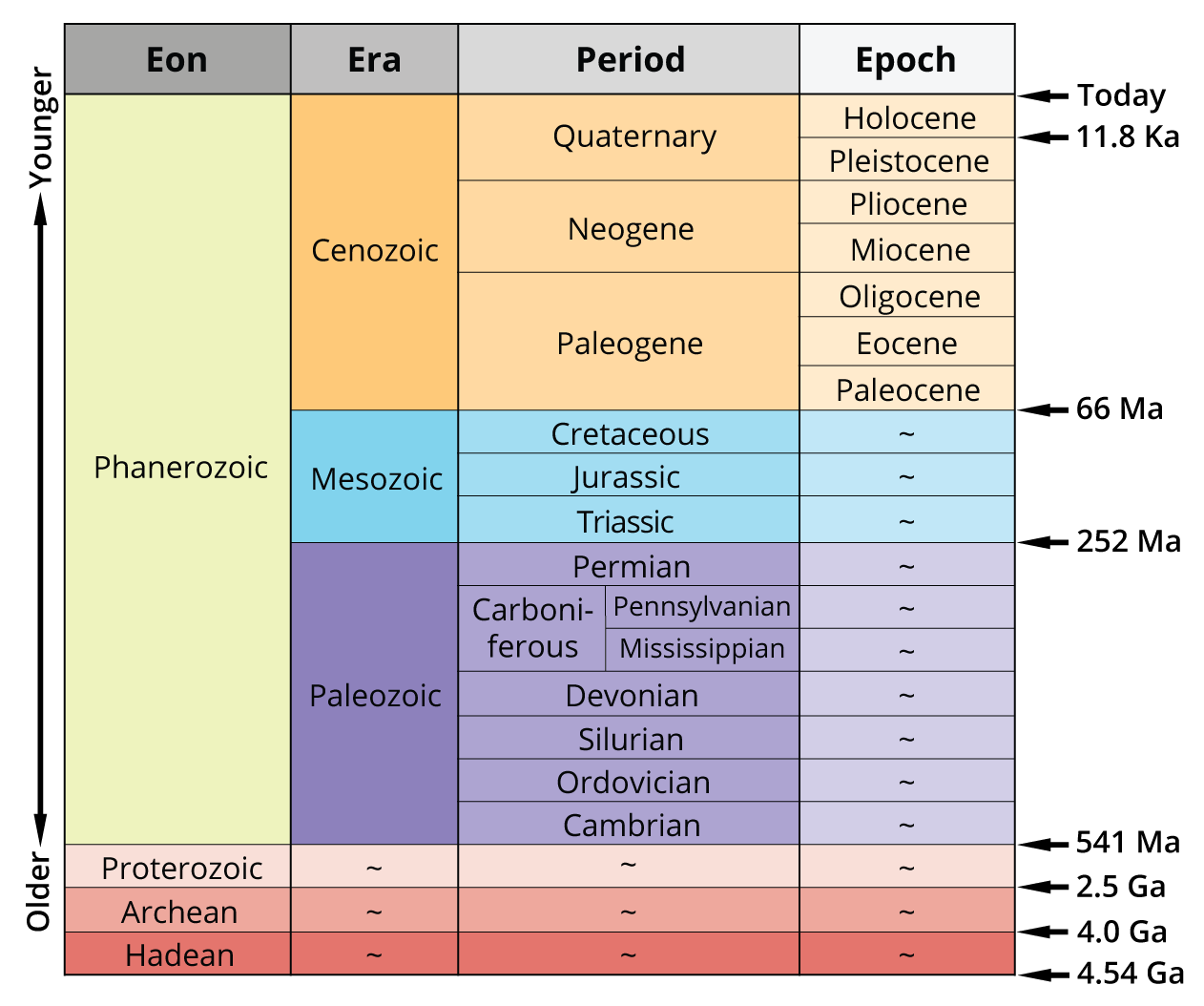
Relative and Absolute Dating Methods
Geologists use two main approaches to unravel Earth's history: relative dating and absolute dating. Relative dating puts events in order (which happened first, second, third), while absolute dating assigns specific numerical ages to rocks and events. Together, these methods let geologists construct the timeline of Earth's 4.6-billion-year history.
