Reactions in Aqueous Solution
When dealing with chemical reactions, it is important to understand that, at least in the case of AP Chemistry, the vast majority of them will be taking place in an aqueous solution, that is, dissolved in water. Therefore, we will not be dealing with molecules, but rather their constituent particles that are created when they dissolve.
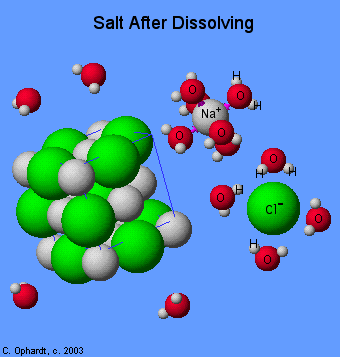
Let's take NaCl for example. When you dissolve table salt in water, it is essentially ripped apart by the water molecules. It is important to understand that water is a polar molecule. Polar molecules have a partial positive end and a partial negative end. Therefore, when an ionic compound like NaCl dissolves, the water molecules rip it apart and trap it, per se.
In other words, ionic compounds dissociate into their ions when dissolved in water. To tie this back to unit three and intermolecular forces, when you dissolve NaCl, you will see ion-dipole interactions. The positive end of the water molecule (hydrogen) is attracted to the negative chloride anion, while the negative end of the water molecule (oxygen) is attracted to the positive sodium cation.
👉 Not sure what ion-dipole forces are or need a refresh? Make sure to review your intermolecular forces with this study guide.
We can write out the dissolution of salt as NaCl (s) → Na⁺ (aq) + Cl⁻ (aq). Thus, if we have a reaction between two compounds in water, it will be the ions/parts of the molecules that dissolve that react together rather than the entire compounds themselves.
Let's look at a specific class of such reaction:

Precipitation Reactions
Sometimes, when two reactants react, one of the products is insoluble. To be insoluble means that it will NOT be ripped apart by water like NaCl would be. In other words, an insoluble product is one that precipitates out as a solid and does not dissolve in water.
👉 In order to fully understand these reactions, you must be aware of some of the general solubility rules that we discussed in the last unit.
One example of a precipitation reaction where one product is insoluble is the precipitation of lead (II) iodide:
2KI (aq) +Pb(NO₃)₂ (aq) → 2KNO₃ (aq) + PbI₂ (s).
Because lead (II) iodide is not soluble in water (according to the general solubility rules linked above), it precipitates and falls out of the solution.
Here is another table of solubility rules, but you do not need to memorize every single one of these. College Board has stated that only the following solubility rule will be assessed on the AP exam: "All sodium, potassium, ammonium, and nitrate salts are soluble in water."
Image Courtesy of Regents Chemistry Reference TableWriting Net Ionic Equations
A net ionic equation is a chemical equation that shows only the species participating in a chemical reaction and omits the spectator ions. Spectator ions are ions that appear on both sides of the arrow in a chemical equation but do not actually participate in the reaction. In precipitation reactions, spectator ions are those that did not participate in the formation of the precipitate, or solid product.
Example #1
Let's see these concepts in action with our previous reaction: 2KI (aq) + Pb(NO₃)₂ (aq) → 2KNO₃ (aq) + PbI₂ (s) If we write this reaction out with all of the ions, we get 2K⁺ (aq) + 2I⁻ (aq) + Pb²⁺ (aq) + 2NO₃⁻ (aq) → 2K⁺ (aq) + 2NO₃⁻ (aq) + PbI₂ (s). This was obtained by dissociating, or separating, each soluble compound into its constituent parts.
Note that PbI₂ does NOT dissociate, since it is insoluble. Be on the lookout for this! Only dissociate soluble compounds. Also, don't dissociate weak acids and bases (you'll learn more about this more in the future).
This equation, with the ions written out, is called the complete ionic equation or the total ionic equation. It shows every ion in the solution that is present during the reaction, including spectator ions.
Now let's take the spectator ions out of the reaction. When looking at the total ionic equation, we see that potassium and nitrate go in and come out the same way, so we can cross them out and cancel them. Potassium and nitrate were the spectator ions!
Once we eliminate the spectator ions, we are left with 2I⁻ (aq) + Pb²⁺ (aq) → PbI₂(s). This is what we call the net ionic equation for the reaction, and it tells us what really happens. Potassium and nitrate were in the flask, but they really didn't do much for us in terms of the formation of the insoluble compound. They could have been any suitable ion and the reaction would have had the same outcome.
