Forms of Energy
As you recall from unit four, matter can undergo physical and chemical changes. Chemical changes involve the forming or breaking of ionic and covalent bonds between elements, but physical changes involve intermolecular changes. Such energy changes are related to changes in temperature. This can easily be seen with the melting of ice, which is a physical change:
H₂O (s) → H₂O (l)
🔥 Energy is supplied to melt the ice into liquid water! Energy is the capacity to do work or transfer heat. There are three types of energy that are commonly tested:

Kinetic Energy
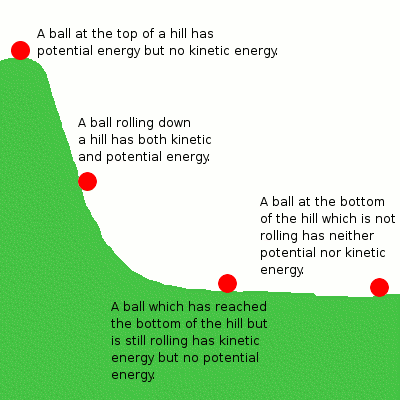
Kinetic energy is the energy that results from the motion of an object. We've discussed kinetic energy a lot so far with regard to temperature since the temperature of particles is directly related to the average kinetic energy of the particles in a substance. Therefore, as temperature increases, the gaseous particles move faster. Remember unit three and the behavior of ideal gases?
KE=½mv^2, where mass, m, is expressed in kilograms and velocity, v, is expressed in meters per second.
KE is expressed in Joules. You don't have to memorize this formula as it is on your AP reference sheet, but be aware that mass and velocity impact the kinetic energy of an object.
Potential Energy
Potential energy is the stored energy in an object based on its position. It usually results from both attractive and repulsive forces. In chemistry, we focus on potential energy as the energy stored in the chemical bonds between atoms and molecules.
PE is related to the stability of a compound as well! Compounds with lower potential energy are more stable than those with higher potential energy. This is because, in order for a compound to be stable, the energy of the bonds between its atoms and molecules must be minimized. When a compound is in its most stable state, the energy of its chemical bonds is at its lowest possible value. We've seen this relationship in several graphs so far throughout this course.
Electrostatic Energy
Electrostatic energy is a form of potential energy that results from the interaction of charged particles. You could associate this with Coulomb's Law, which we discussed several times in previous chapters.
PE of an electron = Q1Q2/d, where charges, Q1 and Q2, are separated by a distance, d
Remember, the attraction between ions only occurs if the charges are opposite and repulsion only occurs if the charges are the same. If you have trouble remembering this, think about magnets!
Image Courtesy of NRGThe Law of Conservation of Energy
This unit is based on one central idea: The Law of Conservation of Energy. This fundamental principle in chemistry and physics states that energy cannot be created or destroyed, only transferred or converted from one form to another. This means that the total amount of energy in a closed system remains constant over time. This applies to all forms of energy.
Let's discuss this in terms of the mechanical system above with the example of the ball rolling down the hill. The potential energy the ball has due to its height can be converted into kinetic energy as it falls, but the total energy remains the same. Similarly, in a chemical reaction, the potential energy stored in the reactants is converted into the potential energy of the products, but the total energy before and after the reaction remains the same.
Since it is so so important, it is also called the First Law of Thermodynamics. Since the universe is considered an isolated system, the total amount of energy in the whole universe is constant.
Studying Energy Changes
The System
To analyze energy changes, we first have to be able to define the system. This is a specific part of the universe that is of interest to us and would typically involve substances used in experiments (ex. HCl, NaOH, etc.). There are several types of systems:
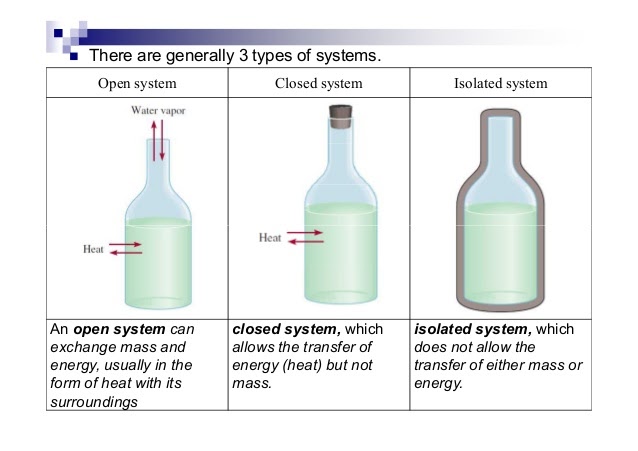
- Open System ⚖- An open system always allows air, heat, and energy from its surroundings into it. It is generally an exchange system. For example, if there was an open system at 90 degrees and the air around it was at 50 degrees, heat and energy would exchange until both would reach 70 degrees. This is called a system at equilibrium which you'll learn more about in unit seven.
