Thermodynamic principles of colloid stability
Thermodynamic stability determines whether colloidal particles stay dispersed in a medium or clump together over time. This topic ties together forces, energy, and environmental factors to explain why some colloidal systems last for years while others aggregate in minutes.
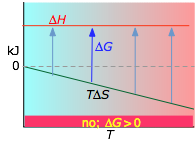
Gibbs free energy in colloidal systems
Gibbs free energy () tells you whether aggregation is thermodynamically favorable. The relationship is:
A colloidal dispersion is thermodynamically stable when for aggregation is positive, meaning the system "prefers" to stay dispersed. A negative for aggregation means the dispersed state is thermodynamically unfavorable, and particles will tend to clump together if they can overcome any kinetic barriers.
Be careful with the language here: a thermodynamically stable colloid genuinely sits at a free energy minimum in the dispersed state. Most colloids, though, are only kinetically stable, meaning aggregation is thermodynamically favorable but an energy barrier slows it down enough that the system appears stable on practical timescales.
Entropy and enthalpy contributions
Entropy () and enthalpy () compete to determine :
- Entropy favors dispersion. Spreading particles throughout a medium increases the number of accessible microstates, raising disorder. This effect is more significant for smaller particles because there are more of them per unit mass.
- Enthalpy reflects the net energy of particle-particle and particle-solvent interactions. Favorable particle-solvent interactions (negative of mixing) promote stability, while strong particle-particle attraction works against it.
The balance shifts with temperature. Since , higher temperatures amplify the entropy term. That's why some marginally stable systems become more stable when warmed (entropy-driven stabilization) while others destabilize (enthalpy-driven aggregation becomes dominant).
Interactions between colloidal particles
Colloidal stability comes down to the balance of attractive and repulsive forces acting between particles. If repulsion wins, particles stay apart. If attraction wins, they aggregate.
Van der Waals forces
Van der Waals forces are the primary attractive interaction between colloidal particles. They originate from fluctuating electron clouds that create temporary dipoles, which then induce dipoles in neighboring particles.
Key characteristics:
- They are always present between any two particles, regardless of surface charge or chemistry.
- Their strength depends on the Hamaker constant, which is determined by the material properties of the particles and the medium. Larger Hamaker constants mean stronger attraction.
- For two identical spheres of radius separated by distance , the van der Waals attraction energy is approximately (where is the Hamaker constant). This means attraction increases with particle size and grows rapidly at short separations.
- They are relatively weak at long range but dominate at short distances, which is why particles that get too close tend to stick.
Electrostatic repulsion
Most colloidal particles acquire surface charge in solution (through ionization of surface groups, ion adsorption, or crystal lattice substitutions). Particles with like charges repel each other.
- The repulsion arises from the overlap of electrical double layers surrounding each particle.
- Its magnitude depends on the surface charge density and the ionic strength of the medium. Higher surface charge means stronger repulsion; higher ionic strength compresses the double layer and weakens it.
- Electrostatic repulsion decays roughly exponentially with distance, controlled by the Debye length (more on this below).
Steric stabilization
Steric stabilization uses adsorbed or grafted polymers and surfactants to physically prevent particles from getting close enough for van der Waals attraction to take over.
- When two polymer-coated particles approach, the adsorbed layers overlap and compress. This compression restricts the polymer chains' conformational freedom (entropy loss) and increases local polymer concentration (osmotic penalty), both of which are energetically unfavorable.
- Effectiveness depends on the thickness and grafting density of the adsorbed layer, as well as the solvent quality. In a good solvent, the polymer chains are well-extended and provide strong repulsion. In a poor solvent, the chains collapse and steric stabilization weakens.
- Steric stabilization works in both polar and nonpolar media, making it more versatile than electrostatic repulsion alone.
Depletion forces
Depletion forces are an attractive interaction that arises when free (non-adsorbing) polymers or small particles are present in solution.
- When two colloidal particles approach within a distance smaller than the diameter of the free polymer coils, those polymers get excluded from the gap between the particles.
- This exclusion creates a region of pure solvent between the particles, while the surrounding solution still contains polymer. The resulting osmotic pressure imbalance pushes the particles together.
- The strength of depletion attraction increases with the concentration and size of the non-adsorbing species.
- Depletion forces can be useful (e.g., controlled phase separation) or problematic (unwanted aggregation), depending on the application.