General Steps
Now that you have an idea of what a net ionic equation is, it is time to learn how to write one out! First, you must know the basics of representing a chemical reaction. Make sure you review those in the last study guide before moving on.
To work through and learn this process, here are the general steps to writing a net ionic equation:
- Figure out which compounds are soluble and insoluble using solubility rules
- Balance the given chemical equation. It may already be balanced, but it also may not, so you always have to check.
- Write the complete ionic equation by dissociating soluble compounds into ions.
- Omit the spectator ions and write the final net ionic equation of the given reaction. Make sure you include the phase of matter each compound is in.
Example #2
Try writing the net ionic equation for the following reaction: KOH + Fe(NO₃)₃ → KNO₃ + Fe(OH)₃
- Using solubility rules, you should find that: Fe(OH)3 is the solid or the precipitate formed. Add the respective states of matter to each compound in the chemical equation.1. KOH (aq) + Fe(NO₃)₃ (aq) → KNO₃ (aq) + Fe(OH)₃ (s)
- You should notice that there is only 1 OH on left and 3 OH on the right. The same goes for NO3, but vice versa. This means we have to balance the equation.1. 3KOH (aq) + Fe(NO₃)₃ (aq) → 3KNO₃ (aq) + Fe(OH)₃ (s)1.
👉 Not sure how to balance chemical equations yet or need more practice? Make sure to check out the next study guide.
- Write out the complete ionic equation by dissociating all soluble compounds into their constituent ions. If you're ever unsure of what you should and shouldn't dissociate, remember that every compound in the aqueous solution will dissociate and the solid will not.1. 3K⁺ (aq) + 3OH⁻ (aq) + Fe⁺³ (aq) + 3NO₃⁻ (aq) → 3K⁺ (aq) + 3NO₃⁻ (aq) + Fe(OH)₃ (s)
- Identify the spectator ions by taking a look at what ions look the same before and after the chemical reaction took place or identifying which ions are not in the formed precipitate: 3K⁺ (aq) + 3OH⁻ (aq) + Fe⁺³ (aq) + 3NO₃⁻ (aq) → 3K⁺ (aq) + 3NO₃⁻ (aq) + Fe(OH)₃ (s)
- Write out your final answer and net ionic equation: 3OH⁻ (aq) + Fe⁺³ (aq) → Fe(OH)₃ (s)
Each of the above steps are different representations of a chemical reaction. Here is a quick summary of each of the terms that you should familiarize yourself with since the AP exam will expect you to know them:
Image Courtesy of Science Notes🎥 Watch Jacob Jeffries discuss solubility, molecular equations, complete ionic equations, and net ionic equations.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
balanced chemical equation | A chemical equation where the number of atoms of each element is equal on both the reactant and product sides. |
chemical change | A process in which substances are rearranged into new combinations, resulting in the formation of new substances with different properties. |
complete ionic equation | A balanced equation that shows all ions and molecules present in solution, including spectator ions. |
conservation of charge | The principle that the total electric charge is neither created nor destroyed in a chemical reaction; total charge is equal on both sides of the equation. |
conservation of mass | The principle that mass is neither created nor destroyed in a chemical reaction; the total mass of reactants equals the total mass of products. |
molecular equation | A balanced chemical equation that represents all reactants and products in their molecular form. |
net ionic equation | A symbolic representation of a chemical reaction that shows only the ions and molecules that actually participate in the reaction, excluding spectator ions. |
physical change | A change in matter that does not alter the chemical composition or identity of the substance. |
Frequently Asked Questions
What is a net ionic equation and how is it different from a regular chemical equation?
A net ionic equation shows only the species that actually change during a reaction (the ions that form a product), leaving out spectator ions that don’t participate. It’s one of three AP-accepted symbolic forms: molecular, complete ionic, and net ionic (CED 4.2.A.3). Net ionic equations still must be balanced for atoms and charge (CED 4.2.A.2). Quick example: - Molecular: AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq) - Complete ionic: Ag+(aq) + NO3-(aq) + Na+(aq) + Cl-(aq) → AgCl(s) + Na+(aq) + NO3-(aq) - Net ionic: Ag+(aq) + Cl-(aq) → AgCl(s) Use solubility rules to decide what dissociates and identify spectators (here Na+ and NO3-). On the AP, you may be asked to write balanced molecular, complete ionic, or net ionic equations (LO 4.2.A). For a focused review, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS). More practice problems are at (https://library.fiveable.me/practice/ap-chemistry).
