foundational principles of fluid statics and the basis for all hydraulic machinery.
Statement of Pascal's law
Pascal's law states: a change in pressure applied to an enclosed fluid is transmitted undiminished to every portion of the fluid and to the walls of its container. The pressure change acts equally in all directions, regardless of the container's shape or size.
Applications of Pascal's law
Pascal's law is what makes hydraulic systems so powerful. A small force applied over a small area creates a pressure that, when transmitted to a larger area, produces a much larger force.
The relationship is:
So if you apply 100 N to a piston with area 0.01 m², the pressure is 10,000 Pa. That same pressure acting on a second piston with area 0.1 m² produces a force of 1,000 N. You've multiplied the force by a factor of 10.
Hydraulic systems and pressure transmission
Hydraulic systems use a fluid (typically oil) to transmit pressure from an input to an output. The basic components are:
- A pump or small piston that generates pressure
- Fluid-filled lines (pipes or hoses) that carry the pressurized fluid
- An actuator (cylinder or motor) that converts fluid pressure back into mechanical force
Hydraulic lifts, brakes, and presses all rely on this principle. The trade-off is that while force is multiplied, the smaller piston must travel a greater distance than the larger piston, conserving energy.
Hydrostatic paradox
The hydrostatic paradox is a result that surprises most students the first time they encounter it.