- Analogy: An example of an open system is a pot of water on a stove. The water in the pot can exchange heat energy with the stove and the air around it, and it can also exchange matter, such as steam, with the surrounding air.
- Closed System - A closed system is usually represented as an open system, but with a stopper. This way, air cannot transfer into or out of the system. Rather than mass and heat/energy being able to transfer, only heat is able to transfer.
- Analogy: An example of a closed system is a thermos. The contents of the thermos can exchange heat energy with the surrounding air, but the liquid inside the thermos cannot escape.
- Isolated System - An isolated system is completely covered on all ends to prevent the transfer of mass, heat, and energy. An example of this is a calorimeter, which is key in this unit! Insulations allow for an isolated system as well.
- Analogy: An example of an isolated system is a sealed container filled with gas. The gas inside the container cannot exchange matter or energy with the surrounding air.
The Surroundings
The rest of the universe outside the system, such as a beaker, constitutes the surroundings. When taking a look at a question, try to always identify what is in the system and surroundings first.
The system is where the reaction takes place and the surroundings are everything around the system.
State Functions
In thermodynamics, we study changes in the state of a system. State functions, such as energy, enthalpy, pressure, volume, and temperature, are properties determined by the state of the system. It does not matter how the condition was achieved, so focus on the initial and final states of the system only.
Heat and work are not state functions, because they vary on the path to the destination.
An analogy to help understand state functions is to think of a road trip. The distance between your starting point and your destination is a state function because it only depends on the starting and ending point and does not depend on the specific route taken or the speed at which you traveled.
Endothermic vs Exothermic
All forms of energy can be described as either exothermic or endothermic processes. We've discussed these terms when looking at potential energy graphs in the kinetics unit, but let's look at them from the perspective of the system and its surroundings.
Before we do that though, we have to define enthalpy. Enthalpy (H) is a state function that describes the total heat content of a system. ΔH (Delta H) is the change in enthalpy of a system or the difference between the final and initial enthalpies of a system. It is a measure of the heat absorbed or released in a chemical reaction or a physical process such as a change of phase or temperature.
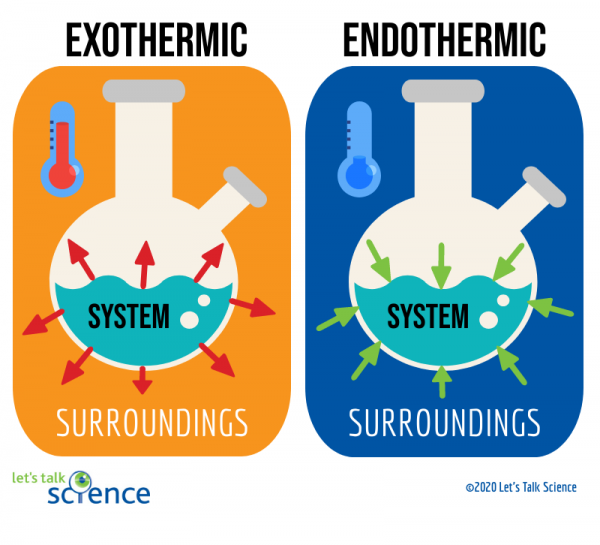
🥵 Endothermic processes, as indicated by the prefix endo-, are processes where heat is supplied to the system by the surroundings. This corresponds with a +ΔH value, indicating that heat is absorbed.
