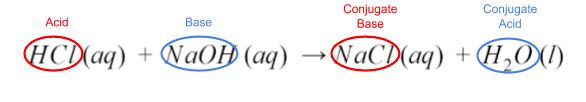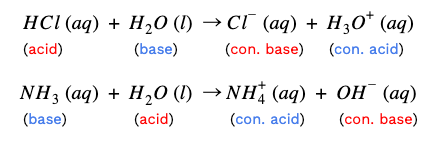What Are Titrations?
Titrations are an experimental method used to determine the unknown concentration of a chemical solution. There are two key substances in a titration: the titrant and the analyte.
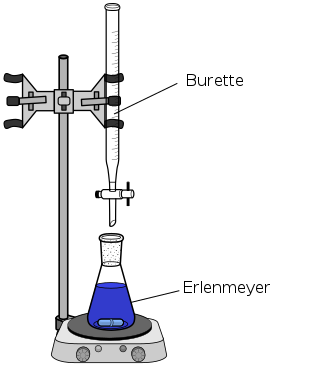
- The titrant is a solution of known concentration that is used to determine the concentration of the unknown solution in a titration. It is usually added to the unknown solution using a burette, which you can see is a long, narrow tube with a stopcock. This allows the chemist to precisely control the volume of titrant they add to the unknown solution.
- The analyte is the unknown solution whose concentration is determined in a titration. It is typically in the Erlenmeyer flask below the burette. You may sometimes see the analyte referred to as the titrand. The image below shows a set-up of the apparatus chemists use to conduct titrations.

Types of Titrations
There are several types of titrations in chemistry, but this course specifically focuses on acid-base titrations. We'll focus on them in this study guide, but here is a quick rundown of the others as well:
- Acid-base titrations: Acid-base titrations are used to determine the concentration of an acid or a base in a solution. The endpoint in an acid-base titration is usually indicated by a pH change, which can be detected using a pH meter or an indicator solution.
- Redox titrations: Redox titrations are used to determine the concentration of an oxidizing or reducing agent in a solution. The endpoint in a redox titration is usually indicated by a color change, which can be detected using an indicator solution or an electronic meter.
- Precipitation titrations: Precipitation titrations are used to determine the concentration of a substance that can form a precipitate with another substance. The endpoint in a precipitation titration is usually indicated by the formation of a precipitate, which can be detected visually or by measuring the light transmittance of the solution.
- Complexation titrations: Complexation titrations are used to determine the concentration of a complexing agent in a solution. The endpoint in a complexation titration is usually indicated by a change in the color or absorption of the solution, which can be detected using an indicator solution or a spectrophotometer. You're probably wondering what an endpoint is, but we'll go over that shortly!
Acid-Base Titrations
Don't worry about the other types of titrations! Let's dig deeper into acid-base titrations.
The titrant in the burette is typically a strong acid or base (and remember, we know its concentration). The analyte of unknown concentration is in the Erlenmeyer flask beneath the burette and is usually a weak acid or base.
Both the burette and Erlenmeyer flask have tick marks for measurement, so we know the initial volumes of both solutions and can do the necessary math to solve for the molarity of the analyte. Remember, molarity (M) is a measure of concentration and can be calculated by dividing the moles of the substance by its volume in liters. This is why we need to keep a record of how much solution is used.
Along with the weak acid or base, the Erlenmeyer has a drop or two of an indicator. There are different types of indicators, and each indicator changes the solution’s color when it is within a certain pH range.
Carrying out a Titration
When you a preparing a titration, you:
- Fill the burette with the titrant of known concentration. Note the concentration and volume of the titrant in the burette before starting the titration.
- Measure out the desired volume of the analyte and place it in the Erlenmeyer flask.
- Add a few drops of indicator to the analyte in the Erlenmeyer flask. Now, you're ready to start the titration! From here, you gradually add the titrant from the burette into the flask, stirring constantly, until the indicator changes the color of the solution.
