Understanding Biogeochemical Cycles
Biogeochemical cycles describe how Earth's essential elements move between living organisms and the physical environment. Understanding these cycles is foundational to biogeochemistry because they explain how elements like carbon, nitrogen, and phosphorus get recycled rather than used up, sustaining life across the planet.
Biogeochemical cycles in ecosystems
A biogeochemical cycle is the continuous movement of an element or compound through the biotic (living) and abiotic (non-living) components of Earth, driven by biological, geological, and chemical processes working together.
These cycles do three critical things for ecosystems:
- Regulate distribution of essential elements across Earth's systems
- Maintain balance between inputs and outputs in different environmental compartments
- Support life by ensuring nutrients are available, used, and recycled
The major cycles you'll encounter throughout this course are the carbon cycle, nitrogen cycle, phosphorus cycle, and water (hydrological) cycle. Each operates on different timescales and through different mechanisms, but they're all interconnected.

Components of biogeochemical cycles
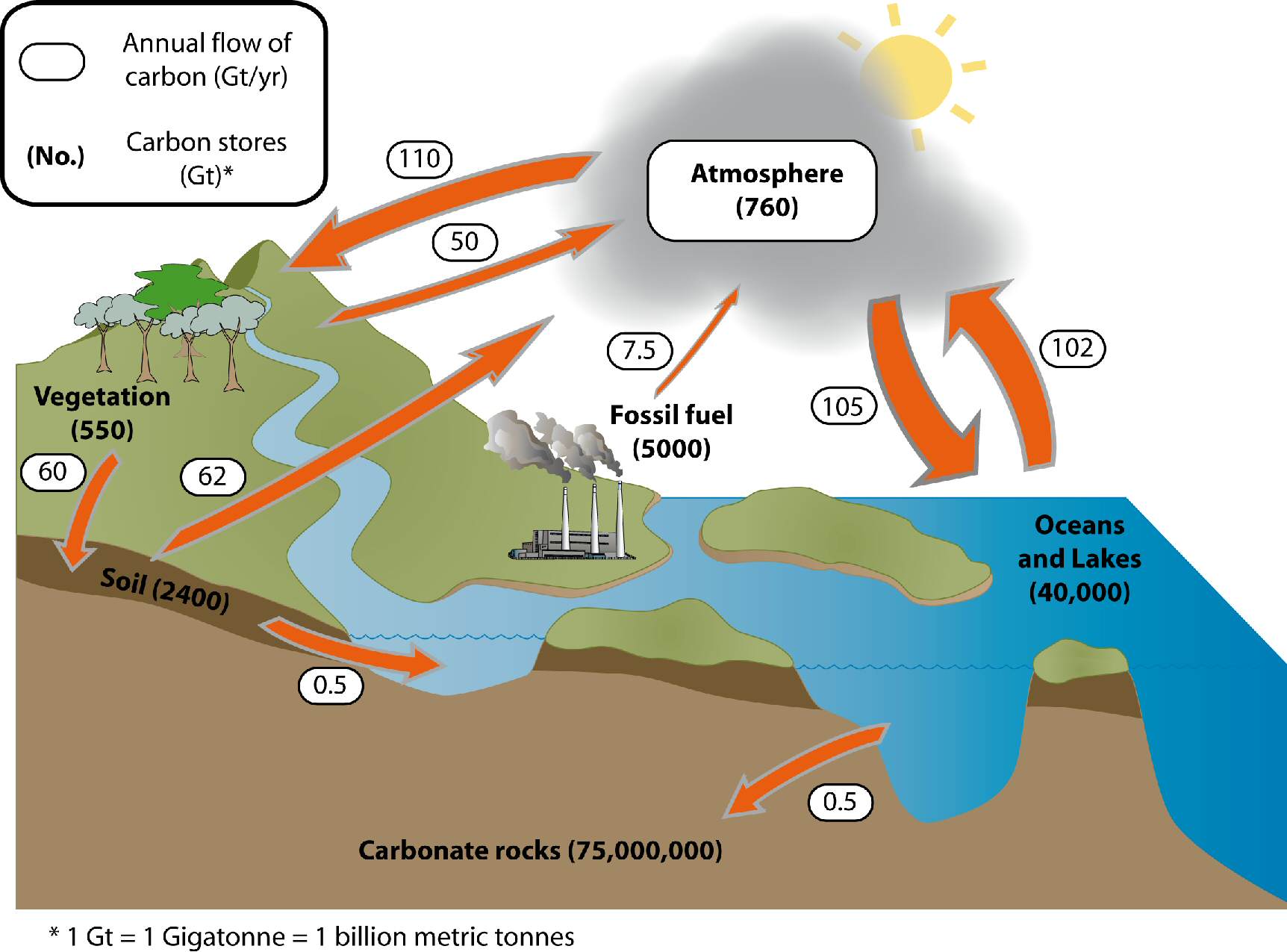
Every cycle can be broken down into the same basic architecture: reservoirs, fluxes, and the processes that drive those fluxes.