- Remember: endo, positive ΔH, heat travels from surroundings to system.
🥶 Exothermic processes, as indicated by the prefix exo-, are processes that give off heat and transfer thermal energy from the system to the surroundings. This corresponds with a -ΔH value, indicating that the system is releasing or losing heat
- Remember: exo, negative ΔH, heat travels from system to surroundings.
Think about these concepts from the point of view of the system. If the system is gaining energy, there is a positive change, so it is an endothermic process. If the system is losing energy, there is a negative change, so it is an exothermic process. We'll go over ΔH more later in this unit.
Hot and Cold Packs: The Chemistry Behind Them
As you know, hot and cold packs are used to provide therapeutic heat or cold to a specific area of the body. Have you ever wondered how these spontaneous packs work? Well, the chemistry behind these packs involves the use of exothermic and endothermic processes.
Inside these packs, there are two separate bags: one containing water and the other containing a chemical substance. When you crack them, the barrier between the bags break, and the two components react.
An instant hot pack typically contains a substance that can absorb water and undergoes an exothermic reaction when it comes in contact with water. An example of such a substance is sodium acetate, which is a common ingredient in hot packs. When the hot pack is activated by squeezing it, a small amount of water is mixed with the sodium acetate, which triggers an exothermic crystallization reaction, releasing heat into the hot pack and warming you up.
A cold pack, on the other hand, typically contains a substance that can release water and undergoes an endothermic reaction when it comes in contact with water. An example of such a substance is ammonium nitrate, which is commonly used in cold packs. When the cold pack is activated by squeezing it, the ammonium nitrate dissolves in the water, absorbing heat and making the pack feel cold to you.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
chemical transformation | A process in which substances are converted into different substances through the breaking and forming of chemical bonds. |
dissolution process | The process by which a solute dissolves in a solvent to form a solution, which can involve energy changes. |
endothermic reaction | A chemical reaction that absorbs thermal energy from the surroundings, resulting in a positive enthalpy change. |
exothermic reaction | A chemical reaction that releases thermal energy to the surroundings, resulting in a negative enthalpy change. |
heat transfer | The process by which thermal energy is transferred between particles in thermal contact through collisions. |
intermolecular interactions | Forces between molecules, such as hydrogen bonding, dipole-dipole forces, and London dispersion forces, that affect the physical and chemical properties of substances. |
interparticle interactions | Forces between particles in a system that affect the energy changes during physical and chemical processes. |
phase transition | The process by which a substance changes from one state of matter to another (solid, liquid, or gas). |
physical transformation | A process in which the physical state or form of a substance changes without altering its chemical composition. |
surroundings | Everything outside the system with which the system can exchange energy and matter. |
system | The part of the universe being studied, which can exchange energy and matter with its surroundings. |
temperature changes | Variations in the thermal energy of a substance that indicate energy changes in a system. |
Frequently Asked Questions
What's the difference between endothermic and exothermic reactions?
Endothermic and exothermic describe heat flow during a process: exothermic reactions release energy (heat) from the system to the surroundings, so the system’s energy decreases and the surroundings’ temperature usually rises; endothermic reactions absorb energy from the surroundings, so the system’s energy increases and the surroundings cool. On the AP CED this connects to enthalpy change (ΔH): exothermic ΔH is negative, endothermic ΔH is positive (CED 6.1.A.3). Temperature changes you observe (hot/cold) indicate these energy changes (CED 6.1.A.1). Examples: combustion is exothermic; melting ice is endothermic. For lab work you measure these with calorimetry (coffee-cup or bomb calorimeters) and calculate q = m·c·ΔT or use ΔH values/Hess’s law (keywords in the CED). For a quick review, see the Topic 6.1 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and try practice problems on Fiveable (https://library.fiveable.me/practice/ap-chemistry).
How do I know if a reaction is endothermic or exothermic just by looking at it?
