Understanding Ocean Acidification
Ocean acidification refers to the ongoing decrease in seawater pH driven by the ocean's absorption of atmospheric . Because roughly 30% of anthropogenic ends up dissolved in the ocean, this process directly links fossil-fuel emissions to marine chemistry changes on a global scale. Understanding the underlying carbonate chemistry is essential for predicting how marine ecosystems will respond.
Definition and Chemistry
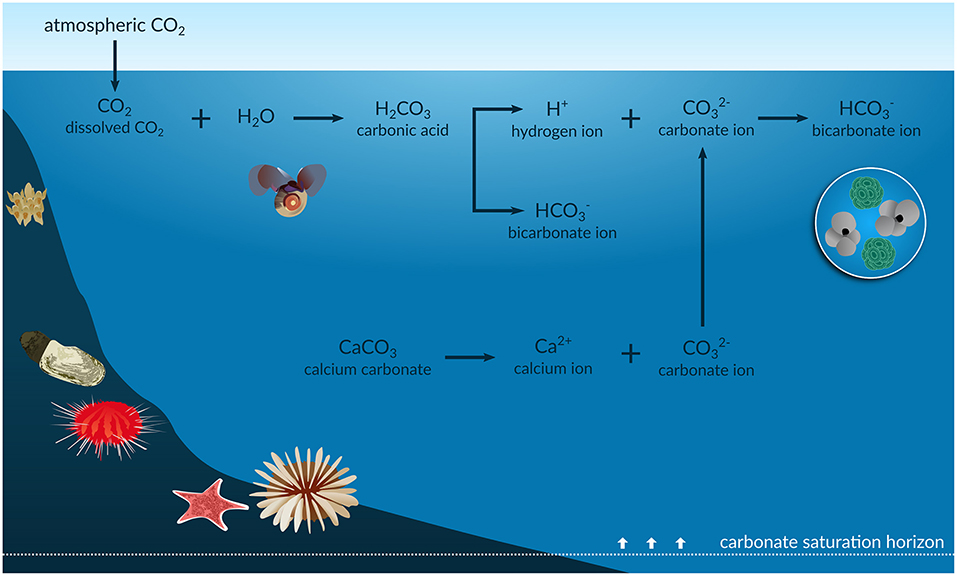
When dissolves in seawater, it reacts with water to form carbonic acid:
Carbonic acid then dissociates in two steps, releasing hydrogen ions that lower pH:
The seawater carbonate system maintains an equilibrium among dissolved , bicarbonate (), and carbonate () ions. As more enters the ocean, that equilibrium shifts: concentrations rise while concentrations fall. The drop in carbonate ion availability is the central problem for shell-building organisms.
Since the Industrial Revolution, surface ocean pH has dropped from about 8.2 to roughly 8.1. That sounds small, but because pH is logarithmic, it represents approximately a 26% increase in hydrogen ion concentration.
The primary drivers of rising atmospheric are:
- Fossil fuel combustion
- Deforestation and land-use change
- Cement production
Impacts on Marine Ecosystems
The decline in availability directly harms calcifying organisms, species that build shells or skeletons from calcium carbonate (). Corals, mollusks, sea urchins, and crustaceans all become less able to calcify, and existing structures can begin to dissolve when the water becomes undersaturated with respect to aragonite or calcite.
Planktonic calcifiers like coccolithophores and pteropods are especially vulnerable. Pteropods form a critical base of polar and subpolar food webs, so their decline can cascade upward through the food chain.
Beyond calcification, acidification causes several other biological disruptions:
- Physiological stress. Marine organisms must spend more energy maintaining their internal acid-base balance, diverting energy away from growth and reproduction.
- Behavioral changes in fish. Elevated interferes with neurotransmitter function (specifically the receptor system), impairing sensory perception, predator avoidance, and homing behavior. Studies on clownfish and damselfish have shown reduced ability to detect predator chemical cues under high- conditions.
- Mixed effects on primary production. Some non-calcifying phytoplankton may benefit from higher dissolved (a fertilization effect), while calcifying phytoplankton experience decreased productivity. The net effect on ocean primary production remains an active research question.
- Altered nutrient cycling. Acidification can change nitrogen fixation rates in cyanobacteria and affect phosphorus availability, with downstream consequences for nutrient limitation patterns across ocean basins.
Consequences and Research on Ocean Acidification
Consequences for Biodiversity and Ecosystem Services
Species that depend on structures face the most direct threat, but the effects propagate through entire ecosystems.
- Community composition shifts. Acid-tolerant species gain a competitive advantage, potentially replacing calcifiers and reducing overall biodiversity.
- Food web disruption. When prey species like pteropods or small mollusks decline, predators at higher trophic levels (salmon, seabirds, marine mammals) lose a key food source.
- Fisheries at risk. Shellfish industries are already feeling impacts. Pacific Northwest oyster hatcheries, for example, experienced severe larval die-offs linked to upwelling of corrosive, low-pH water, prompting real-time monitoring of intake water chemistry.
- Coral reef degradation. Weakened reef structures provide less coastal protection from storm surges and support fewer associated species. Coral reefs harbor roughly 25% of all marine species despite covering less than 1% of the ocean floor.
- Reduced carbon sequestration. Calcifying organisms contribute to the ocean's biological carbon pump. Their decline could weaken the ocean's capacity to draw down atmospheric , creating a positive feedback loop.