There are two points during a titration that are kept note of:
- Equivalence point: The equivalence point is the point at which the number of moles of titrant added is equal to the number of moles of the analyte. At the equivalence point, the reactants are completely consumed and the products are present in stoichiometric amounts. It indicates the end of the reaction.
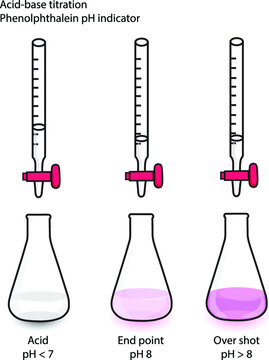
- Endpoint: The endpoint is the point at which there is a visible change in the Erlenmeyer flask, such as a color change or a pH change.
When the correct indicator is used, the endpoint of the titration indicated by a color change occurs at the equivalence point where the moles of titrant = the moles of the analyte.
A common indicator is phenolphthalein, which makes the solution light-pink at the equivalence point. If the analyte is a weak acid and the titrant is a strong base, the pH of the analyte is less than 7 before the titration begins. At this point, the phenolphthalein makes the solution colorless. However, as the base is added, the solution's pH increases. At the endpoint, once a certain amount of strong base is added, the solution changes color.
Image Courtesy of Adobe StockGraphical Representations of Titrations
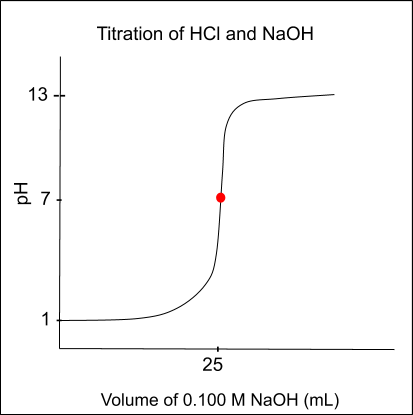
A titration curve can be produced after completing a titration. A titration curve is a graphical representation of the relationship between the volume of titrant added and the corresponding pH of the analyte.
Below is a sample graph of the curve:
There are three main regions on a titration curve:
- The linear region: The linear region is the portion of the curve where the pH of the analyte is pretty constant as more titrant is being added to the solution.
- The inflection point: The inflection point is the point on the curve where the slope changes. It indicates the equivalence point, which is the point at which the number of moles of titrant added is equal to the number of moles of the analyte. In the graph above, it is the red dot at a pH of 7.
- The endpoint: The endpoint is the point on the curve where the chemical reaction between the titrant and the analyte is complete and is indicated by a color change. Remember, you typically want the endpoint and equivalence point to be the same.
Simple Titration Calculations
Since the equivalence point is when the number of moles of acid and base are equal, we can make an equation using molarity (moles/L) and volume:
moles of acid = moles of base
Since moles = molarity x liters, we can say that the molarity of the titrant times the volume of the titrant equals the molarity of the analyte times the volume of the analyte or MaVa = MbVb.
We know the titrant’s concentration going into the titration, so we can use that for Ma. Va is the amount of titrant added to the Erlenmeyer flask. Vb is the analyte’s initial volume. We can plug in these values and then solve for Mb.
It’s important to note that if the mole ratio of the acid and base is NOT 1:1, then you would need to multiply the appropriate sides by their mole ratio. For example, if an acid and base have a mole ratio of 1:2, then this would be the equation: MaVa = 2MbVb
Titration Calculations Example
The following question is from the Advanced Placement YT Channel.
A solution of vinegar contains an unknown amount of acetic acid, HC₂H₃O₂. A 25.0 mL sample of vinegar is titrated with 0.650 M NaOH according to the chemical reaction below. If it requires 32.04 mL of the titrant to reach the equivalence point, what is the concentration of HC₂H₃O₂ in the vinegar?
HC₂H₃O₂ (aq) + NaOH (aq) → H₂O (l) + NaC₂H₃O₂ (aq)
This requires the simple use of MaVa = MbVb. Since the mole ratio is 1:1, we can just plug in and solve!
