Fundamentals of surface tension
Surface tension is a property of liquids that causes their surfaces to behave like elastic sheets. It arises from cohesive forces between liquid molecules and drives phenomena like capillary action, meniscus formation, and the behavior of soap films and bubbles.
Understanding surface tension matters in fluid dynamics because it governs how fluids behave in narrow spaces, at interfaces, and at small scales. You'll encounter its effects in microfluidics, inkjet printing, respiratory physiology, and even insect locomotion on water.
Molecular origin of surface tension
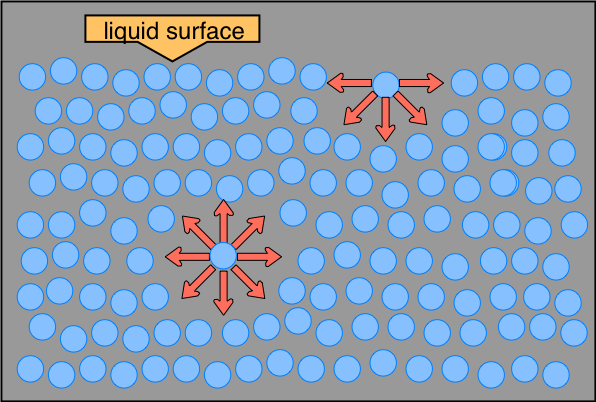
Think about a molecule sitting deep inside a liquid. It's surrounded on all sides by other molecules pulling on it equally, so the net force on it is zero. Now think about a molecule at the surface. It has neighbors below and to the sides, but very few above (just air). That imbalance creates a net inward force on every surface molecule.
This inward pull causes the liquid to minimize its surface area, which is why the surface behaves like a stretched elastic membrane. A sphere has the smallest surface area for a given volume, which is exactly why free-floating droplets are spherical.
Cohesive vs. adhesive forces
- Cohesive forces are attractive forces between molecules of the same substance (liquid-liquid interactions).
- Adhesive forces are attractive forces between molecules of different substances (liquid-solid or liquid-gas interactions).
The balance between these two determines wetting behavior. When adhesive forces dominate (water on clean glass), the liquid spreads out and wets the surface. When cohesive forces dominate (mercury on glass), the liquid beads up and resists spreading.
Surface tension coefficient
The surface tension coefficient, denoted , is expressed in units of force per unit length (N/m) or equivalently energy per unit area (J/m²). These two interpretations are physically identical but useful in different contexts:
- As force per length, represents the force required to stretch a liquid surface along a line of unit length.
- As energy per area, represents the energy needed to create a new unit area of surface.
Typical values at room temperature range from about 20 to 80 mN/m. Water has a relatively high surface tension of about 72 mN/m due to strong hydrogen bonding, while ethanol sits around 22 mN/m.
Factors affecting surface tension
- Temperature: Surface tension decreases as temperature rises. Higher thermal energy means molecules move more vigorously, weakening cohesive forces at the surface.
- Solutes: Surfactants (like soap) lower surface tension by accumulating at the interface and disrupting cohesive interactions. Some inorganic salts slightly increase surface tension by strengthening the water structure in the bulk.
- Electric fields: An applied electric field can redistribute charge at the surface, modifying the effective surface tension. This is the basis of electrowetting.
- Surface contamination: Even trace impurities at the surface can significantly alter , which is why clean surfaces are critical in measurement.
Capillary action
Capillary action is the ability of liquids to flow through narrow spaces without external forces, and sometimes against gravity. It drives fluid transport in plants (water moving from roots to leaves), wicking in paper towels, and flow in microfluidic devices.
Capillary rise in tubes
When you dip a narrow tube into a liquid, the liquid level inside the tube can spontaneously rise above (or drop below) the surrounding liquid level. Whether it rises or falls depends on the contact angle:
- Wetting liquid (contact angle ): The liquid rises. Adhesive forces pull the liquid up the tube wall, and surface tension sustains the column.
- Non-wetting liquid (contact angle ): The liquid is depressed below the outside level. Cohesive forces dominate, and the liquid resists climbing the wall.
The height of rise depends on the tube radius, the liquid's surface tension, and the contact angle.
Contact angle and wettability
The contact angle is measured at the three-phase contact line where liquid, solid, and vapor meet. It's the angle between the solid surface and the tangent to the liquid surface at that line.
