Understanding the structure of an atom is essential to comprehending the photoelectric effect and interpreting photoelectron spectroscopy. Remember that an atom is made up of three subatomic particles:
-
Protons - located in the nucleus with a +1 charge and a mass of approximately 1 amu.
-
Neutrons - located in the nucleus with no charge and a mass of approximately 1 amu.
-
Electrons - found around the nucleus in orbitals with a charge of -1 and no mass.
The movement of electrons will be our focus in this study guide. Be sure to review how to write out the electron configuration of an atom and understand that electrons are not only found in different energy levels or shells but are also located in different subshells:
- When writing out electron configuration, the number (1, 2, 3, etc) represents the principle energy level or electron shell.
- Following the electron shell, you will letters (s, p, d, and f). These letters represent the four unique subshells where electrons can be located. The maximum number of electrons in each subshell, respectively, is 2, 6, 10, and 14.
The following is the electron configuration of boron, element 5 on the periodic table:
If you would like to review how this electron configuration was obtained, check out our study guide on Atomic Structure and Electron Configuration.
The Quantum-Mechanical Model
Before this section, we have been discussing the atom with regard to Bohr's model. However, today chemists use the quantum-mechanical model of an atom to more accurately display how electrons behave. Rather than precisely defining where electrons may exist around a nucleus, the quantum-mechanical model displays how electrons have the probability of existing almost anywhere.

Although you don't have to know this for the AP exam, it may help your understanding of this section. The biggest takeaway of the quantum-mechanical model is Heisenberg's uncertainty principle, which basically states that it is virtually impossible to know both the exact position and momentum of a particle at the same time.
The Properties of Light 🌈
Before we continue our exploration of electrons, we have to understand the properties of light. Light has many properties in common with electrons, such as its wave-particle duality. Electrons and light exist as both a particle and a wave simultaneously. An electron's wave-like properties are basically the reason for Heisenberg's uncertainty principle.
Light as Particle
Let's focus on light as a particle, otherwise known as a photon! Albert Einstein proposed that light is made up of photons, each of which has a specific energy.
Frequency of Light ⚡
Frequency (v) represents the number of waves that pass a point in space in one second. You can think about the frequency of light as the number of times a slinky goes back and forth in a second.
The frequency of light is also related to its energy in a direct relationship. Light with a higher frequency has more energy than light with a lower frequency.
The Photoelectric Effect
The photoelectric effect demonstrates that when a photon of sufficient energy hits a metal surface, it can emit an electron. In other words, electrons are emitted from a metal surface when light strikes it if its frequency is high enough.
Basically, the photoelectric effect only occurs if the frequency of light reaches a certain threshold:
- If the frequency is low, the metal absorbs the light. It is not high enough to reach the threshold and therefore does not exhibit the photoelectric effect.
- If the frequency is high enough and reaches the threshold, a specified number of electrons are ejected from the metal.

We'll go over this concept in greater depth in unit three of the AP Chemistry curriculum.
Photoelectron Spectroscopy (PES)
Photoelectron spectroscopy (PES) is a technique used to compare the relative energies of atoms, ions, and molecules. PES uses energy from electrons emitted through the photoelectric effect to provide insight into the electronic configuration of a sample.
How does PES work?
When light of a certain frequency shines upon a sample, a limited number of electrons are emitted. The released energy reflects the energy or energy levels within an atom.
In viewing the photoelectron spectrum of an element, you are also able to:
- Distinguish the different orbital levels in an atom
- Determine the electron configuration of an atom.
Each peak in a photoelectron spectrum represents a different orbital level where electrons can be found.
Here is a diagram putting all of these concepts together, but don't worry, we'll break it down further!
Image Courtesy of ChemdxInterpreting a Photoelectron Spectrum
Here is a photoelectron spectrum of carbon without the markups which is how the AP exam would give you a graph like this. Let's dissect it.
Image Courtesy of ChemmybearThe Axes of a PES
First, let's look at the axes. When looking at a graph for the first time, the first thing you want to look at is what the graph is representing.
The x-axis is the binding energy, which is used very similarly to the term "ionization energy" on the AP Chemistry exam. We'll go over ionization energy in the next study guide, but for now, think of it as the amount of energy required to remove an electron from an atom.
The closer an electron is to the nucleus of an atom, the more energy will be required to remove it. Therefore, the binding energy, or ionization energy, will be higher as well. Think about this with regard to the fact that positive charges attract negative charges. The super-positive nucleus is going to be strongly attracted to the negative electrons around it. Electrons that are closer to the nucleus are thus more strongly attracted to it.