(Ma)(25.0 mL) =(0.650 M)(32.04 mL)
Ma = 0.833 M
Acids and Bases
Acid-base reaction equations illustrate what is happening during titrations. In the Brønsted-Lowry definition, acids are proton donors and bases are proton acceptors. The species that donated the proton becomes the acid’s conjugate base. Similarly, the species that accepted the proton is now the conjugate acid.
By looking at chemical equations, we can identify which species are the acid, base, conjugate acid, and conjugate base. Check out the example below:
HCl (aq) + NaOH (aq) → NaCl (aq) + H₂O (l)
Take a look at the reactants and the products. Notice that this is a double replacement reaction. The reactants’ ions split up, and the H⁺ from HCl joined with OH⁻ to make water. The remaining ions are then combined.
Since HCl gave up its proton, and hydroxide from NaOH accepted it, HCl is the acid, and NaOH is the base. Their corresponding products are their conjugates.
Additionally, the equation illustrates another property of acid-base reactions. When reacted completely, the acid and base neutralize each other, creating a salt and water. Water is a special case--it’s amphiprotic. This is a fancy term to describe how water can be both a proton donor and a proton acceptor. In other words, water can act as either an acid or a base.
In the first example, HCl donated a proton to water, so water acts like a base here. However, in the second equation, NaOH accepts a proton from water, so water acts like an acid. Finally, another note: ionizing acids and bases in water reveal the strengths of their conjugate pairs.
Shortcut
There is a quick way to figure out the conjugate acid and base! First, find the pairs!
Let's use NH₃ + H₂O → NH₄⁺ + OH⁻ as the example.
NH₃ and NH₄⁺ are the first pair.
H₂O and OH⁻ are the second pair.
But how do we know which one is the acid and which one is the base in each pair?💭
Simple! The compound with the extra hydrogen is the acid. Therefore, NH₄⁺ is the conjugate acid and H₂O is the acid.
Review Activity
Identify the acid and base and their conjugates.
- H₂SO₄ (aq) + CH₃NH₂ (aq) → CH₃NH₃⁺ (aq) + HSO₄⁻ (aq)
- NH₃ (aq) + HNO₂ (aq) → NH₄⁺ (aq) + NO₂⁻ (aq) Answers:
🎥 Watch Dylan Black discuss titrations and how to determine the molarity of a solution at the equivalence point.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
analyte | The substance in a solution whose amount or concentration is being determined during a titration. |
endpoint | The observable event or change in property (such as color) that indicates when the equivalence point has been reached in a titration. |
equivalence point | The point in a titration where the analyte is completely consumed by the titrant in a quantitative reaction. |
titrant | A solution of known concentration that is added to an analyte during a titration to determine the amount of analyte present. |
titration | An analytical procedure used to determine the amount of an analyte in solution by reacting it with a titrant of known concentration. |
Frequently Asked Questions
What is titration and why do we use it in chemistry?
Titration is a lab method for finding how much of an analyte is in a solution by adding a titrant of known concentration until they react completely. The titrant (delivered from a burette) contains a species that reacts stoichiometrically and quantitatively with the analyte; the equivalence point is reached when the analyte is totally consumed (use the balanced reaction and stoichiometric ratio to identify it). In practice you watch for an endpoint—an observable change (often color from an indicator like phenolphthalein) that signals you’re essentially at the equivalence point. Titrations let you determine moles (and thus mass or concentration) of unknowns using volume of titrant and its standard concentration (standard solution or primary standard). This skill is tested on the AP exam (identify equivalence point from amounts—CED 4.6.A). For a focused study guide and practice problems, check the Fiveable topic page (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4).
How do you find the equivalence point in a titration?