DLVO theory
DLVO theory (named after Derjaguin, Landau, Verwey, and Overbeek) is the foundational quantitative model for colloidal stability. It predicts the total interaction energy between two particles by summing van der Waals attraction and electrostatic repulsion:
The shape of this total energy curve as a function of particle separation distance determines whether a system is stable, unstable, or somewhere in between.
Electric double layer
When a charged particle sits in an electrolyte solution, ions redistribute around it to form the electric double layer:
- Stern layer — a thin layer of counterions tightly bound to the particle surface. These ions are essentially immobile.
- Diffuse layer — a region extending outward where counterions are loosely associated and co-ions are partially excluded. Ion concentrations gradually approach bulk values.
The characteristic thickness of the diffuse layer is the Debye length (), defined as:
where is the ionic strength. In practical terms: higher ionic strength means a shorter Debye length and a more compressed double layer. For example, in pure water can be hundreds of nanometers, but in 0.1 M NaCl it shrinks to about 1 nm.
Zeta potential
Zeta potential () is the electric potential measured at the slipping plane, the boundary between the fluid that moves with the particle and the fluid that doesn't.
- It's not the same as the surface potential, but it's the most experimentally accessible measure of surface charge.
- Measured using techniques like electrophoretic light scattering (the particle moves in an applied electric field, and its velocity reveals ).
- As a rule of thumb, suggests good electrostatic stability, while indicates the system is prone to aggregation.
- Zeta potential changes with pH (surface groups ionize differently), ionic strength (double layer compression), and adsorbed species (surfactants or polymers can shift the slipping plane).

Energy barriers and minima
The DLVO interaction energy curve typically shows three important features:
- Primary minimum — a deep energy well at very short separation distances where van der Waals attraction dominates. Particles trapped here form strong, usually irreversible aggregates.
- Energy barrier — a peak at intermediate distances where electrostatic repulsion is strongest. The height of this barrier determines kinetic stability. If the barrier is much larger than the thermal energy (), particles rarely have enough energy to cross it, and the system stays dispersed.
- Secondary minimum — a shallow energy well at larger separations. Particles can sit here in loose, reversible aggregates (flocs) that are easily redispersed by mild agitation.
The practical takeaway: even if the primary minimum is very deep (thermodynamically favoring aggregation), a tall energy barrier can keep the system kinetically stable for long periods.
Limitations of DLVO theory
DLVO theory is powerful but has notable blind spots:
- It assumes smooth, spherical particles. Real particles have surface roughness, irregular shapes, and chemical heterogeneity.
- It ignores non-DLVO forces such as hydrophobic interactions, hydrogen bonding, hydration forces, and steric effects.
- It breaks down at high ionic strengths (above ~0.1 M for monovalent ions) and performs poorly with multivalent ions, which can cause charge reversal and other effects the theory doesn't predict.
- It treats the solvent as a continuous dielectric medium, ignoring molecular-scale solvent structure near surfaces.
Despite these limitations, DLVO theory remains the starting point for understanding and predicting colloidal stability. Extended DLVO (XDLVO) models incorporate additional forces to address some of these gaps.
Colloidal stability in different media
The dispersing medium isn't just a passive background. Its properties directly control the strength and range of interparticle interactions.
Effect of pH on stability
pH controls the ionization state of surface groups, which determines the surface charge.
- At the isoelectric point (IEP), the net surface charge is zero. Electrostatic repulsion vanishes, and particles aggregate readily. For example, silica has an IEP around pH 2, while alumina's is near pH 9.
- Moving the pH away from the IEP (in either direction) increases the magnitude of the surface charge and strengthens electrostatic repulsion.
- The optimal pH for stability depends on the specific surface chemistry. For oxide particles, this means working well above or below the IEP.
Ionic strength and Debye length
Ionic strength () quantifies the total concentration of ions in solution and directly controls the Debye length.
- Low ionic strength → long Debye length → extended double layer → strong, long-range electrostatic repulsion → better stability.
- High ionic strength → short Debye length → compressed double layer → weakened repulsion → easier aggregation.
This is why adding salt to a charge-stabilized colloid often causes flocculation. The critical coagulation concentration (CCC) is the minimum electrolyte concentration needed to eliminate the energy barrier entirely. According to the Schulze-Hardy rule, the CCC drops dramatically with increasing ion valence: divalent ions are roughly 100× more effective at destabilizing than monovalent ones, and trivalent ions are even more potent.