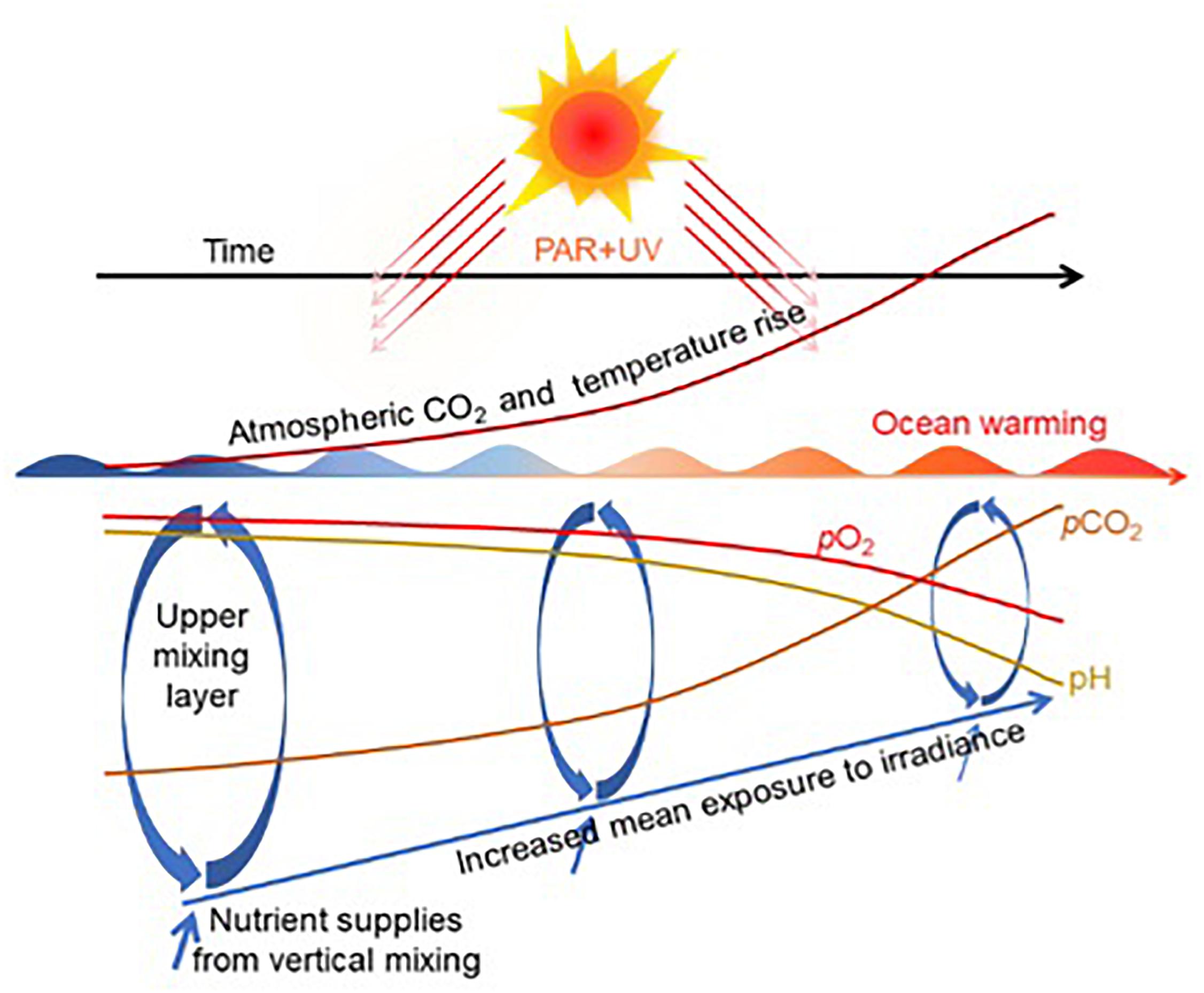
These impacts don't occur in isolation. Synergistic stressors like ocean warming, deoxygenation, and nutrient pollution compound the effects of acidification. Warm water holds less oxygen and increases metabolic demand, so organisms already stressed by low pH face additional physiological burdens.
Role of Biogeochemical Research
Research on ocean acidification spans observation, experimentation, modeling, and policy, and it depends on integrating biogeochemistry with marine biology, ecology, and social science.
Monitoring and observation:
- Long-term time series (such as the Hawaii Ocean Time-series at Station ALOHA) track pH, dissolved inorganic carbon, and alkalinity over decades.
- Global observation networks like the Global Ocean Acidification Observing Network (GOA-ON) coordinate measurements across regions and provide standardized data for trend analysis.
Experimental approaches:
- Mesocosm experiments enclose parcels of seawater and manipulate levels to simulate future conditions, capturing community-level responses that lab studies miss.
- Laboratory studies isolate individual species responses, measuring calcification rates, gene expression, and metabolic costs under controlled pH treatments.
- Free-ocean enrichment (FOCE) systems test acidification effects directly on the seafloor.
Modeling:
- Earth system models project future pH trajectories under different emissions scenarios (e.g., RCP/SSP pathways).
- Ecosystem models couple carbonate chemistry with biological responses to assess long-term impacts on fisheries and biodiversity.
Mitigation and adaptation strategies:
- Carbon dioxide removal techniques (enhanced weathering, direct ocean capture) aim to reduce the burden.
- Restoration of blue carbon ecosystems like mangroves, seagrass beds, and salt marshes enhances local carbon sequestration and can buffer adjacent waters against pH swings.
- Research findings inform international climate agreements (such as the Paris Agreement) and help develop local adaptation strategies for vulnerable coastal communities.
The interdisciplinary nature of this work is worth emphasizing. Biogeochemists provide the chemical framework, marine biologists document organism responses, ecologists trace community-level effects, and social scientists assess economic and cultural impacts on fishing communities. Effective policy depends on all of these perspectives working together.