The equivalence point is when the analyte has been completely consumed by the titrant—i.e., moles titrant added match the stoichiometric ratio needed to react with the analyte (CED 4.6.A.1). Practically: calculate moles analyte (or use its unknown volume × concentration), then use the titrant concentration to find the volume that supplies the required moles (moles = M × V). In the lab you find it by: (1) watching an indicator color change (the endpoint) chosen so its pH-change range overlaps the equivalence pH, or (2) reading the titration curve: the equivalence volume is at the steepest slope (largest ΔpH/ΔV). Note equivalence pH depends on whether you titrate strong/weak acids or bases, so pick an appropriate indicator (phenolphthalein is common for many base/acid titrations). For AP-aligned review and examples see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between equivalence point and endpoint in titrations?
The equivalence point is the true stoichiometric point in a titration—the exact moment when moles of titrant added react exactly with the moles of analyte (what the CED calls the analyte being “totally consumed”). The endpoint is the observable signal you use in the lab (usually a color change from an indicator like phenolphthalein). Ideally the indicator’s endpoint matches the equivalence point, but they can differ slightly—that difference causes systematic error. On the AP you may be asked to identify the equivalence point from amounts or a titration curve (Learning Objective 4.6.A), so remember equivalence = stoichiometry, endpoint = what you observe. For a quick refresher, see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and practice problems at (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about what a titrant and analyte are - can someone explain?
Titrant = the solution you add from the burette; it has a known concentration (a standard solution or one you’ve standardized). Analyte = the substance in the flask whose amount or concentration you’re trying to find (unknown). During a titration you add titrant until the reacting species totally consumes the analyte—that moment (by stoichiometry) is the equivalence point. The endpoint is the observable change (e.g., phenolphthalein color change) that signals you’ve reached—or very nearly reached—that equivalence point. Quick example: if 0.100 M HCl (titrant) reacts 1:1 with NaOH (analyte), equivalence happens when moles HCl added = moles NaOH originally in the flask. Use volume × M to convert. Knowing the titrant concentration and the titrant volume at the endpoint lets you calculate the analyte concentration (LO 4.6.A in the CED). For a focused review, see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does the color change during a titration and what does that tell us?
The color change comes from an indicator—a molecule that has different colors in different chemical forms (usually protonated vs. deprotonated). In an acid–base titration, the titrant reacts with the analyte until the analyte is consumed at the equivalence point (stoichiometric amounts). Around that point the solution’s pH shifts quickly; the indicator switches form and you see a sudden color change. That observable event is the endpoint, which tells you you’ve essentially reached the equivalence point and can use the volume of titrant to calculate moles of analyte (titrant, analyte, equivalence point, endpoint are CED keywords). Indicators must be chosen so their color-change pH range overlaps the expected equivalence pH (phenolphthalein is common for strong base added to weak acid). For AP review, see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I calculate the concentration of an unknown solution using titration data?
Use the titrant’s known concentration and the volume of titrant at the equivalence (endpoint) to get moles, convert by stoichiometry to moles of analyte, then divide by analyte volume. Steps: 1. Calculate moles of titrant: n(titrant) = M(titrant) × V(titrant in L). 2. Use balanced reaction to get moles analyte: n(analyte) = n(titrant) × (stoich. ratio: mol analyte / mol titrant). 3. Find analyte concentration: [analyte] = n(analyte) / V(analyte in L). Quick example (monoprotic acid titrated by NaOH): - 0.1000 M NaOH used, volume at endpoint = 23.45 mL; acid sample = 25.00 mL. - n(NaOH) = 0.1000 × 0.02345 = 0.002345 mol. - Reaction 1:1 → n(HCl) = 0.002345 mol. - [HCl] = 0.002345 / 0.02500 = 0.0938 M. Remember: identify the equivalence point (indicator or titration curve), use correct stoichiometric ratio, and report with proper sig figs (AP free-response often checks these skills). For a focused review, see the Topic 4.6 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when they say the titration reaction goes to completion?