Explanation of hydrostatic paradox
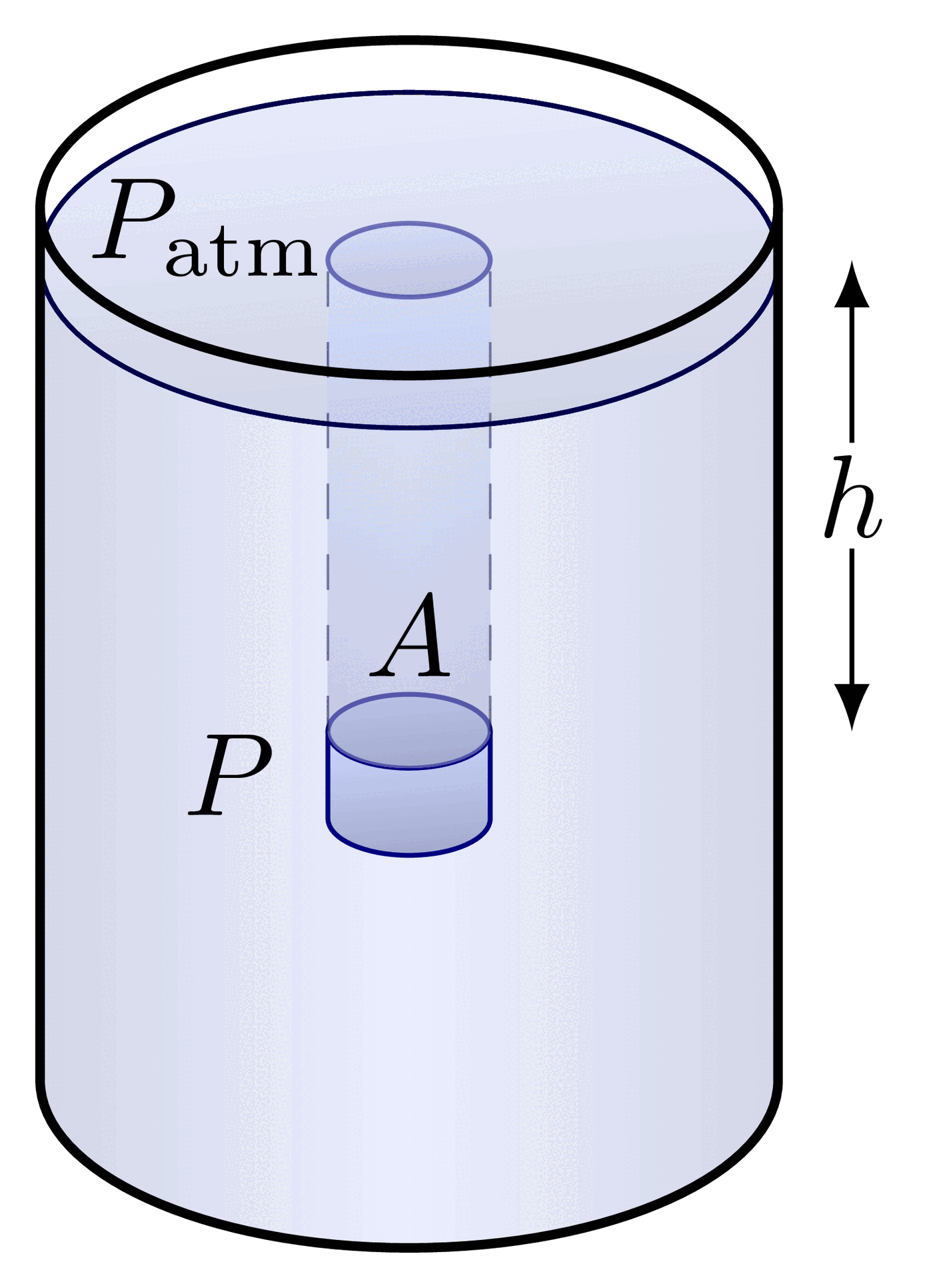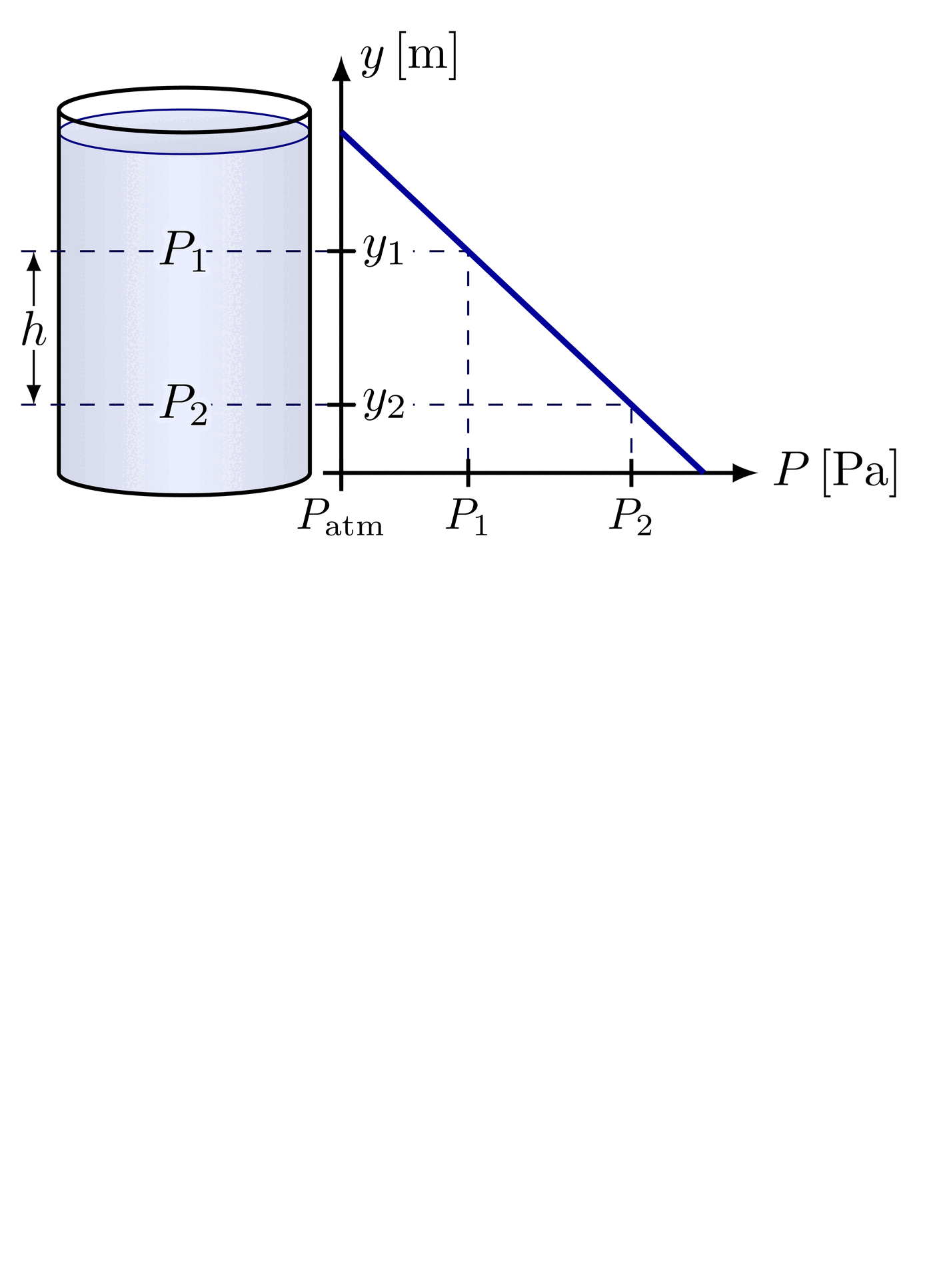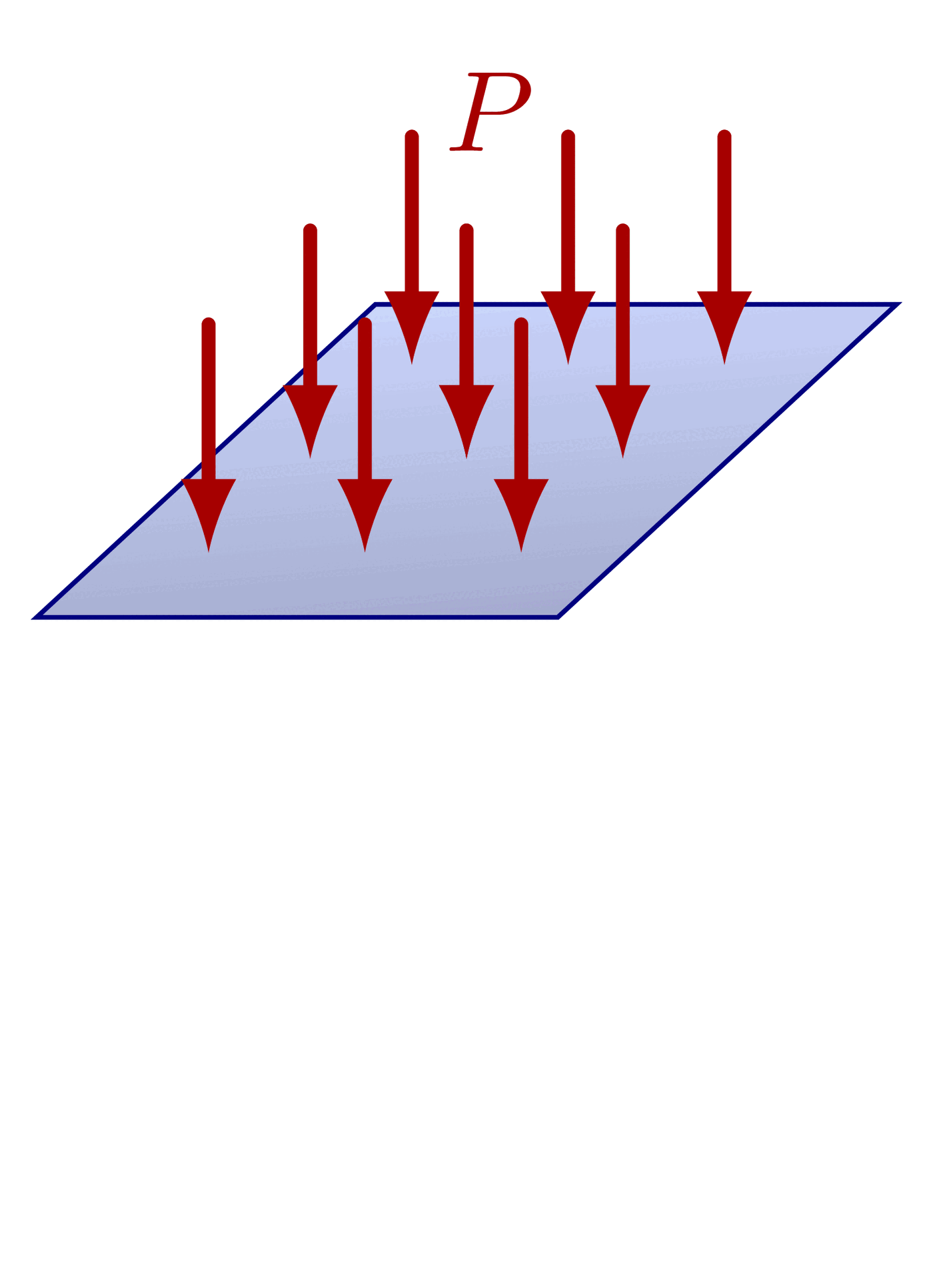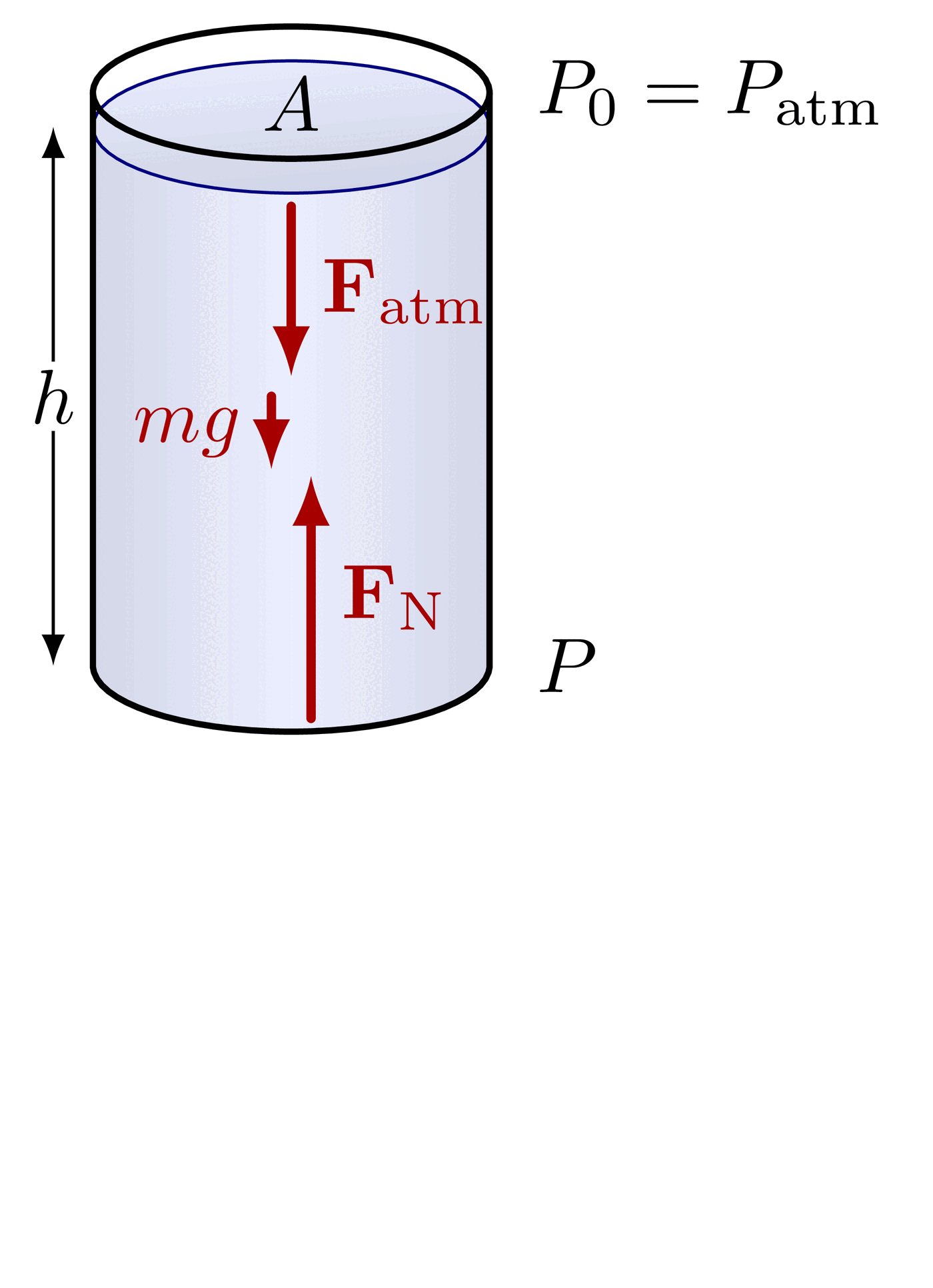
The pressure at the bottom of a container depends only on the height of the fluid and its density, not on the shape or total volume of fluid in the container. A tall, narrow tube and a wide, open tank filled to the same height with the same fluid will have identical pressures at their bases.