Stability in polar vs. nonpolar solvents
The stabilization strategy you choose depends heavily on the solvent:
- Polar solvents (especially water): Electrostatic stabilization works well because the high dielectric constant supports charge separation and double layer formation. Hydrogen bonding and hydration forces also contribute.
- Nonpolar solvents (oils, hydrocarbons): The low dielectric constant means charges are poorly screened and double layers barely form. Electrostatic repulsion is largely ineffective. Steric stabilization using compatible polymers or surfactants is the primary strategy here.
- Some systems use electrosteric stabilization, combining both charged and polymeric surface layers, which can work across a range of solvent polarities.
Experimental techniques for stability assessment
No single measurement tells the whole story. Combining several techniques gives a much more complete picture of colloidal stability.
Zeta potential measurements
Zeta potential is typically measured by electrophoretic light scattering (ELS), also called laser Doppler electrophoresis. An electric field is applied across the sample, particles migrate at a rate proportional to their zeta potential, and scattered laser light detects their velocity.
- : generally good stability
- between 15 and 30 mV: moderate stability
- : expect aggregation
Tracking zeta potential as you vary pH or ionic strength produces a stability map for your system.
Dynamic light scattering
Dynamic light scattering (DLS) measures particle size by analyzing fluctuations in scattered light intensity caused by Brownian motion.
- Smaller particles diffuse faster, producing more rapid intensity fluctuations. The diffusion coefficient is converted to a hydrodynamic diameter using the Stokes-Einstein equation.
- DLS also reports the polydispersity index (PDI), which indicates how broad the size distribution is.
- Monitoring particle size over time is a straightforward stability test: if the average diameter grows, aggregation is occurring.
- DLS is quick, non-invasive, and requires very small sample volumes, but it's most reliable for dilute, relatively monodisperse samples.
Sedimentation and creaming rates
Gravity-driven separation reveals stability on practical timescales:
- Sedimentation occurs when particles are denser than the medium (they sink). Creaming occurs when particles are less dense (they rise, as in emulsions).
- Stable systems show very slow separation. Unstable systems separate visibly within hours or days.
- Analytical centrifugation (e.g., LUMiSizer) accelerates the process, allowing you to predict long-term stability from short experiments. It measures transmission profiles across the sample as a function of time and centrifugal force.
- Stokes' law relates sedimentation velocity to particle size, density difference, and medium viscosity:
Rheological behavior of colloids
Rheology probes how a colloidal system flows and deforms, which reflects its microstructure and stability.
- Stable, well-dispersed colloids tend to show Newtonian behavior (constant viscosity) at low concentrations, or shear-thinning behavior at higher concentrations.
- Aggregated or flocculated systems often display a yield stress (you need a minimum force to make them flow) and may show shear-thickening behavior.
- Oscillatory rheometry measures the storage modulus (, elastic response) and loss modulus (, viscous response). A system where behaves more like a gel, which can indicate a network of aggregated particles.
- Tracking rheological properties over time or after stress can reveal whether a system is stable or slowly aggregating.
Strategies for enhancing colloidal stability
Choosing the right stabilization strategy depends on the particles, the medium, and the application requirements.
Surface modification of particles
Changing the particle surface directly alters how particles interact with each other and the medium:
- Charge modification: Grafting ionizable groups onto the surface increases electrostatic repulsion.
- Polymer grafting: Covalently attaching polymer chains (e.g., PEG) provides steric stabilization that's more robust than physical adsorption because the chains can't desorb.
- Inorganic coatings: Depositing a shell of silica or alumina can change the Hamaker constant, shift the IEP, or improve compatibility with a particular solvent.
- Surface modification is often more permanent than adding free stabilizers, making it preferred for long-term applications.
Addition of stabilizing agents
Stabilizing agents adsorb onto particle surfaces and provide repulsive forces:
- Surfactants adsorb at the particle-medium interface. Ionic surfactants add both steric and electrostatic stabilization. Nonionic surfactants provide purely steric stabilization and are less sensitive to ionic strength.
- Polymers (e.g., polyvinyl alcohol, polyethylene glycol) create thick steric barriers. Their effectiveness depends on molecular weight, concentration, and solvent quality.