When the CED says a titration reaction “goes to completion,” it means the chemical reaction between titrant and analyte proceeds fully according to its stoichiometry (no significant reverse reaction or leftover reactive species) so that once you reach the equivalence point the analyte has been totally consumed. Practically, you assume mols titrant added × (stoichiometric ratio) = mols analyte initially present. For example, in a 1:1 HCl + NaOH titration, 0.0100 L × 0.100 M NaOH = 0.00100 mol NaOH will neutralize 0.00100 mol HCl. That assumption lets you calculate the analyte amount from buret volumes and concentrations. On the AP exam this is exactly what LO 4.6.A expects—identify the equivalence point from amounts assuming the reaction is quantitative. Use an indicator to spot the endpoint (observable change), but remember endpoint ≈ equivalence point only if the indicator is chosen well. For a quick review see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can you explain step by step what happens during an acid-base titration?
Start: you have an analyte (unknown amount) in a flask and a titrant (known concentration, the standard solution) in a burette. You usually pipette an aliquot of analyte into the flask and add a few drops of an indicator (e.g., phenolphthalein) or use a pH meter. Step 1—deliver titrant slowly from the burette while swirling the flask so reaction is complete and mixed. The titrant reacts quantitatively with the analyte in the stoichiometric ratio of the balanced equation. Step 2—as you approach the equivalence point (exact stoichiometric consumption of analyte by titrant), the indicator will change color (that observed change is the endpoint) or the pH curve shows a steep jump. Endpoint ≈ equivalence point if chosen well. Step 3—record volume of titrant used (final − initial burette reading). Use molarity × volume to get moles titrant, convert by stoichiometry to moles analyte, then to mass or concentration. AP tip: you must be able to identify the equivalence point from amounts or a titration curve (CED 4.6.A). For a quick study guide and practice problems, see the Fiveable titration guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to know the exact concentration of the titrant before starting?
You need the exact concentration of the titrant because titration calculations rely on a known stoichiometric relationship between titrant and analyte at the equivalence point. If the titrant’s molarity is off, your computed moles (and therefore the amount or concentration of the analyte) will be wrong. That’s why titrants are prepared as standard solutions and often standardized against a primary standard before use—the CED calls this out in 4.6.A.1: the titrant must react specifically and quantitatively so the equivalence point (and observed endpoint) tells you how many moles reacted. On the AP exam you’ll be expected to use the known titrant concentration, buret volume change, and stoichiometric ratio to calculate analyte amount (practice that math for free at Fiveable: study guide for Topic 4.6 (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry)).
I don't understand how to identify when the equivalence point is reached - help?
Equivalence point = the moment the analyte is exactly consumed by the titrant (moles reacted = moles added × stoichiometric ratio). Practically: - Do the mole math: at equivalence, n(analyte) = n(titrant) × (stoichiometric factor). For 1:1 acid–base titrations use MaVa = MbVb (so if you start with 25.00 mL of 0.100 M HCl and use 0.100 M NaOH, equivalence is at 25.00 mL NaOH added). - Use the titration curve: equivalence is the center of the steep vertical jump (where pH changes fastest—maximum slope). - Use an indicator chosen so its color-change range overlaps the pH at the equivalence point (endpoint ≈ observable signal; endpoint ≠ always exactly equivalence). Phenolphthalein is commonly used for many strong acid–strong base titrations. - Experimentally read buret volume at the indicator endpoint, then correct for any systematic differences if needed. This is exactly what the CED asks you to show (4.6.A—identify equivalence from amounts). For worked examples and practice, check the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and more practice problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the point of doing titrations instead of just measuring pH directly?
Titrations let you find how much analyte is present using stoichiometry, not just a single pH number. The titrant is a standard solution of known concentration that reacts quantitatively with the analyte; at the equivalence point the analyte is completely consumed. Detecting that point (the endpoint) with an indicator or a titration curve is more accurate for concentration calculations than a single pH reading, especially for weak acids/bases or buffered solutions where pH changes slowly near equivalence. pH electrodes can drift, need calibration, and give a value—but they don’t tell you moles reacted. Titrations convert volume of titrant to moles (useful on the AP: LO 4.6.A and stoichiometry problems). For more on indicators, titration curves, and practice problems for the exam, check the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and the unit resources (https://library.fiveable.me/ap-chemistry/unit-4). For extra practice, see Fiveable’s AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
How do indicators work in titrations and how do you choose the right one?