Pressure independence of container shape
This follows directly from . The equation contains no term for the container's cross-sectional area or total fluid volume. Only (depth), (density), and (gravity) matter. The container walls redirect the weight of the fluid so that the base pressure depends solely on the vertical height of the fluid column.
Implications for fluid system design
This paradox has real engineering consequences. A thin vertical pipe connected to a large tank can generate enormous pressure at the base if the pipe is tall enough, even though the pipe holds very little fluid. Engineers designing dams, storage tanks, and sealed systems must account for the maximum fluid height, not just the total volume stored. Ignoring this has led to actual structural failures.
Measuring pressure
Accurate pressure measurement is essential for monitoring and controlling fluid systems. Different devices suit different applications.
Types of pressure gauges
Common mechanical pressure gauges include:
- Bourdon tube gauges: A curved tube straightens under pressure, moving a needle. These are rugged and widely used in industrial settings.
- Diaphragm gauges: A flexible membrane deflects under pressure. Good for low-pressure measurements and corrosive fluids.
- Piezoresistive gauges: Use a material whose electrical resistance changes with applied pressure. Common in electronic systems.
Manometers and pressure measurement
A manometer measures pressure by balancing it against a column of liquid (usually water or mercury). The most common type is the U-tube manometer:
- A U-shaped tube is partially filled with a manometer fluid.
- One end connects to the system whose pressure you want to measure.
- The other end is open to the atmosphere.
- The height difference between the two liquid columns gives the gauge pressure: .
Manometers are simple, have no moving parts, and require no calibration, which makes them reliable reference instruments.
Pressure transducers and sensors
Pressure transducers convert pressure into an electrical signal, making them ideal for automated systems and data logging. Common types include strain gauge, capacitive, and piezoelectric transducers. They offer high accuracy, fast response times, and easy integration with electronic control systems.
Pressure forces on submerged surfaces
When a surface is submerged in a fluid, the hydrostatic pressure distribution creates forces that engineers must account for in structural design.
Pressure distribution on submerged planes
The pressure on a submerged plane surface varies linearly with depth, following . At the top of the surface (closest to the free surface), pressure is lower; at the bottom, it's higher. The pressure always acts perpendicular to the surface, regardless of whether the surface is horizontal, vertical, or inclined.
Resultant force and center of pressure
To find the total force on a submerged plane surface:
- Determine the pressure at the centroid of the surface: , where is the depth of the centroid.
- Multiply by the surface area: .
The center of pressure is the point where this resultant force effectively acts. For a vertical or inclined surface, the center of pressure is always below the centroid because pressure increases with depth, weighting the force distribution toward the bottom.
Buoyancy and Archimedes' principle
Buoyancy is the net upward force a fluid exerts on a submerged or partially submerged object. Archimedes' principle states:
The buoyant force on an object equals the weight of the fluid displaced by that object.
Mathematically:
The buoyant force acts through the center of buoyancy, which is the centroid of the displaced fluid volume. An object floats when the buoyant force equals its weight, and sinks when its weight exceeds the buoyant force. The relative positions of the center of gravity and center of buoyancy determine whether a floating object is stable or tends to tip over.