Calorimetry
The absolute enthalpy of a system (H) cannot be measured directly, but changes in enthalpy (ΔH) can be measured using calorimetry. Calorimetry is the study of heat flow and heat exchange between a system and its surroundings and it can be used to calculate ΔH by measuring changes in temperature, which represent heat being lost or gained.

Types of Calorimeters
Calorimetry is a process by which a reaction takes place in a controlled vessel in which there is a liquid (typically water) and a temperature gauge like a thermometer to measure how the liquid heats up. There are several types of calorimeters that can be used to measure the heat flow in a chemical reaction or physical process:
- In a bomb calorimeter, a reaction takes place in a sealed container, called a bomb, and the heat generated by the reaction is used to raise the temperature of the surrounding water. By measuring the rise in temperature of the water and knowing its specific heat, the heat of the reaction can be calculated.
- In a constant-pressure calorimeter, the heat of a reaction is measured by monitoring the change in temperature of the reaction mixture at a constant pressure. This is because at constant pressure, the amount of heat transferred to a system is equal to ΔH.
- In a coffee-cup calorimeter, which is a simple calorimeter, the heat released or absorbed by a reaction is used to raise or lower the temperature of the surrounding water. By measuring the temperature change of the water and knowing its mass and specific heat, the heat of the reaction can be calculated. Coffee-cup calorimetry is going to be our focus in this unit, and we will learn how to use it to observe changes in energy.
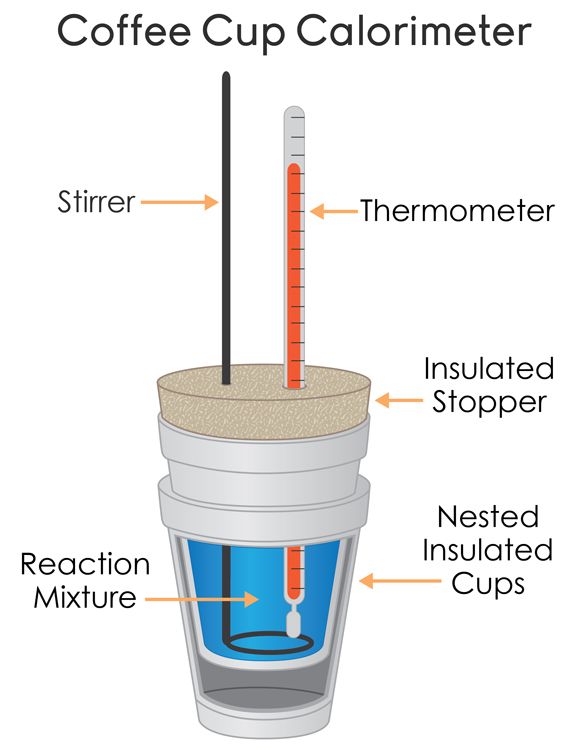
Coffee-Cup Calorimetry
We have a few parts to this coffee-cup calorimeter:
- The thermometer
- The reaction mixture
- The stirrer to stir the reaction mixture and allow for more accurate temperature measurements.
- An insulated container (such as a styrofoam cup)
- Heat-proof lid to cover the calorimeter
The calorimeter is meant to insulate the sample, meaning that heat cannot come in or out of the system. The more insulated, the better the understanding of heat changes during a chemical reaction
Quantifying Energy
The First Law of Thermodynamics
Since a calorimeter is insulated, we can think of it as an isolated system where the First Law of Thermodynamics is significant. To recall, it states that energy cannot be created or destroyed, only transferred or converted from one form to another. This means that the total amount of energy in the closed calorimeter remains constant over time.
👉 Need a quick review of this fundamental principle of chemistry and physics? Check out our study guide discussing endothermic and exothermic processes, featuring this concept.
Measuring Heat Transferred
Okay, so let's back up. Now, we know of a technique we can use to measure how much energy is absorbed or released during a chemical reaction. When we need to quantify this transfer of energy, we can use coffee-cup calorimetry and allow the chemical reaction to occur in the calorimeter.
Once the chemical reaction occurs, we can use the equation q=mCΔT to quantify the magnitude of heat transferred between the system and its surroundings in a calorimeter. Let's break it down:
- q is the heat in Joules
- m is the mass in grams or kilograms
- C is the specific heat of the substance, and
- ΔT is the change in temperature in Kelvin
In most questions, you are going to be solving for q. Regardless, you should be mindful of the units of each of these components. Note that because Celcius and Kelvin are equivalent scales, ΔT for Celcius is going to be the same in Kelvin.