Relative vs. Absolute Dating Methods
Relative dating determines the order of geologic events without providing specific numerical ages. If you know that Rock Layer A formed before Rock Layer B, that's relative dating. The tools include superposition, cross-cutting relationships, and index fossils.
Absolute dating determines the actual numerical age of a rock, mineral, or fossil, expressed in years or millions of years. Radiometric dating is the most common technique, and it relies on the predictable decay of radioactive isotopes. Methods like potassium-argon (K-Ar), uranium-lead (U-Pb), and carbon-14 dating each work best for different age ranges.
The two approaches complement each other. Relative dating establishes the sequence, and absolute dating pins specific ages onto that sequence.

Principles of Relative Dating
These are the foundational rules geologists use to determine the relative order of rock layers and geologic events:
- Principle of Superposition: In an undeformed sequence of sedimentary rocks, the oldest layers are at the bottom and the youngest are at the top. Think of it like a stack of papers on a desk: the first one you set down is on the bottom.
- Principle of Original Horizontality: Sedimentary layers are originally deposited in a nearly horizontal position. If you find tilted or folded layers, something deformed them after they were deposited.
- Principle of Cross-Cutting Relationships: Any geologic feature that cuts across another feature must be younger than the feature it cuts. A fault slicing through a rock layer, for example, formed after that layer. The same applies to igneous intrusions and erosional surfaces.
- Principle of Inclusions: If fragments of one rock are found enclosed within another rock, those fragments (inclusions) must be older than the rock surrounding them. The fragments had to already exist before the host rock formed around them.
Radiometric Dating for Absolute Ages
Radiometric dating works because certain isotopes have unstable nuclei that decay into different elements at a constant, measurable rate. The rate of decay is described by the half-life: the time it takes for half of the parent isotope atoms in a sample to decay into daughter product atoms.
For example, if you start with 1,000 atoms of a parent isotope and its half-life is 1 million years, after 1 million years you'll have 500 parent atoms and 500 daughter atoms. After another million years, you'll have 250 parent atoms and 750 daughter atoms.
To calculate the age of a sample, geologists measure the ratio of parent isotope to daughter product in a mineral and apply the decay equation:
Where:
- = age of the sample
- = decay constant (related to the half-life by )
- = number of daughter isotope atoms measured in the sample
- = number of parent isotope atoms remaining in the sample
For an intro course, the key concept is the half-life relationship, not the equation itself. Focus on understanding that more daughter product relative to parent isotope means an older sample.
Common radiometric methods and their uses:
- Potassium-Argon (K-Ar): Used for volcanic rocks older than about 100,000 years. Potassium-40 decays to argon-40 with a half-life of ~1.25 billion years.
- Uranium-Lead (U-Pb): Used for very old rocks (older than ~1 million years). Uranium-238 decays to lead-206 with a half-life of ~4.47 billion years, making it ideal for dating Earth's oldest minerals.
- Carbon-14 (Radiocarbon): Used for organic materials (wood, bone, shell) less than about 50,000 years old. Carbon-14 has a half-life of only ~5,730 years, so it decays too quickly to be useful for anything older.
Limitations of Dating Methods
No dating method is perfect. Understanding the limitations helps you evaluate how reliable a given age estimate is.
Relative dating limitations:
- It cannot tell you how old something is, only the order in which events occurred
- It cannot determine the duration of time between events
Radiometric dating relies on three key assumptions:
- The decay rate has remained constant over time (well-supported by physics)
- The system has remained closed, meaning no parent or daughter isotopes have been added or removed since the rock formed
- The initial amount of daughter product can be known or estimated
Practical limitations of radiometric dating:
- The rock or fossil must contain suitable radioactive minerals or organic material. Not every rock can be dated this way.
- The sample must not have been chemically altered or contaminated after formation, since that would change the parent-to-daughter ratio.
- The half-life of the chosen isotope must match the age range you're measuring. Using carbon-14 on a billion-year-old rock won't work because all the carbon-14 would have decayed long ago.