- : Complete wetting (liquid spreads into a thin film)
- : Partial wetting (liquid spreads but forms a definite droplet edge)
- : Non-wetting (liquid beads up on the surface)
Two classic examples: water on clean glass has (strong adhesion), while mercury on glass has (strong cohesion).
Jurin's law
Jurin's law gives the equilibrium height of a liquid column in a capillary tube:
where:
- = height of the liquid column
- = surface tension
- = contact angle
- = liquid density
- = gravitational acceleration
- = tube radius
The key takeaway: height is inversely proportional to tube radius. Halve the radius, and the liquid rises twice as high. This is why capillary effects become dominant at small scales and are negligible in wide containers.
Capillary pressure
The Young-Laplace equation describes the pressure difference across any curved liquid-vapor interface:
where and are the two principal radii of curvature of the interface.
- A concave meniscus (wetting liquid) produces a positive capillary pressure that pulls liquid upward.
- A convex meniscus (non-wetting liquid) produces a negative capillary pressure that pushes liquid downward.
This pressure difference is what drives liquid flow through porous media and maintains the stability of liquid bridges and menisci.
Meniscus shapes
A meniscus is the curved liquid-vapor interface that forms where a liquid contacts a solid surface. Its shape is set by the competition between surface tension (which curves the interface) and gravity (which flattens it), along with the wetting properties of the liquid on the solid.
Concave vs. convex menisci
- Concave menisci form with wetting liquids (). The liquid surface curves upward near the solid wall. Water in a glass tube is the classic example.
- Convex menisci form with non-wetting liquids (). The liquid surface curves downward near the wall. Mercury in a glass tube shows this behavior.
The meniscus shape directly determines the sign and magnitude of the capillary pressure, which in turn controls how the liquid behaves in confined geometries.
Radius of curvature
The curvature of the interface at any point is described by two principal radii of curvature, and , measured in two perpendicular planes. Smaller radii mean a more tightly curved surface and a larger capillary pressure (per the Young-Laplace equation).
For a spherical interface (like a small bubble), , and the equation simplifies to .
Laplace pressure
Laplace pressure is the pressure difference across a curved interface, as given by the Young-Laplace equation. A few important consequences:
- The pressure inside a soap bubble is higher than outside. A smaller bubble has a higher internal pressure than a larger one (because is smaller).
- Laplace pressure explains why small bubbles shrink and large ones grow when connected (coarsening in foams).
- It also accounts for the pressure drop that drives capillary flow in narrow channels and porous materials.
Surface tension effects
Soap films and bubbles
Soap molecules are surfactants: they have a hydrophilic (water-loving) head and a hydrophobic (water-repelling) tail. When dissolved in water, they migrate to the surface and orient with their tails pointing outward, reducing the surface tension.
A soap film is a thin water layer sandwiched between two layers of surfactant molecules. This structure is what allows the film to be stable enough to form bubbles. Inside a bubble, the air pressure is slightly higher than outside due to Laplace pressure. For a soap bubble with two surfaces (inner and outer), the pressure difference is rather than .
Marangoni effect
The Marangoni effect is fluid flow driven by gradients in surface tension along an interface. Whenever surface tension varies from point to point (due to temperature differences, concentration differences, or uneven surfactant distribution), liquid flows from regions of low surface tension toward regions of high surface tension.
This effect shows up in many contexts:
- Spreading of oil spills on water
- Uneven drying patterns in paint films
- Behavior of the tear film on the eye's surface
- Convection cells in thin liquid layers heated from below
Tears of wine phenomenon
After you swirl wine in a glass, you'll notice droplets ("tears") forming and running down the inside of the glass. This is a direct consequence of the Marangoni effect.
- A thin film of wine coats the glass above the bulk liquid.
- Alcohol evaporates from this thin film faster than from the bulk (more surface area relative to volume).
- As alcohol leaves, the remaining liquid has a higher water fraction and therefore higher surface tension.
- The surface tension gradient pulls liquid upward from the bulk (lower surface tension) toward the film (higher surface tension).
- The accumulated liquid eventually forms droplets heavy enough to run back down under gravity.
This cycle repeats continuously, producing the characteristic "tears" or "legs."
Capillary waves
Capillary waves are small ripples on a liquid surface where the restoring force is surface tension rather than gravity. They have short wavelengths (typically a few millimeters or less) and are quickly damped by viscosity.