Since the heat of reaction is an extensive property, it is dependent on the mass of the substance. This is why mass is included in this heat transfer equation.
Specific Heat
As you can see, the measurement of heat changes involves knowing specific heat, the amount of heat required to raise the temperature of a gram of a substance by 1° C. This can be used to determine heat capacity, the amount of heat required to raise the temperature of an object by 1° C.
Well...specific heat is the amount of energy required to raise the temperature of a gram of a substance by 1° C, but what the heck does this actually mean?
💧Water
Let's take a look at a real-life example: boiling water. Do you ever get annoyed at the speed at which water boils? It's soooo slow, it becomes annoying to wait when hungry.
The reason why water takes a long time to boil is because of its high specific heat. This means that water takes a long time to take in energy, or enough energy, for the water to boil. The specific heat of water is 4.184 J/g.
🏖️Sand
Now let's take a look at sand: does it heat up faster than water? When you go to the beach, you probably feel the scorching hot sand at your feet. It heats up a lot faster than water—it takes much less energy to heat sand up than it takes to heat water up. The specific heat of sand is about 0.840 J/g.
The specific heat of sand is much less than the specific heat of water. Therefore, we can conclude that the higher the specific heat, the more energy it takes for an object to heat up and cool down.
Usually, the specific heat of an object is given on the AP Chemistry exam. You just have to know when to plug it in (q=mCΔT is only one of the few equations in this unit😉).
Calorimetry Examples
Question 1
An insulated cup contains 255.0 grams of water and the temperature changes from 25.2 °C to 90.5 °C. Calculate the amount of heat released by the system. The specific heat capacity of water is 4.184 J/g°C.
When looking at this question, you should recognize that calorimetry is being used. Therefore, use q = mcΔT and plug in the values you know:
q = (255.0 g)(4.184 J/g°C)(90.5-25.2°C)
Make sure the units that you are using match since mass can be given in grams or kilograms. As long as the units of your mass match your specific heat, you should be good to go! For example, if you were to use 0.2550 kg and 4.184 J/g°C, you are mixing kilograms and grams, messing up your calculation. Let's solve the rest:
q = (255.0 g)(4.184 J/g°C)(65.3°C)
q = 69,700 J or 69.7 kJ since there are 1000 J in 1 kJ.
We will be going over more equations later in this unit, so whenever you see a temperature change in a problem, think about using this specific formula.
Misconception - q vs. ΔH
Right now, we can look at q and ΔH and think about heat. However, q does not equal ΔH.
For the purpose of this course, q is always going to be positive. You can think of it as a magnitude of energy. ΔH could be positive or negative though; it depends on if the heat is released or absorbed.
Question 2
The following question is from the Advanced Placement YT Channel. The following laboratory procedure is being done in a calorimeter.
| Mass of Copper | 50.00g |
|---|---|
| Initial Temperature of Copper | 100.0°C |
| Mass of Water | 100.00g |
| Initial Temperature of Water | 20.0°C |
| Final Temperature of System (Copper and Water) | 23.6°C |
| Specific heat capacity of water | 4.18 J/g°C |
| Specific heat capacity of copper | ? |
(a) What is |ΔT| for the copper? What is |ΔT| for the water?
Whenever you see changes in temperature, you should automatically find the change in temperature (ΔT). ΔT is always calculated by final temperature - initial temperature.
- Copper: |23.6°C - 100.0°C| = 76.4°C
- Water: 23.6°C - 20°C = 3.6°C
(b) A student claims that, since the magnitude of ΔT for the copper is greater than that of the water, it means that the magnitude of heat (q) lost by the copper is greater than the magnitude of heat (q) gained by the water. Do you agree with this claim?
Let's recall the first law of thermodynamics: no energy could be created or destroyed. Therefore, the heat lost by the copper must be equal to the heat gained by the water.
Sample Response: Assuming the calorimeter lost no heat to its surroundings, the heat lost by the copper must equal the heat gained by the water, despite their significant changes in temperature.
(c) Find the specific heat of copper.
Considering we only learned one formula so far, we must use q=mcΔT. In general, however, if you see specific heat, the formula you have to use is most likely this one.
Since we are solving for c, we must be given q, the mass of the copper, and the change in temperature. One problem: we don't have q! Let's go back to part b...the magnitude of heat lost by the copper is equal to the magnitude of heat gained by the water.
This enables us to use q=mcΔT twice, by setting the water calculation equal to the copper calculation (q for copper is equal to q for water so what q equals for copper must be equivalent to what q equals for water).