How do I write net ionic equations step by step?
Steps to write a net ionic equation (clear, AP-style): 1. Write the balanced molecular equation for the reaction. (CED 4.2.A: equations must be balanced.) 2. Identify states (aq, s, g, l). Use solubility rules to decide which ionic compounds are soluble (so dissociate) and which form a precipitate. 3. Write the complete ionic equation: split all strong electrolytes (soluble salts, strong acids/bases) into ions; keep solids, gases, and weak electrolytes intact. 4. Cancel spectator ions (ions appearing unchanged on both sides). What’s left is the net ionic equation—show only species that change. Check atom and charge conservation. (Keywords: spectator ions, complete ionic, net ionic, precipitation reaction.) 5. Label physical states and make sure the net ionic is balanced for atoms and charge. Quick example: Molecular: AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq) Complete ionic: Ag+ + NO3– + Na+ + Cl– → AgCl(s) + Na+ + NO3– Net ionic: Ag+(aq) + Cl–(aq) → AgCl(s) For more practice and AP-aligned tips, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to learn net ionic equations when we already know molecular equations?
Molecular equations show what compounds react, but net ionic equations show what actually changes at the particle level—the ions and molecules that participate. Net ionic equations remove spectator ions (unchanged ions in solution) so you can: identify the true reacting species (important for precipitation, acid–base, and redox reactions), check charge conservation, track electrons for oxidation–reduction, and do stoichiometry on the species that matter. That aligns with CED LO 4.2.A and EKs 4.2.A.2–4.2.A.3: balanced molecular, complete ionic, and net ionic forms are all useful depending on context. On the AP Exam you may be asked to represent changes using a net ionic equation (Unit 4 tasks and free-response prompts), so practicing net ionic forms builds the “models and representations” skill the exam tests. For a focused review, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS). For more practice problems across Unit 4, try the AP practice bank (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about spectator ions - what are they and why do we remove them?
Spectator ions are ions present in solution that don’t participate in the chemical change—their chemical form and oxidation state stay the same before and after the reaction. We remove them when writing a net ionic equation to focus only on species that actually change (the reacting ions and any precipitate, gas, or weak electrolyte formed). That makes the equation show the true chemistry and helps you check conservation of mass and charge. Quick example: Molecular: AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq) Complete ionic: Ag+(aq) + NO3−(aq) + Na+(aq) + Cl−(aq) → AgCl(s) + Na+(aq) + NO3−(aq) Net ionic: Ag+(aq) + Cl−(aq) → AgCl(s) (Na+ and NO3− are spectators and cancel.) On the AP, net ionic work checks that you know aqueous dissociation, strong electrolytes, precipitation reactions, and charge conservation (Topic 4.2). For more practice and tips, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and hundreds of practice problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between complete ionic equations and net ionic equations?
Complete ionic equations show every strong electrolyte dissociated into ions—you write aqueous strong acids, bases, and soluble salts as their ions (with states) while keeping solids, gases, and weak electrolytes intact. A net ionic equation is just the complete ionic equation with all spectator ions (ions that appear unchanged on both sides) removed—it shows only the species that actually change (usually the precipitate, gas, or molecular product plus the reacting ions). Why it matters for AP Chem: the CED expects you to represent reactions in molecular, complete ionic, and net ionic forms and to conserve mass and charge (4.2.A, 4.2.A.2–3). Use solubility rules and strong/weak electrolyte knowledge to decide what dissociates and which ions are spectators. For practice, check the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and try problems from the Unit 4 page (https://library.fiveable.me/ap-chemistry/unit-4) or the practice bank (https://library.fiveable.me/practice/ap-chemistry).
How do I know which compounds dissociate into ions when writing ionic equations?
You decide which compounds dissociate by asking: is the substance a strong electrolyte (fully dissociates), a weak electrolyte (partially dissociates), or a nonelectrolyte/insoluble (doesn’t give free ions)? For AP net ionic work: - Fully split into ions: soluble ionic salts, strong acids (HCl, HBr, HI, HNO3, HClO4, H2SO4—first H) and strong bases (Group 1 OH–, Ba2+, Sr2+ hydroxides). - Partially split: weak acids/bases (acetic acid, NH3)—write them as molecules, not complete ions. - Do not split: pure solids, liquids, gases, and insoluble salts (precipitates) stay as formulas. Use solubility rules (or Ksp for borderline cases) to decide if a salt is soluble. In complete ionic equations, show all strong electrolytes as ions; cross out spectator ions to get the net ionic. Practice these steps in the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
Why does charge have to be balanced in net ionic equations?