- Polyelectrolytes combine charge and steric effects (electrosteric stabilization) and are particularly effective in aqueous systems.
- Too much stabilizer can actually cause problems: excess free polymer can induce depletion flocculation, and excess surfactant can form micelles that complicate the system.
Controlling environmental conditions
Sometimes the simplest approach is adjusting the medium:
- pH: Maintain it well away from the IEP. Buffer the system if pH drift is a concern.
- Ionic strength: Keep it low for electrostatically stabilized systems. Use deionized water where possible.
- Temperature: Higher temperatures increase collision frequency (promoting aggregation) but also increase thermal energy (helping particles escape shallow energy minima). The net effect depends on the system. For sterically stabilized systems, there may be a critical temperature above which the solvent quality drops and stability is lost.
Optimizing particle size and distribution
Particle size affects stability through multiple mechanisms:
- Smaller particles experience stronger Brownian motion relative to gravitational settling, so they resist sedimentation better.
- Smaller particles also have a higher surface-area-to-volume ratio, meaning surface interactions (repulsive or attractive) play a larger role relative to body forces.
- A narrow size distribution (low polydispersity) helps because all particles behave similarly. Broad distributions can lead to differential sedimentation rates and Ostwald ripening.
- Size control methods include milling, high-pressure homogenization, ultrasonication, and controlled precipitation.
Destabilization and flocculation
Destabilization is the loss of colloidal stability, leading to aggregation. Flocculation specifically refers to the formation of loose aggregates (flocs). Sometimes you want this (water treatment), and sometimes you don't (paint shelf life).
Mechanisms of flocculation
Flocculation can proceed through several distinct pathways:
- Bridging flocculation: A high-molecular-weight polymer adsorbs onto two or more particles simultaneously, physically linking them. This requires the polymer to be long enough to span the gap and present at the right concentration. Too much polymer saturates the surfaces and re-stabilizes the system (steric stabilization).
- Charge neutralization: Adding oppositely charged ions, polymers, or particles reduces the net surface charge, weakening electrostatic repulsion. Multivalent ions are especially effective (e.g., or salts in water treatment).
- Depletion flocculation: Non-adsorbing polymers create osmotic pressure gradients that push particles together (as described above).
Reversible vs. irreversible flocculation
The distinction maps directly onto the DLVO energy profile:
Reversible flocculation occurs in the secondary minimum. The attractive well is shallow (a few ), so gentle agitation or dilution can redisperse the particles. The aggregates are loose and open-structured.
Irreversible flocculation (also called coagulation) occurs in the primary minimum. Van der Waals attraction at close contact is strong, and particles form dense, permanent aggregates that cannot be redispersed by simple mixing.
In practice, many systems start with reversible flocculation in the secondary minimum, and over time particles slowly transition into the primary minimum as they rearrange within the flocs.
Kinetics of flocculation
The rate of flocculation depends on how often particles collide and how likely those collisions are to result in attachment.
- Rapid (diffusion-limited) flocculation occurs when there is no energy barrier. Every collision leads to attachment. The rate is described by the Smoluchowski equation: where is the medium viscosity. This sets the upper limit for flocculation rate.
- Slow (reaction-limited) flocculation occurs when an energy barrier exists. Only a fraction of collisions have enough energy to overcome the barrier. The ratio of the rapid rate to the actual rate is called the stability ratio (). Higher means better kinetic stability.
- Monitoring flocculation kinetics (e.g., by tracking particle size with DLS over time) provides a direct, practical measure of system stability.
Controlling flocculation in applications
Different applications require different levels of flocculation control:
- Water treatment: Flocculants like alum () or polyacrylamide are added to aggregate suspended particles, which then settle out during sedimentation. Dosage must be optimized because too little flocculant is ineffective and too much can restabilize the particles.
- Mineral processing: Selective flocculation separates valuable minerals from waste (gangue) by exploiting differences in surface chemistry. One mineral is flocculated while the other remains dispersed.
- Food and pharmaceutical products: Controlled, reversible flocculation can give desirable textures (e.g., creamy consistency in sauces), but irreversible aggregation causes product failure. Stability testing under accelerated conditions (elevated temperature, centrifugation) helps predict shelf life.
- Paints and coatings: Particles must remain dispersed during storage but may need to flocculate in a controlled way during application and drying.