The setup looks like this:
mcΔT (copper) = mcΔT (water)
(50.00 g)(C)(76.4 °C) = (100.00 g)(4.18 J/g°C)(3.6 °C)
C = 0.39 J/g°C
Another Formula
So we know that we could use q=mcΔT, but when we have a heat loss and heat gain question, we could also use:
heat loss = heat gain or mcΔT (1st substance) = mcΔT (2nd substance) like we just did!
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
calorimetry | An experimental technique used to measure the amount of heat transferred between systems. |
dissolution | The process by which a solute dissolves in a solvent to form a solution, involving the breaking of bonds or interactions in the solute and formation of new interactions with the solvent. |
endothermic reaction | A chemical reaction that absorbs thermal energy from the surroundings, resulting in a positive enthalpy change. |
exothermic reaction | A chemical reaction that releases thermal energy to the surroundings, resulting in a negative enthalpy change. |
first law of thermodynamics | The principle that energy is conserved in chemical and physical processes; energy cannot be created or destroyed, only transferred or transformed. |
heat | Energy transferred between two systems due to a difference in temperature. |
heat transfer equation | The mathematical relationship q = mcΔT used to calculate heat absorbed or released, where m is mass, c is specific heat capacity, and ΔT is the change in temperature. |
molar heat capacity | The amount of thermal energy required to raise the temperature of one mole of a substance by one degree Celsius. |
phase transition | The process by which a substance changes from one state of matter to another (solid, liquid, or gas). |
specific heat capacity | The amount of thermal energy required to raise the temperature of one gram of a substance by one degree Celsius. |
thermal energy | The total kinetic energy of particles in a substance due to their random motion. |
Frequently Asked Questions
What is heat capacity and how is it different from specific heat?
Heat capacity (C) is the amount of heat required to raise the temperature of an object or sample by 1°C (units: J/°C or J/K). Specific heat capacity (c, often just “specific heat”) is an intensive property: the heat required to raise 1 gram of a substance by 1°C (J/g·°C). Use q = m c ΔT when you know mass and specific heat—that’s the AP-tested formula for calculating heat transferred (CED 6.4.A.1). If you instead know the whole object’s heat capacity, use q = C ΔT. Related: molar heat capacity is heat per mole per degree (J/mol·°C). In calorimetry, you often measure temperature change and use specific (or molar) heats, plus calorimeter constants for heat absorbed by the apparatus. For a focused review, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN). More practice problems across Unit 6 are at (https://library.fiveable.me/practice/ap-chemistry).
How do you use the equation q = mcΔT in calorimetry problems?
Use q = mcΔT to calculate the heat gained or lost by a substance: q (J) = m (g) × c (J/g·°C) × ΔT (°C, = Tfinal − Tinitial). Steps you’ll use on AP problems (Topic 6.4): 1. Identify which body you’re finding q for (usually water or the system). Use mass in grams and the correct specific heat (water ≈ 4.184 J/g·°C unless told otherwise). 2. Compute ΔT = Tfinal − Tinitial. If T rises, ΔT positive → q positive (system gained heat). If T falls, ΔT negative → q negative (system lost heat). 3. Plug into q = mcΔT and keep units (J). For calorimetry, apply conservation of energy: qlost + qgained = 0 (or qsystem = −qsurroundings). 4. If asked per mole, convert mass → moles and use molar heat capacity or divide q by moles. On the exam you may need the calorimeter constant or include calorimeter heat (qcal = CcalΔT). For review and worked examples see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and extra practice (https://library.fiveable.me/practice/ap-chemistry).
Why does water have such a high specific heat capacity compared to other substances?
Water’s specific heat is high (about 4.18 J/g·°C) because a lot of the added thermal energy goes into changing intermolecular interactions rather than immediately raising kinetic energy (temperature). Water is small and highly polar, so molecules form a dense network of hydrogen bonds. To raise water’s temperature you must partially overcome and excite those H-bonded arrangements (stretching/rotational/vibrational modes), which consumes energy per gram more than in substances with weaker intermolecular forces. That’s why, for the same q in q = mcΔT, ΔT is smaller for water (CED 6.4.A.1 & 6.4.A.3). Practically: water moderates temperature changes in calorimetry and is why it’s commonly the calorimeter medium. For more review on specific heat and calorimetry examples, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6). Want extra practice? Try the AP Chem problem set list (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about calorimetry - can someone explain how those coffee cup experiments actually work?
