The Nernst equation connects cell potential to concentration and temperature in electrochemical reactions. It's crucial for understanding how these factors affect the energy available in a system, helping predict reaction spontaneity and equilibrium conditions.
This equation bridges thermodynamics and electrochemistry, allowing us to calculate cell potentials under non-standard conditions. It's a powerful tool for analyzing real-world electrochemical systems and their behavior in various environments.
Thermodynamic Principles and the Nernst Equation
Derivation of Nernst equation
- Relates Gibbs free energy () to cell potential () using
- represents number of electrons transferred in redox reaction
- is Faraday's constant (96,485 C/mol)
- Change in Gibbs free energy also depends on standard Gibbs free energy change () and reaction quotient () via
- is the gas constant (8.314 J/mol·K)
- is the temperature in Kelvin
- Combining equations and solving for yields Nernst equation
- At standard temperature (298 K), Nernst equation simplifies to
Applications of the Nernst Equation
Application of Nernst equation
- Calculates cell potentials under non-standard conditions (concentrations ≠ 1 M or gas pressures ≠ 1 atm)
- Steps to calculate non-standard cell potential:
- Determine standard cell potential () from table of standard reduction potentials
- Calculate reaction quotient () based on concentrations or partial pressures of reactants and products
- Substitute values into Nernst equation and solve for
- Same process applies to calculate potential of individual electrodes under non-standard conditions
- Use standard reduction potential of electrode instead of standard cell potential
Concentration effects on cell potentials
- Nernst equation reveals cell potential depends on concentrations of reactants and products
- Increasing reactant concentration or decreasing product concentration increases cell potential
- Decreasing reactant concentration or increasing product concentration decreases cell potential
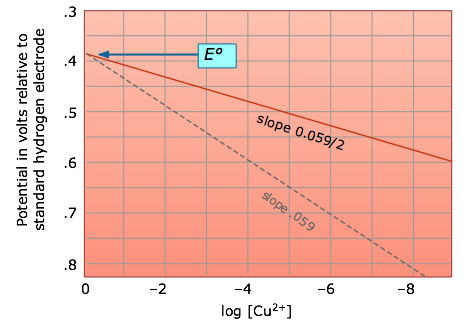
- Magnitude of change in cell potential depends on reaction stoichiometry
- For 1:1 stoichiometry, tenfold concentration change results in change in cell potential
- For other stoichiometries, change in cell potential calculated using Nernst equation

Relationship between Cell Potential, Free Energy, and Equilibrium Constants
Cell potential vs free energy
- Cell potential and Gibbs free energy related by
- Under standard conditions, becomes
- Standard Gibbs free energy change related to equilibrium constant () by
- Combining equations yields relationship between standard cell potential and equilibrium constant
- These relationships allow:
- Calculation of equilibrium constant from standard cell potential
- Calculation of standard cell potential from equilibrium constant
- Determination of redox reaction spontaneity based on sign of cell potential or Gibbs free energy change