Because charge is conserved just like mass, any symbolic representation of a reaction—including complete ionic and net ionic equations—must show equal total charge on both sides. Net ionic equations remove spectator ions, but they still represent the actual ionic species that gain or lose electrons or recombine. If the charges didn’t balance, the equation would imply creation or destruction of net electric charge, which violates conservation of charge (CED 4.2.A.2). For AP work you must write balanced molecular, complete ionic, and net ionic equations that conserve both atoms and charge (CED 4.2.A). If you get an imbalance, check: did you drop a spectator ion incorrectly, or forget coefficients that multiply ionic charges? For more practice and examples on writing and checking net ionic equations, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS), the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and AP practice sets (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain how to identify spectator ions in a chemical reaction?
Spectator ions are the ions that appear unchanged on both sides of a reaction. To find them quickly: 1. Write the balanced molecular equation. 2. Split all strong electrolytes (soluble salts, strong acids/bases) into their ions to make the complete ionic equation. (Use solubility rules and strong/weak electrolyte ideas from Topic 4.2.) 3. Cancel any ions that are identical on both reactant and product sides—those canceled ions are spectators. 4. The remaining species form the net ionic equation; it shows only the chemical change and conserves charge and atoms (CED 4.2.A). Example: NaCl(aq) + AgNO3(aq) → AgCl(s) + NaNO3(aq) Complete ionic: Na+ + Cl– + Ag+ + NO3– → AgCl(s) + Na+ + NO3– Cancel Na+ and NO3– (spectators). Net ionic: Ag+ + Cl– → AgCl(s). This skill is tested on AP (represent ions and net ionic equations—4.2.A). For review and more examples, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and extra practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens to polyatomic ions like sulfate when writing net ionic equations?
Treat polyatomic ions the same as single ions: keep them intact in complete and net ionic equations unless the reaction actually changes their chemical identity. If a salt with a soluble polyatomic ion (like SO4^2− from Na2SO4) is in aqueous solution, write it as the ion (Na+ and SO4^2−) in the complete ionic equation, then cancel spectator ions to get the net ionic equation. Don’t split sulfate into S and O atoms—you only break species into ions for strong electrolytes that dissociate in water. If the polyatomic ion participates in the reaction (forms an insoluble precipitate, is protonated, or is oxidized/reduced), show that change (you may need to write a molecular or redox equation and then form the ionic forms). Remember to balance atoms and conserve charge (CED 4.2.A.2–3). For a quick refresher, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and practice problems at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand when to use molecular vs complete ionic vs net ionic equations - when do I use each one?
Use the form that best fits what you need to show: - Molecular equation: write full formulas for reactants and products (e.g., NaCl(aq) + AgNO3(aq) → AgCl(s) + NaNO3(aq)). Use this when you just need the overall balanced reaction or when reagents/products are given as compounds (CED: 4.2.A.1–3). - Complete ionic equation: break all strong electrolytes (soluble salts, strong acids/bases) into ions to show every aqueous species (Na+ + Cl– + Ag+ + NO3– → AgCl(s) + Na+ + NO3–). Use this to track all ionic species and charge conservation. - Net ionic equation: cancel spectator ions (ions unchanged) from the complete ionic equation to show only species that react (Ag+ + Cl– → AgCl(s)). Use this when the goal is to show the actual chemical change (precipitation, acid–base, redox) or to apply solubility rules/Ksp. On the AP exam you’ll often be asked explicitly for a net ionic or complete ionic equation (Topic 4.2). For step-by-step practice, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I balance net ionic equations if the charges don't match up?