Coffee-cup calorimetry is just a simple way to measure heat transfer at constant pressure (like in many AP problems). You mix a hot (or reactive) sample into water inside an insulated cup, wait until thermal equilibrium, measure the temperature change ΔT, and use q = mcΔT to find the heat gained or lost. Because of energy conservation (First Law), q(system) + q(surroundings) = 0—so if the solution/water warms by qwater = mwater·cwater·ΔT, the reaction or added object released qreaction = −qwater. Remember to include heat absorbed by the calorimeter itself if it’s not ideal (use a calorimeter constant or include the mass & specific heat of the cup/spoon). Use cwater ≈ 4.18 J/g·°C unless told otherwise. On the AP exam you’ll be asked to calculate sign (exothermic vs endothermic), use specific/molar heat as needed, and show energy bookkeeping. For a focused review, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN). Want more practice? Try the AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between specific heat capacity and molar heat capacity?
Specific heat capacity (usually c) is the energy required to raise 1 gram of a substance by 1°C: units J/(g·°C). Molar heat capacity (Cm) is the energy required to raise 1 mole of a substance by 1°C: units J/(mol·°C). Both show up in AP Thermochemistry problems (CED 6.4.A)—use whichever matches the amount you have: - If you know mass: q = m c ΔT (q in J). - If you know moles: q = n Cm ΔT. They’re directly related: Cm = M × c, where M is molar mass (g/mol). Example: water c ≈ 4.18 J/(g·°C); M = 18.02 g/mol → Cm ≈ 18.02×4.18 ≈ 75.3 J/(mol·°C). On the AP exam pick the form that fits the given quantity and units (you’ll see both forms in questions). For a focused refresher, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I know if a reaction is endothermic or exothermic from calorimetry data?
Look at the temperature change of the calorimeter contents and use q = mcΔT (plus any calorimeter constant). Simple rules: - If the measured temperature of the solution/mixture rises, the process released heat → reaction is exothermic. - If the temperature falls, the process absorbed heat → reaction is endothermic. Do the math: calculate q_surroundings = m·c·ΔT (include the calorimeter’s heat capacity if given). By energy conservation (1st law), q_system + q_surroundings = 0, so q_reaction = −q_surroundings. Example: 100.0 g water (c = 4.18 J/g·°C) with ΔT = +5.00 °C gives q_water = 100·4.18·5 = 2090 J, so q_reaction = −2090 J (exothermic). For constant-pressure coffee-cup calorimetry, ΔH ≈ q_p. This is exactly the type of reasoning AP asks you to use (CED 6.4.A; q = mcΔT). For a quick topic review see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What does the first law of thermodynamics have to do with heat capacity calculations?
The first law of thermodynamics—energy is conserved—is the foundation for heat-capacity and calorimetry calculations. In practice that means the heat lost by one part of the experiment equals the heat gained by the other: q(system) + q(surroundings) = 0. So when you use q = m c ΔT (CED 6.4.A.1) to find the heat change of a substance, you’re applying the first law to track energy flow. In a coffee-cup calorimeter you usually set q(reaction) = −q(water + calorimeter); in a bomb calorimeter you account for the calorimeter constant (q = Ccal ΔT). Watch sign convention: ΔT positive → q absorbed (endothermic), ΔT negative → q released (exothermic) (CED 6.4.A.2, 6.4.A.7). For AP: be ready to calculate q with specific or molar heat capacity and justify energy conservation in your answer (Topic 6.4). Review worked examples in the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do equal masses of different materials heat up at different rates?
Because different materials store different amounts of thermal energy per gram, equal masses heat at different rates. The heat equation q = mcΔT tells you why: for the same heat input q and the same mass m, the temperature change ΔT = q/(m c). A large specific heat c means the substance needs more energy to raise its temperature, so ΔT is smaller (it heats more slowly). Example: water’s c ≈ 4.18 J/g·°C, many metals have c ≈ 0.1–1 J/g·°C—the metal will warm much faster than the same mass of water for the same heat added. On a molecular level, specific heat depends on how energy is stored (translational, rotational, vibrational modes), bond strength, and structure. In calorimetry you use q = mcΔT to measure heat flows and find c or ΔH; practice these setups for the AP exam (topic 6.4). For a clear study guide on this topic, see Fiveable’s heat-capacity/calorimetry page (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN). For more practice questions, check (https://library.fiveable.me/practice/ap-chemistry).
How do you calculate the heat absorbed or released when something changes temperature?
