Nucleotides are the building blocks of DNA and RNA. They're made of a nitrogenous base, a sugar, and phosphate groups. These components work together to store and transmit genetic information in living organisms.
Understanding nucleotides is crucial for grasping how genetic material functions. The structure of nucleotides determines how DNA and RNA form, replicate, and carry out their roles in biological processes like protein synthesis and gene expression.
Nucleotide Components
Building Blocks of Nucleic Acids
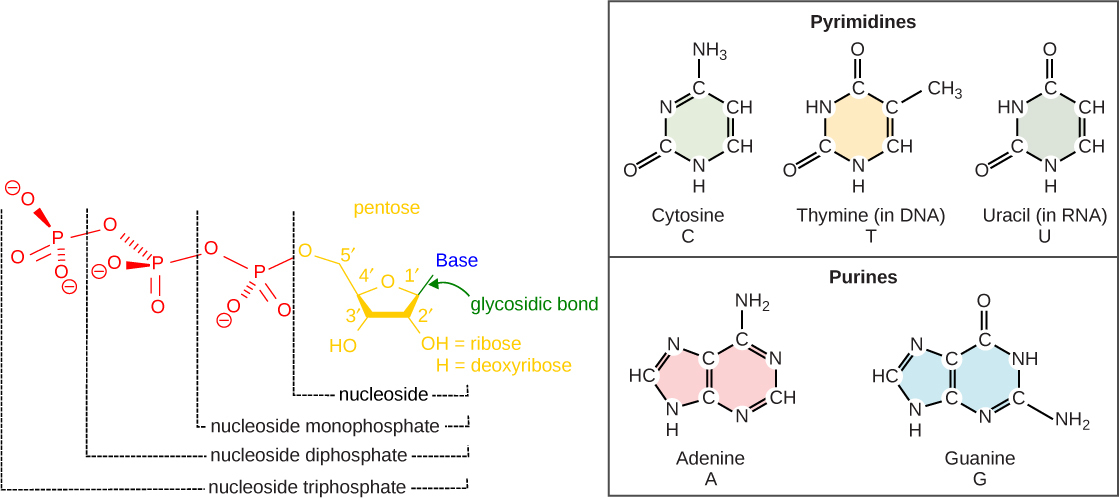
A nucleotide has three parts: a nitrogenous base, a pentose sugar, and one or more phosphate groups. Remove the phosphate group(s) and you're left with a nucleoside, which is just the base attached to the sugar.
The nitrogenous bases are heterocyclic aromatic compounds containing nitrogen. They carry the actual genetic information. There are two families:
- Purines: adenine (A) and guanine (G)
- Pyrimidines: cytosine (C), thymine (T), and uracil (U)
Sugar and Phosphate Backbone
The pentose sugar is either ribose (in RNA) or 2'-deoxyribose (in DNA). The key structural difference: ribose has a hydroxyl group () at the 2' carbon, while deoxyribose has just a hydrogen () there. That single missing oxygen is where the "deoxy" in deoxyribose comes from, and it makes DNA more chemically stable than RNA.
Phosphate groups attach to the 5' carbon of the sugar. Nucleotides can carry one, two, or three phosphate groups, giving you nucleoside mono-, di-, or triphosphates (e.g., AMP, ADP, ATP).
When nucleotides polymerize into a strand, phosphodiester bonds link the 3' hydroxyl of one sugar to the 5' phosphate of the next. This sugar-phosphate chain forms the backbone of the nucleic acid, and it gives each strand a directionality: a 5' end and a 3' end.

Nitrogenous Bases
Purine and Pyrimidine Structures
Purines (A and G) have a fused double-ring structure: a six-membered ring joined to a five-membered ring, both containing nitrogen. They're the larger of the two base types.
Pyrimidines (C, T, and U) have a single six-membered ring containing nitrogen. They're smaller than purines. A useful way to remember which is which: pYrimidine has a "Y," and so does cYtosine, thYmine, and Uracil (okay, that last one doesn't help, but two out of three isn't bad).
Thymine is found exclusively in DNA, while uracil replaces it in RNA. Structurally, thymine is just uracil with a methyl group at the C-5 position.
Nomenclature and Tautomerism
Nucleotide names encode their components. For example, deoxyadenosine 5'-triphosphate (dATP) tells you:
- "deoxy" → deoxyribose sugar
- "adenosine" → adenine base + sugar (the nucleoside)
- "triphosphate" → three phosphate groups
Tautomerism is a concept that matters for understanding mutations. Certain bases can shift between structural isomers called tautomers. The two main forms are:
- Keto form (): the dominant, "normal" form
- Enol form (): a rare alternative
When a base briefly adopts its rare tautomeric form during replication, it can hydrogen-bond with the wrong partner. This leads to non-canonical base pairing and, if uncorrected, a point mutation in the next round of replication.
Nucleotide Interactions
Hydrogen Bonding and Base Pairing
Hydrogen bonding between complementary bases is what holds the two strands of DNA together. The pairing follows strict rules:
- A pairs with T (in DNA) through two hydrogen bonds
- G pairs with C through three hydrogen bonds
- In RNA, A pairs with U through two hydrogen bonds
Because G-C pairs have three hydrogen bonds compared to A-T's two, DNA regions rich in G-C content are harder to denature (separate) and have higher melting temperatures.
These base-pairing rules also explain Chargaff's rules: in any double-stranded DNA molecule, the amount of A equals T, and the amount of G equals C. Notice that purines always pair with pyrimidines, which keeps the width of the double helix uniform.
Beyond stabilizing the DNA double helix, hydrogen bonding between bases also drives the formation of secondary structures in RNA, such as hairpins, stem-loops, and internal loops. These structures are critical for RNA function in processes like translation and gene regulation.