Reservoirs (also called pools or stocks) are where elements accumulate and are stored. The four major reservoirs are the atmosphere, oceans, soil/lithosphere, and biomass (living organisms). Each reservoir holds a different quantity of a given element, and the amount stored at any point is called the stock or pool size.
Fluxes are the rates at which elements move between reservoirs, measured in mass per unit time (e.g., or for the global carbon cycle). A flux tells you not just that something moves, but how fast it moves. If the flux into a reservoir equals the flux out, that reservoir is in steady state.
Three categories of processes drive fluxes between reservoirs:
- Biological processes transform matter through living organisms. Photosynthesis pulls from the atmosphere into biomass; respiration and decomposition release it back.
- Geological processes shape Earth's surface and move elements on long timescales. Weathering breaks down rock and releases minerals, erosion transports sediment, and sedimentation buries material in ocean floors.
- Chemical processes alter the chemical state of elements. Oxidation, reduction, dissolution, and precipitation reactions all convert elements between forms that may be more or less available to organisms.
Living organisms in biogeochemical cycles
Organisms aren't passive passengers in these cycles. They actively drive fluxes at every trophic level.
Primary producers (phytoplankton in oceans, trees and plants on land) capture solar energy and fix inorganic carbon into organic molecules through photosynthesis. They form the base of food webs and represent the main biological pathway for pulling out of the atmosphere.
Consumers transfer energy and matter through food chains. Herbivores, carnivores, and omnivores all contribute to nutrient cycling through metabolic waste products and, eventually, through their own decomposition.
Decomposers (fungi and bacteria) break down dead organic matter and release nutrients back into the soil, water, or atmosphere. Without decomposers, nutrients would stay locked in dead biomass and cycle to a halt.
Microorganisms deserve special attention because they facilitate chemical transformations no other organisms can perform. For example:
- Nitrogen fixation by Rhizobium bacteria converts atmospheric into biologically available ammonium ()
- Nitrification by Nitrosomonas oxidizes ammonium to nitrate ()
- Denitrification by Pseudomonas returns nitrogen to the atmosphere as or
These microbial transformations are often the rate-limiting steps in nutrient cycles, which is why they receive so much attention in biogeochemistry.
Gaseous vs. sedimentary cycles
Not all biogeochemical cycles operate the same way. One of the most useful distinctions is between gaseous and sedimentary cycles.
Gaseous cycles have their primary reservoir in the atmosphere. Carbon and nitrogen are the classic examples. Because gases mix rapidly through the atmosphere, these cycles tend to have faster turnover rates and operate on shorter timescales (years to centuries for the active pools).
Sedimentary cycles have their primary reservoir in the Earth's crust (lithosphere). Phosphorus and sulfur are the main examples. Elements in rock and sediment move slowly through weathering and tectonic uplift, so these cycles operate on timescales of thousands to millions of years.
Key differences at a glance:
| Feature | Gaseous Cycles | Sedimentary Cycles |
|---|---|---|
| Primary reservoir | Atmosphere | Lithosphere (rock, sediment) |
| Turnover rate | Relatively fast | Slow |
| Timescale | Years to centuries (active pools) | Thousands to millions of years |
| Global distribution | Rapid, via atmospheric mixing | Localized, dependent on geology |
| Anthropogenic impact | Effects felt quickly (e.g., emissions raising atmospheric concentrations within decades) | Effects accumulate more slowly (e.g., phosphorus runoff building up in waterways over years) |
This distinction matters practically: when humans perturb a gaseous cycle, the consequences tend to show up faster and spread globally. Sedimentary cycle disruptions are often more regional but can persist for very long periods because the natural recovery mechanisms are so slow.