An indicator is a weak acid or base whose protonated and deprotonated forms are different colors. During an acid–base titration its color switches over a specific pH range (the indicator’s transition interval). The equivalence point is when stoichiometrically all analyte is consumed; the endpoint is the observable indicator color change. For a good indicator you want the indicator’s transition range to overlap the titration curve’s steep region around the equivalence pH. How to choose: - Predict the equivalence pH from the reactants (strong acid + strong base ≈ pH 7; weak acid + strong base >7; weak base + strong acid <7). - Pick an indicator whose color change range falls inside the steep part of the titration curve (e.g., phenolphthalein 8.2–10.0 works well for weak acid/strong base; bromothymol blue 6.0–7.6 for strong acid/strong base; methyl orange 3.1–4.4 for strong acid/weak base). On the AP exam you may be asked to identify equivalence vs endpoint—use stoichiometry and expected equivalence pH to justify your indicator choice. For more examples and practice, see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens if you add too much titrant past the equivalence point?
Past the equivalence point you’ve added more titrant than needed to exactly react with the analyte, so the analyte is fully consumed and the solution’s composition is dominated by excess titrant. For an acid–base titration that means the pH will swing past the equivalence pH (acidic → neutral → basic or vice versa) and the indicator’s color (the endpoint) will stay changed. On a titration curve you’ll see the steep jump at equivalence, then the curve levels off as excess titrant accumulates. Practically, over-titrating makes you read a larger volume of titrant and so you’ll overestimate the amount of analyte if you use that volume in calculations—always stop at the indicator’s correct endpoint (or use the curve/stoichiometry to correct for excess moles: moles excess = moles titrant added − moles required). For a clear review, see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
Why is the equivalence point different from the endpoint and does it matter for calculations?
The equivalence point is the stoichiometric moment when moles of titrant added exactly react with all the analyte (what the CED calls the analyte being “totally consumed”). The endpoint is the observable signal (color change from an indicator, a pH jump on the titration curve) you actually see in the lab. They’re different because an indicator changes color at a particular pH, which may be slightly before or after the true equivalence pH. Does it matter for calculations? Yes, but only if the endpoint and equivalence point are noticeably offset. For AP problems you assume the equivalence point is reached when the volume at the endpoint is used (CED 4.6.A.1), but in real labs you choose an indicator whose transition pKa is close to the equivalence pH so the error is minimal. Large indicator mismatch or poor technique gives systematic error in moles (and therefore concentration/mass). Practice reading titration curves and picking indicators so endpoint ≈ equivalence (see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and more practice problems (https://library.fiveable.me/practice/ap-chemistry)).
Can titrations be used for reactions other than acid-base reactions?
Yes—titrations aren’t limited to acid–base chemistry. The CED definition in 4.6.A focuses on the general idea: a titrant of known concentration reacts specifically and quantitatively with an analyte and the equivalence point is reached when the analyte is consumed. That framework applies to other reaction types too. Common non–acid–base titrations: - Redox titrations (e.g., MnO4– vs. Fe2+; endpoint shown by color change or potentiometry) - Complexometric titrations (EDTA to determine metal ions; metal-ion indicators) - Precipitation titrations (AgNO3 to find Cl–; endpoint by indicator or potentiometric jump) - Back titrations (useful when direct titration isn’t practical) Endpoints can be detected by color indicators, voltage changes, or other property shifts—just like acid–base titrations. For AP purposes, know the equivalence point concept, stoichiometric ratio, and how an endpoint signals equivalence (CED 4.6.A.1). For a quick review, see the Topic 4.6 study guide (https://library.fiveable.me/ap-chemistry/unit-4/intro-titrations/study-guide/8XHQYjYki6GqAcrp18I2) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).