Look for where heat/energy is in the equation and whether products are higher or lower in enthalpy than reactants. Quick rules: - If heat appears on the product side (or ΔH is written negative), the system released energy → exothermic. Surroundings get hotter; temperature rises. On an enthalpy diagram, products sit lower than reactants (ΔH < 0). - If heat appears on the reactant side (or ΔH is positive), the system absorbed energy → endothermic. Surroundings cool; temperature falls. On a diagram, products are higher than reactants (ΔH > 0). - Bond idea: forming bonds releases energy (favors exothermic); breaking bonds requires energy (favors endothermic). - Common processes: melting, vaporization, and dissolving (sometimes) are often endothermic; freezing and condensation are exothermic. On the AP exam you’ll be asked to interpret temperature changes, enthalpy diagrams, and ΔH signs (CED 6.1.A). For a quick review, check the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why does the temperature go up in some reactions and down in others?
Temperature goes up when a reaction releases heat to the surroundings (exothermic) and goes down when a reaction absorbs heat from the surroundings (endothermic). In AP terms, temperature change is an experimental sign of an energy change (CED 6.1.A.1). Chemically, exothermic reactions have ΔH < 0: the system’s enthalpy decreases and that lost energy shows up as higher temperature in the surroundings. Endothermic reactions have ΔH > 0: the system gains energy and the surroundings cool. Whether dissolving, phase change, or a bond-making/bond-breaking reaction is endo or exo depends on relative strengths of interactions (CED 6.1.A.2, 6.1.A.4). On the exam you might be asked to explain this qualitatively or use calorimetry math (coffee-cup/bomb calorimeter, q = m·c·ΔT) to quantify heat flow. For a concise review, check the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6). For more practice, try the AP problems at (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when they say energy is absorbed or released in a chemical reaction?
When we say energy is absorbed or released in a chemical reaction, we’re talking about heat flow between the system (the reacting chemicals) and the surroundings. If energy is released—an exothermic reaction—the system’s enthalpy decreases (ΔH < 0) and heat flows out to the surroundings, so the surroundings get warmer (you see a temperature increase). If energy is absorbed—an endothermic reaction—the system’s enthalpy increases (ΔH > 0) and heat flows in from the surroundings, so the surroundings cool. Temperature changes are the experimental signal of these energy changes (CED EK 6.1.A.1–6.1.A.3). On the AP exam you might identify endo/exo from ΔH sign, temperature data, or calorimetry calculations (coffee-cup or bomb calorimeter). For a clear AP-aligned summary and examples, check the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ). For extra practice problems, see Fiveable’s AP Chemistry practice set (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about heat transfer - does the system gain energy or lose energy in an endothermic process?
In an endothermic process the system gains energy (ΔH > 0)—heat flows from the surroundings into the system, so the surroundings lose energy and usually cool. That matches the CED Essential Knowledge: temperature changes indicate energy changes, and for endothermic reactions the energy of the system increases as heat is transferred to (or work is done on) the system. By contrast, exothermic processes release heat to the surroundings (system loses energy, ΔH < 0). On the AP exam you might be asked to identify sign of ΔH, interpret temperature changes, or relate system vs. surroundings in calorimetry problems (Topic 6.1 and 6.4). If you want a quick recap and practice problems for this topic, see the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6).
Can melting ice be considered an endothermic process and why?
Yes—melting ice is an endothermic process. During melting a solid (ice) becomes liquid, and the system (the ice) must absorb heat from the surroundings to break some of the intermolecular hydrogen bonds without increasing temperature. That absorbed heat is the enthalpy of fusion (ΔHfus), which is positive for melting. Experimentally you’d see energy flow into the system (surroundings cool or heat source supplies energy) while the ice melts—exactly the CED idea that phase changes can be endothermic or exothermic and that temperature changes indicate energy transfer (6.1.A.1–6.1.A.3, keywords: phase change, enthalpy change, heat flow). On the exam you might justify this with ΔH > 0, or by describing heat flow from surroundings into the system during melting. For more review, see the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) or the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6). For practice problems, check https://library.fiveable.me/practice/ap-chemistry.
How do you tell if dissolving salt in water is endothermic or exothermic?