This information should be able to tell us which side the nucleus is on in this diagram. With this photoelectron spectrum of carbon, it is on the left side because the binding energy is greatest there (1000>0.1). So let's read the diagram from left to right.
The y-axis of this graph simply tells you how many electrons there are in each peak.
The Peaks on a PES
Since peaks represent an orbital where electrons can be found, the first orbital (closest to the nucleus) must be 1s. Because the graph goes up to 2 on the y-axis, there are 2 electrons in the 1s orbital. This should make sense because there is a maximum of two electrons in the s suborbital.
The next orbital has to be 2s and the PES indicates there are 2 electrons in this orbital as well.
So far, the electron configuration seems to be 1s^2 2s^2.
There is one more peak though, which corresponds to the 2p orbital. However, this orbital isn't filled to maximum capacity, there are only 2 electrons in it.
The full electron configuration of this element is 1s^2 2s^2 2p^2. If I didn't tell you that this PES was for carbon, you should be able to guess it given the graph and the periodic table.
Big Ideas with Photoelectron Spectroscopy
When breaking down a PES,
- The position of the peak indicates how much energy is required to remove an electron from that sublevel.
- The height of the peak indicates how many electrons occupy that sublevel.
Practice MCQs
The next two questions are adapted from a Quizizz that kkehesci created.
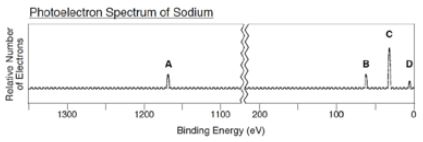
- Refer to the photoelectron spectrum of neon shown below to answer the following question. Which of the following statements best accounts for peak A being to the left of peaks B and C?
a. The electron configuration of neon is 1s^2 2s^2 2p^6.
b. Neon has 8 electrons located in its valence shell.
c. Core electrons of an atom experience a much greater attraction to the nucleus than valence electrons.
d. Peaks B and C show 1st ionization energies (I.E.) in neon, whereas peak A shows the 2nd I.E. of Neon.
2. Which peak shows electrons closest to the nucleus? A, B, C, or D?
Answers to MCQs
- The answer to #1 is C: core electrons of an atom experience a much greater attraction to the nucleus than valence electrons. This goes back to the concept that electrons closest to the nucleus have a higher ionization energy/binding energy.
- The answer to #2 is peak A. This goes back to that same exact concept. If asked about which peak corresponds to the energies of the valence electrons, you should say peak D (since they are furthest from the nucleus).
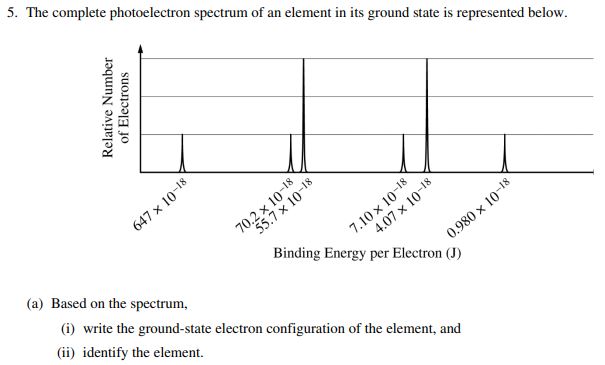
AP Practice Question - 2019 #5
This question is taken from the AP Chemistry Exam in 2019.
In part a, they are asking you to simply write the electron configuration and identify the element. We just did this a few times! If you feel comfortable with this, try it on your own first before looking at the answer.
Since the binding energy is the largest on the left, the peak on the left is the 1s orbital. The electron configuration is:
You could use the noble gas shortcut here as well, but I often leave the electron configuration like this when given a PES. You may make a mistake when trying to write out the noble gas shortcut with a photoelectron spectrum.
To identify the element, just pull out your periodic table! You should get Ca.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
electron configuration | The arrangement of electrons in an atom or ion, describing which orbitals and subshells are occupied and how many electrons are in each. |
ionization energy | The energy required to remove an electron from an atom in the gas phase. |
photoelectron spectroscopy | An experimental technique that measures the energy required to remove electrons from different subshells of an atom or ion. |
photoelectron spectrum | A graphical representation of data from photoelectron spectroscopy showing peaks that correspond to electrons in different subshells of an atom or ion. |
subshell | A subdivision of an electron shell characterized by a specific orbital type (s, p, d, or f) and containing orbitals of similar energy. |
Frequently Asked Questions
What is photoelectron spectroscopy and how does it work?