By contrast, gravity waves have longer wavelengths and are restored by gravitational forces. The crossover between the two regimes occurs at a wavelength called the capillary length, , which is about 17 mm for water.
Capillary waves are relevant to light scattering from liquid surfaces, the formation of capillary bridges between particles, and the breakup of liquid jets into droplets.

Measurement techniques
Several experimental methods exist for measuring surface tension, each suited to different situations.
Capillary rise method
This method measures the height a liquid climbs in a narrow tube and calculates from Jurin's law. It's simple and inexpensive, but accuracy depends on knowing the tube radius precisely and having a well-defined contact angle. It works best for liquids with moderate to high surface tension.
Wilhelmy plate method
A thin plate (typically platinum or glass) is partially immersed in the liquid, and the downward force on the plate due to the meniscus is measured with a sensitive balance. Surface tension is calculated from:
where is the measured force, is the wetted perimeter of the plate, and is the contact angle. This method is widely used for studying dynamic surface tension of surfactant solutions because measurements can be taken continuously as the surface ages.
Du Noüy ring method
A thin platinum or platinum-iridium wire ring is slowly pulled upward through the liquid surface. The maximum force needed to detach the ring from the surface is recorded and converted to surface tension using a correction factor that accounts for ring geometry and liquid density.
This method is quick and reliable, making it common in industrial quality control. However, results are sensitive to ring cleanliness and alignment.
Pendant drop method
A droplet is formed at the tip of a needle and allowed to hang. Its shape results from the balance between surface tension (which makes it spherical) and gravity (which elongates it). By capturing a high-resolution image of the drop profile and fitting it to the Young-Laplace equation numerically, you can extract with high accuracy.
This method is non-invasive and works well for liquids with low surface tension or when only a small sample volume is available. It does require a good imaging setup and stable drop formation.
Applications of surface tension
Microfluidics and lab-on-a-chip devices
At the microscale, surface tension dominates over gravity and inertia. This makes it a powerful tool for controlling fluids in microchannels:
- Capillary forces can passively pump liquids without external pressure sources.
- Controlled surface chemistry allows precise droplet formation and stable two-phase interfaces.
- Surface tension-driven flows enable mixing, sorting, and separating particles or cells on chip.
Engineering the surface properties of microchannel walls (through electrowetting, chemical patterning, or surface coatings) is central to designing functional microfluidic systems.
Inkjet printing technology
In inkjet printing, tiny ink droplets are ejected from a nozzle and deposited on a substrate. Surface tension affects every stage of this process:
- Droplet formation: Surface tension determines the droplet size and stability as it detaches from the nozzle.
- Flight: Surface tension keeps the droplet spherical during transit.
- Impact and spreading: The ink's surface tension relative to the substrate's surface energy controls how much the droplet spreads, which directly affects print resolution.
Surfactants are commonly added to ink formulations to tune surface tension for optimal performance on different substrates (paper, plastic, textiles).
Lung surfactants and respiratory disorders
The alveoli in your lungs are tiny air sacs lined with a thin layer of fluid. Without surfactant, the surface tension of this fluid would cause the alveoli to collapse during exhalation (following Laplace pressure: smaller alveoli would have higher pressure and empty into larger ones).
Lung surfactant, a mixture of lipids and proteins, reduces alveolar surface tension and stabilizes the alveoli. When surfactant is insufficient or dysfunctional, serious conditions can result:
- Neonatal respiratory distress syndrome (NRDS): Premature infants often lack sufficient surfactant. Treatment involves administering exogenous surfactant directly into the lungs.
- Acute respiratory distress syndrome (ARDS): Surfactant dysfunction in adults due to lung injury or infection.
Insect locomotion on water surfaces
Water striders and similar insects can walk on water because their weight is supported by surface tension forces acting on their legs. This works because:
- The insect's legs are coated with hydrophobic wax and covered in microscopic hairs (setae), making them extremely water-repellent.
- Each leg creates a dimple in the water surface without breaking through.
- The upward component of the surface tension force along the dimple's perimeter supports the insect's weight.
These insects also use surface tension for propulsion. Water striders row their legs to generate surface waves and vortices that push them forward. Studying these mechanisms has inspired the design of water-walking robots and novel aquatic propulsion systems.