You tell by measuring the temperature change of the surroundings (usually the solution) or by calculating the enthalpy change. If the solution warms, the process is exothermic (system releases heat, ΔH < 0). If the solution cools, the process is endothermic (system absorbs heat, ΔH > 0). Experimentally you can use a simple coffee-cup calorimeter: qsolution = m·c·ΔT (mass of solution, specific heat, temperature change) and qsystem = −qsurroundings to find ΔH per mole dissolved. Physically, whether dissolution is endo- or exothermic depends on lattice energy (energy required to separate ions) versus hydration (solvation) enthalpy (energy released when ions interact with water). If hydration > lattice energy → exothermic; if hydration < lattice → endothermic. Many common salts (e.g., NaCl) are nearly thermoneutral or slightly endothermic. This matches CED essential knowledge 6.1.A.1–6.1.A.4 (temperature change indicates energy transfer; enthalpy of solution depends on interparticle interactions). For a quick review, see the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ). For extra practice, check Fiveable’s Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between temperature changes and energy changes in chemical reactions?
Temperature changes are direct evidence of energy flow between the system and surroundings: if the system’s temperature rises, it’s released energy to the surroundings (exothermic); if the system’s temperature falls, it’s absorbed energy from the surroundings (endothermic). On the AP CED this is EK 6.1.A.1–A.3: you’ll describe processes as exothermic (ΔH < 0, heat leaves the system) or endothermic (ΔH > 0, heat enters the system). Experimental observations like a cup warming or cooling, phase changes, or solution formation tell you the sign and direction of heat flow—which you can quantify with calorimetry using q = mcΔT or coffee-cup/bomb calorimeter setups (CED keywords: enthalpy change, calorimetry, specific heat). For practice, review the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and try problems on Fiveable’s AP practice page (https://library.fiveable.me/practice/ap-chemistry) to build skill with ΔT → q → ΔH reasoning needed on the exam.
Why do some phase changes require energy while others release energy?
Phase changes that require energy are endothermic (ΔH > 0); those that release energy are exothermic (ΔH < 0). Why? Think about intermolecular (or interparticle) attractions: breaking attractions (solid → liquid → gas) requires input of energy to overcome bonds/IMFs, so the system’s enthalpy increases and it absorbs heat from the surroundings. Forming attractions (gas → liquid → solid) releases energy because potential energy is lowered when particles get closer and bonds/IMFs form, so heat flows out to the surroundings. Temperature changes show these energy flows (CED 6.1.A.1–6.1.A.3). On the AP exam you may be asked to identify whether a phase change is endo/exo by the sign of ΔH, connect it to heat flow between system and surroundings, or explain using bond/IMF strength. For a clear review and examples (including practice problems), see the Topic 6.1 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand the difference between the system and surroundings in thermodynamics - can someone explain?
Think of the system as the stuff you choose to study (the reacting chemicals, the substance changing phase)—everything else is the surroundings (the container, air, calorimeter, your hand). In thermochemistry we track energy flow between them. If the system loses heat, the surroundings gain it (exothermic: ΔH < 0; you’ll see the surroundings warm). If the system gains heat, the surroundings cool (endothermic: ΔH > 0). A handy rule: qsystem = −qsurroundings (what one loses the other gains). Temperature change in the system is experimental evidence of an energy change (CED 6.1.A.1–6.1.A.3), and calorimetry (coffee-cup or bomb) measures qsurroundings to find ΔH for the system. For AP problems, label which is your system, state the sign of ΔH, and use q = m·c·ΔT when given masses and specific heats. For more examples and CED-aligned practice, check the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do intermolecular forces affect whether dissolving something is endothermic or exothermic?
When a substance dissolves, three energy steps matter: (1) break solute–solute attractions (endothermic), (2) break solvent–solvent attractions (endothermic), and (3) form solute–solvent attractions (exothermic). The sign of the enthalpy of solution (ΔHsoln) depends on the net balance: if energy released forming solute-solvent interactions > energy required to separate particles, dissolution is exothermic (surroundings warm); if not, it’s endothermic (surroundings cool). AP terms you should use: lattice energy (energy to separate an ionic solid), hydration/solvation energy (energy released when ions/molecules interact with solvent), and enthalpy of solution. Temperature change in a calorimeter shows whether heat flowed into or out of the system (CED 6.1.A.1, 6.1.A.4). For practice, study examples like NH4NO3 (strongly endothermic) and many ionic salts (small ± ΔH) in the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and try problems at https://library.fiveable.me/practice/ap-chemistry.