PES (photoelectron spectroscopy) measures how tightly electrons are held in atoms/ions by knocking them out with photons and measuring the ejected electrons’ energies. A photon with known energy (hν) hits the atom; an electron absorbs that energy and is ejected. The kinetic energy (KE) of the ejected electron is measured, and binding energy (BE) is found from BE = hν − KE. Peaks on a PES plot correspond to electrons from specific subshells; peak position = binding energy (higher BE → electrons closer to nucleus), peak height (ideally) ∝ number of electrons in that subshell. Interpretations use concepts like effective nuclear charge, shielding, and Koopmans’ theorem; spin–orbit splitting can split peaks for certain subshells. On the AP exam you’ll be asked to connect peak positions/heights to electron configuration and electron–nucleus interactions (CED Topic 1.6). For a focused review, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA). Practice problems are at (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to learn about photoelectron spectroscopy in AP Chemistry?
You need to learn photoelectron spectroscopy (PES) because it’s an experimental way to connect the electron configuration model to real data—exactly what the CED expects in Topic 1.6 (binding energy, subshell peaks, core vs valence electrons, effective nuclear charge, shielding). PES shows peaks whose positions equal the energy needed to remove electrons from specific subshells (so you can identify 1s, 2s, 2p, etc.) and peak heights relate to electron counts. That lets you justify statements about electron configuration and nucleus–electron interactions on the exam (Unit 1: 7–9% of the test; PES appears in short free-response questions like the sample FRQ). Practice reading spectra to explain why inner electrons have higher binding energy (Coulomb’s law) and how Zeff and shielding shift peaks. For a focused study and practice problems, check the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and thousands of practice questions at (https://library.fiveable.me/practice/ap-chemistry). If you want, I can walk through a sample PES and map peaks to the electron configuration.
How do you read a PES spectrum and what do the peaks mean?
Read a PES by treating each peak as electrons from a specific subshell: the x-axis is binding energy (how strongly electrons are held; often in eV or kJ·mol⁻¹) and the y-axis shows relative intensity (peak height ≈ number of electrons in that subshell). Peaks at higher binding energy are from electrons closer to the nucleus (core electrons) because Coulomb’s law means greater attraction → larger ionization energy. Valence peaks are at lower binding energy (easier to remove). Peak splitting or small doublets can show spin–orbit effects. Use Koopmans’ theorem: the peak position approximates the ionization energy of that orbital. Compare relative positions to expected subshell energies (1s < 2s/2p, etc.) and peak heights to deduce electron configuration and effective nuclear charge/shielding effects. On the AP exam you should be able to identify which peaks are core vs valence and justify shifts using shielding/Zeff (CED 1.6.A). For a focused review, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA). For broader unit review or extra practice, check Unit 1 (https://library.fiveable.me/ap-chemistry/unit-1) and AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between the height and position of peaks in photoelectron spectroscopy?
Position (horizontal) of a PES peak tells you the binding energy (how much energy is needed to remove an electron)—peaks farther left (higher binding energy) are from electrons held more tightly (closer to the nucleus, inner/core orbitals). That links to effective nuclear charge, shielding, and Koopmans’ idea that peak position approximates subshell ionization energy. Height (vertical) is (ideally) proportional to the number of electrons in that subshell—a taller peak means more electrons contributed. Small caveats: spin–orbit splitting can split a subshell into two nearby peaks, and experimental intensities can be affected by cross-sections, so heights aren’t always exact counts. For AP-style questions, use peak position to identify which orbital (core vs valence) and peak height to infer electron count or compare subshell occupancies. For a focused review, see the Topic 1.6 PES study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and extra practice (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about how PES relates to electron configuration - can someone explain?
Think of a PES spectrum as a photo of where electrons live and how tightly they’re held. Each peak’s position (binding energy) tells you how much energy it takes to remove electrons from a specific subshell: peaks farther left (higher binding energy) are electrons closer to the nucleus (core electrons), peaks to the right are less tightly bound valence electrons. Peak height (ideally) matches the number of electrons in that subshell—so a 2-electron 1s gives a taller peak than a 1-electron 3s. Differences in binding energy come from effective nuclear charge and shielding (more Zeff → higher binding energy). Koopmans’ theorem connects measured binding energies to orbital ionization energies. For the AP exam you should be able to use a PES diagram to write an element’s electron configuration and explain nucleus–electron interactions (CED 1.6.A). For extra practice and a clear walkthrough, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do electrons in different subshells require different amounts of energy to remove?