If the charges don’t match in a net ionic equation, you haven’t conserved charge—so fix it. Quick steps you can use on AP problems (CED 4.2.A: conserve mass and charge): 1. Write the balanced molecular equation. 2. Dissociate strong electrolytes into ions to get the complete ionic equation. 3. Cancel spectator ions to get the net ionic. If charge is unbalanced, adjust stoichiometric coefficients of ionic species (you can’t change charges). That will balance both atoms and total charge. 4. For acid/base contexts, balance remaining H and O by adding H2O and H+ (acidic) or H2O and OH– (basic). 5. For redox reactions, balance charge using electrons via half-reaction method, then combine halves so electrons cancel. Always check that total atoms and total charge are identical on both sides (CED 4.2.A.2). For practice, see the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and try problems at Fiveable’s practice page (https://library.fiveable.me/practice/ap-chemistry).
Why do some compounds stay together as molecules instead of breaking into ions?
Because whether a compound breaks into ions depends on its bonding and how favorable dissociation is. Ionic solids (like NaCl) have ions held in a lattice; if water’s solvation energy overcomes the lattice energy, the solid dissociates into free ions (strong electrolyte). Covalent molecular compounds (like H2O, CO2, most organics) share electrons and don’t form separate ions in solution, so they “stay together” as molecules. Partial cases: weak electrolytes (e.g., acetic acid) only partially ionize because ion formation is not energetically favored. Solubility rules, lattice energy vs. hydration energy, and acid/base tendencies govern which species appear as ions in a complete ionic or net ionic equation (CED keywords: strong/weak electrolytes, solubility rules, spectator ions). For net ionic work, write species that actually exist as ions in solution—leave intact the molecular or insoluble species. See the Topic 4.2 study guide for examples (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the point of removing spectator ions if they're still there in the actual reaction?
Good question—spectator ions are still present physically, but we remove them in net ionic equations because they don’t undergo any chemical change. The point of a net ionic equation is to show only the species that actually participate: bonds forming/breaking, precipitate formation, proton transfer, or redox changes. That makes it easier to check conservation of mass and charge (CED 4.2.A.1–4.2.A.3) and to focus on the reactive core for things like Ksp, acid–base, or stoichiometry reasoning. Example: AgNO3 + NaCl → AgCl(s) + NaNO3. The net ionic is Ag+ + Cl– → AgCl(s). Na+ and NO3– are spectators—they’re there in solution but don’t change, so removing them clarifies what actually reacted. If you want more practice writing complete vs. net ionic equations and exam-style problems, use the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and the AP practice bank (https://library.fiveable.me/practice/ap-chemistry).
How do I write net ionic equations for precipitation reactions?
Steps to write a net ionic equation for a precipitation reaction (quick method): 1. Write the balanced molecular equation for the reactants (double-displacement form). Example: AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq). 2. Use solubility rules and the CED idea that strong electrolytes fully dissociate to write the complete ionic equation. Ag+(aq) + NO3−(aq) + Na+(aq) + Cl−(aq) → AgCl(s) + Na+(aq) + NO3−(aq). 3. Identify and cancel spectator ions (ions unchanged on both sides). Here Na+ and NO3−. 4. Write the net ionic equation with only species that change (must be mass- and charge-balanced): Ag+(aq) + Cl−(aq) → AgCl(s). Notes: always check solubility rules (insoluble product = precipitate) and only split strong electrolytes. Don’t split solids, liquids, gases, or weak electrolytes. This process matches CED 4.2.A (represent ionic species, conserve mass & charge). For practice and the topic study guide, see Fiveable’s Net Ionic Equations study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about which substances are aqueous vs solid when writing ionic equations - how do I tell the difference?
You figure aqueous vs. solid two ways: (1) use the state symbols the problem gives (always follow those), and (2) if states aren’t given, apply solubility rules / chemistry knowledge to decide whether a compound dissociates (aq) or stays a solid (s). Strong electrolytes (soluble ionic salts like NaCl, KNO3, most nitrate salts) are written as dissociated ions in the complete ionic equation; weak electrolytes (weak acids/bases) stay mostly molecular; insoluble salts (AgCl, BaSO4, PbSO4, etc.) are written as solids—they become precipitates and remain as (s) in the net ionic. Also remember gases and pure liquids are not broken into ions (CO2(g), H2O(l)). On the exam you must show molecular → complete ionic → net ionic and cancel spectator ions to conserve mass and charge (CED 4.2.A). If you want a quick refresher and solubility reminders, check the Topic 4.2 study guide (https://library.fiveable.me/ap-chemistry/unit-4/net-ionic-equations/study-guide/VTCEO9cDrSHfPodoWGxS) and practice problems (https://library.fiveable.me/practice/ap-chemistry).