What are some real world examples of endothermic and exothermic processes I can use for my FRQ?
Use clear, testable examples that show temperature change and ΔH sign. Endothermic (ΔH > 0): ice melting or water evaporating (system absorbs heat → surroundings cool), photosynthesis (plants absorb sunlight to make glucose), dissolving ammonium nitrate in water (common “cold packs”)—measured as a drop in temperature in calorimetry. Exothermic (ΔH < 0): freezing water or condensation (system releases heat → surroundings warm), combustion (burning hydrocarbons or candle wax), neutralization (HCl + NaOH releases heat), dissolving strong bases like NaOH in water (common lab heat release). In an FRQ, state whether system or surroundings gains heat, give ΔH sign, and tie to observed temperature change (CED 6.1.A.1–6.1.A.3). For calorimetry/quantitative parts mention q = m·c·ΔT and that formation/dissolution can change sign depending on bond/lattice vs. solvation energies (CED 6.1.A.4). For more examples and AP-aligned review, see the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
If a reaction feels cold to touch, does that automatically mean it's endothermic?
Not automatically. If something “feels cold” you’re noticing the surroundings losing heat—which usually means the system absorbed heat (an endothermic process). That’s exactly the CED idea: a temperature change in the surroundings indicates energy flow between system and surroundings (6.1.A.1–6.1.A.3). But a cold sensation alone isn’t definitive because other effects can mimic cooling: evaporative cooling, heat capacity differences, rapid heat flow through the container, or very small temperature changes that your hand misreads. On the AP exam you should connect the observation to energy transfer and, when asked, justify with calorimetry or enthalpy language (q into system > 0 for endothermic). The safest answer in a lab or FRQ is to say the cold feel suggests the reaction may be endothermic and then propose a test—measure temperature change of the system/surroundings and calculate q (q = m·c·ΔT) or use a calorimeter—to confirm. Want quick practice applying this? See the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
How do I remember which direction energy flows in endothermic vs exothermic reactions?
Think “where does heat go?” and label the system vs the surroundings. If the system loses energy (heat flows out), the surroundings warm—that’s exothermic: ΔH < 0. If the system gains energy (heat flows in), the surroundings cool—that’s endothermic: ΔH > 0. A quick mnemonic: “Exit = exothermic (energy exits the system)” and “Enter = endothermic (energy enters the system).” On the AP, link this to observations: measure temperature—a temperature increase in the surroundings means the reaction was exothermic (CED 6.1.A.1–6.1.A.3). Draw an enthalpy diagram: products lower than reactants for exothermic (release), products higher for endothermic (absorb). Use these ideas on calorimetry problems (coffee-cup/bomb calorimeter) and when predicting ΔH sign. For more practice and a short study guide on this Topic 6.1, see Fiveable’s guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and thousands of practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why does breaking bonds require energy but forming bonds releases energy?
Breaking a bond requires energy because you're pulling charged particles (nuclei and electrons) apart against attractive electrostatic forces. You must input energy to overcome that attraction and raise the potential energy of the separated atoms—that’s an endothermic step (bond enthalpy > 0). Forming a bond does the opposite: as atoms come together, attractive forces pull electrons into lower-energy arrangements, so potential energy decreases and that excess energy is released to the surroundings—an exothermic step (bond enthalpy released). In a chemical reaction the overall ΔH depends on the balance: total energy absorbed to break bonds minus total energy released forming new bonds. If breaking costs more than forming releases, the reaction is net endothermic (system gains energy); if forming releases more, it’s net exothermic (system loses energy and surroundings warm). On the AP exam you’ll connect these ideas to temperature changes and enthalpy (CED 6.1.A.1–6.1.A.3). For a clear review and practice problems, see the Topic 6.1 study guide (https://library.fiveable.me/ap-chemistry/unit-6/endothermic-exothermic-processes/study-guide/IBke22j0ulvsM9ps3QPJ) and more unit review (https://library.fiveable.me/ap-chemistry/unit-6). Fiveable also has tons of practice questions to help you apply bond enthalpies.