Different subshells need different energy to remove an electron because of how strongly the nucleus pulls on those electrons (effective nuclear charge, Zeff) and how well other electrons shield that pull. Electrons in s orbitals penetrate closer to the nucleus and feel a larger Zeff, so they have higher binding (ionization) energy than p, d, or f electrons in the same shell. Electrons farther from the nucleus or behind more shielding electrons are easier to remove. Electron–electron repulsion and pairing can also raise energy for an electron (making it easier to eject) in a filled subshell. Photoelectron spectroscopy (PES) measures those differences: peak position = binding energy (how hard to remove), peak height ≈ number of electrons in that subshell (CED 1.6.A, Koopmans’ idea). For a focused PES review and practice problems, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and the Unit 1 overview (https://library.fiveable.me/ap-chemistry/unit-1).
How does the number of electrons in a subshell affect the height of peaks in PES?
Ideally, the height (intensity) of a PES peak is proportional to how many electrons come from that subshell—so a full 1s or 2s peak (2 electrons) will be shorter than a full 2p peak (6 electrons). In practice: s = 2, p = 6, d = 10, f = 14—more electrons → taller peak (CED 1.6.A.1). Keep one caveat in mind for the AP exam: peak height is a primary clue to electron count, but experimental factors can change heights (photoionization cross-sections, instrument sensitivity, overlapping peaks, spin–orbit splitting). Position (binding energy) tells you how strongly electrons are held (closer to nucleus → higher binding energy). Use both height and position together to deduce electron configuration (Topic 1.6). Want more practice? Review the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens when you shine light on an atom during photoelectron spectroscopy?
When you shine high-energy light (UV or X-rays) on an atom in PES, photons transfer energy to electrons. If a photon’s energy exceeds an electron’s binding energy (ionization energy for that orbital), the electron is ejected; its measured kinetic energy lets you calculate the binding energy: EB = hν − KE. In a PES spectrum each peak’s position (binding energy) corresponds to a particular subshell (1s, 2s, 2p, etc.), and peak height is (ideally) proportional to the number of electrons in that subshell. Core electrons appear at much higher binding energy than valence electrons because they’re closer to the nucleus (higher effective nuclear charge, less shielding). PES therefore reveals electron configuration and electron–nucleus interactions (useful with Koopmans’ theorem and spin–orbit splitting). This is exactly what AP Topic 1.6 tests: connect peaks to subshells and explain why core vs valence energies differ. For a focused review, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can you use PES data to figure out the electron configuration of an unknown element?
Yes—PES can often let you determine an atom’s electron configuration, but with some limits. Peaks on a PES plot map to subshells: their position (binding energy) shows how tightly electrons are held (inner/core electrons have larger binding energies), and peak height is (ideally) proportional to the number of electrons in that subshell. Using that info plus known subshell ordering and Hund’s/Pauli rules you can assign electrons and write a configuration (this is exactly what LO 1.6.A expects). Caveats: peaks can overlap or show spin–orbit splitting, intensities aren’t always perfectly proportional, and shifts occur from effective nuclear charge and electron shielding (Koopmans’ theorem is a helpful approximation). So PES gives strong experimental evidence for subshell energies and electron counts, but you may need periodic-table context or complementary data to be certain. For AP-style practice and worked examples, check the Photoelectron Spectroscopy study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do 1s electrons require more energy to remove than 2s electrons?
1s electrons need more energy to remove than 2s electrons because they experience a stronger attraction to the nucleus. Two facts explain this: (1) 1s orbitals are closer to the nucleus (greater penetration), so by Coulomb’s law the electron–nucleus attraction is larger; (2) 1s electrons are less shielded by other electrons, so the effective nuclear charge (Zeff) they feel is higher than for 2s electrons. In PES terms, higher binding energy (peak farther left/higher BE) corresponds to a more tightly bound electron—that’s why the 1s peak appears at larger binding energy than the 2s peak. This idea ties directly to the CED keywords: binding energy, effective nuclear charge, and electron shielding (Topic 1.6). For a quick refresher and PES practice, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and more AP practice questions (https://library.fiveable.me/practice/ap-chemistry).
How do you interpret PES spectra for ions versus neutral atoms?
Short answer: compare peak positions (binding energy) and heights (electron count). For an ion vs neutral, the same subshell peaks appear but their binding energies shift because of effective nuclear charge and shielding. A cation (lost electrons) has a higher effective nuclear charge on the remaining electrons, so all peaks shift to higher binding energy (left on many PES plots if x-axis is BE increasing to left)—core and valence both are held more tightly. An anion shows lower binding energies. Peak heights still reflect number of electrons in each subshell (ideally). Remember final-state effects and Koopmans’ theorem: PES gives approximate removal energies from the initial orbital, but electron correlation/relaxation can tweak peak positions. On the AP exam, justify shifts by Coulomb’s law / Zeff (CED 1.6.A.1). For a quick review, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and more unit practice at (https://library.fiveable.me/ap-chemistry/unit-1) or practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between binding energy and the distance of electrons from the nucleus?
Short answer: electrons closer to the nucleus have larger binding energies. PES peak position corresponds to the energy needed to remove an electron from a subshell: core electrons (small r, less shielded, higher effective nuclear charge) show peaks at higher binding energy, while valence electrons (larger r, more shielding) appear at lower binding energy. Use Coulomb’s law: attraction ∝ (charge product)/r, so smaller r → stronger attraction → more energy required to remove the electron. Factors that shift binding energy: effective nuclear charge (Zeff) raises BE, electron shielding lowers BE, and spin–orbit or relaxation effects can split/shift peaks (Koopmans’ theorem gives a first approximation). On the AP exam expect to link PES peak positions to electron configuration and nucleus–electron interactions (CED 1.6.A). For a topic review and PES practice, see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why PES peaks get shorter as you move to higher energy levels - help?
Short answer: peak height in a PES is (ideally) proportional to how many electrons come from that subshell, and peak position tells you the binding energy. So peaks usually get shorter at higher (less tightly bound) energy levels because there are fewer electrons in those subshells and those electrons give smaller signals. Why that makes sense: - Inner (core) subshells often contain more electrons (e.g., 2s/2p vs a single 3s), so their peaks are taller. (CED keyword: height ∝ number of electrons.) - Valence electrons are farther from the nucleus, more shielded, and have lower binding energy (easier to remove), so their peaks appear at lower binding energy and are often smaller. - Experimental effects (ionization cross-sections) can also reduce peak height for higher-energy/valence electrons, but the primary AP-level reason is electron count + effective nuclear charge/shielding. For more on interpreting PES (positions = binding energy; heights ≈ electron count) see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA). Practice with spectra on Fiveable’s practice problems (https://library.fiveable.me/practice/ap-chemistry) to get comfortable.
How does photoelectron spectroscopy prove that electrons exist in specific energy levels?
PES proves electrons occupy specific energy levels because when atoms are hit with photons, electrons are ejected with specific kinetic energies, so the spectrum shows distinct peaks at particular binding energies. Each peak corresponds to electrons from a particular subshell (1s, 2s, 2p, etc.); the position (binding energy) tells you how strongly those electrons were held (core electrons have larger binding energy because of greater Coulomb attraction / higher effective nuclear charge), and the peak height (ideally) reflects how many electrons were in that subshell. That one-to-one mapping between peaks and subshells lets you read electron configuration and infer electron–nucleus interactions (Koopmans’ theorem connects measured binding energy to ionization energy). In short: discrete peaks = discrete energy levels. For more examples and AP-style practice, see the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and hundreds of practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why would scientists use PES instead of just looking up electron configurations?
Because electron configurations are a model—useful, but idealized. PES gives experimental binding energies for electrons in each subshell, so you can measure things the simple configuration doesn’t show: actual ionization energies, differences from the "Aufbau" picture caused by electron–electron interactions, effective nuclear charge, shielding, spin–orbit splitting, and relaxation effects (Koopmans’ theorem is an approximation). Peaks in a PES tell you which subshells are core vs valence and how many electrons are in each (peak positions = binding energy; heights ≈ electron count). That matters when theory and reality disagree (transition metals, heavy elements, or chemical environments). For AP Topic 1.6 you should be able to connect peak position/height to subshells and nucleus–electron interactions (LO 1.6.A). If you want practice reading spectra, check the Topic 1.6 study guide (https://library.fiveable.me/ap-chemistry/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).