Use q = mcΔT. Here q is heat (J), m is mass (g), c is specific heat (J/g·°C) or use molar heat capacity (J/mol·°C) with moles, and ΔT = Tfinal − Tinitial (°C or K). Positive q = heat absorbed (endothermic); negative q = heat released (exothermic). In calorimetry problems remember energy is conserved: qsystem + qsurroundings = 0, so qsystem = −qsurroundings. Example: heat needed to warm 50.0 g of water (c = 4.18 J/g·°C) from 20.0 °C to 80.0 °C: q = (50.0 g)(4.18 J/g·°C)(80.0−20.0 °C) = 12,540 J (absorbed). On the AP exam you'll be asked to calculate q from mass or moles and c (CED 6.4.A, use q = mcΔT). For calorimetry labs, use q lost by hot object = −q gained by cold object and include the calorimeter constant if given. For more examples and aligned study notes, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how to set up calorimetry problems - do I always use q = mcΔT?
Short answer: usually, but not always. q = mcΔT is the go-to for heating/cooling a single substance (mass m, specific heat c, temp change ΔT)—that’s Essential Knowledge 6.4.A.1. But real calorimetry problems often need extra pieces: - Add/subtract heat terms: qreaction + qwater + qcalorimeter = 0. For a coffee-cup calorimeter you’ll use qwater = mcΔT and qcalorimeter = Ccal·ΔT (calorimeter constant), then set qrxn = −(qwater + qcalorimeter). - Phase changes use q = m·ΔHfus or m·ΔHvap (latent heat) not mcΔT. - Use molar heat capacity (Cmol) when given moles: q = n·Cmol·ΔT. - Bomb calorimeter: heat measured at constant volume; use qrxn = −(Cbomb·ΔT) or include calorimeter heat capacity plus water. On the AP exam, show conservation of energy (first law) and label signs (+/-) clearly. For a focused study guide and practice problems, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6). For lots of practice, try Fiveable’s AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
What happens to energy when you heat up a system versus when you cool it down?
When you heat a system, thermal energy is transferred into it—the system’s internal energy increases and its temperature usually rises; when you cool a system, thermal energy leaves it—its internal energy decreases and temperature falls. Quantitatively AP expects you to use q = mcΔT (q positive when the system absorbs heat, negative when it releases heat) and remember the first law: energy is conserved (ΔE = q + w). In calorimetry you measure these transfers (coffee-cup for constant-pressure, bomb calorimeter for constant-volume) and use temperature changes plus specific or molar heat capacities to calculate q (Topic 6.4 / LO 6.4.A). If a dissolution raises the calorimeter temperature the process is exothermic (releases heat); if it lowers temperature it’s endothermic (absorbs heat). For practice, review the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
How do phase transitions affect heat capacity calculations?
Phase changes break the simple q = mcΔT rule because temperature doesn't change while a phase transition happens. For heating/cooling you use q = mcΔT (CED 6.4.A.1, 6.4.A.5), but for melting/boiling you must use latent (transition) heat: q = m·L (L = heat of fusion or vaporization). In calorimetry problems treat the process in steps—heat or cool to the phase-change temperature (use q = mcΔT), then apply q = mL for the phase change (ΔT = 0), then continue heating/cooling the new phase with its specific heat. Sum all q terms (and include calorimeter heat if given) and conserve energy (first law) to solve. Remember specific vs. molar heat capacities and include correct units. For guided examples and AP-style practice, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does the temperature of the solution change when you dissolve salt in water?
When you dissolve salt in water the temperature changes because energy is transferred between the solution and its surroundings as bonds are broken and formed—that’s the enthalpy of solution (ΔHsoln). Breaking the ionic lattice (and hydrating ions) requires energy; forming ion–water interactions releases energy. If the net process releases more energy than it consumes, the solution heats up (exothermic); if it absorbs more, the solution cools (endothermic). In a calorimeter the heat lost or gained by the solution equals q = mcΔT (CED 6.4.A.1, 6.4.A.7), so the measured ΔT tells you the direction and magnitude of heat flow. Remember energy is conserved (first law), so the temperature change reflects the system reaching thermal equilibrium after that energy exchange. For a quick review, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
What are the three main ways chemical systems can change their energy?
There are three main ways a chemical system changes its energy (CED 6.4.A.6): - Heating/cooling (thermal energy transfer): energy flows into or out of a system as heat, changing temperature. You calculate this with q = mcΔT using specific or molar heat capacity (CED 6.4.A.1, 6.4.A.5). Calorimetry measures these transfers. - Phase transitions (latent heat): energy is absorbed or released without a temperature change when a substance melts, vaporizes, etc. Use latent heats (ΔHfusion, ΔHvap) in energy bookkeeping (related Topic 6.5). - Chemical reactions (bond making/breaking): reaction enthalpies (ΔHrxn) change system energy—exothermic reactions release heat, endothermic absorb it (CED 6.4.A.7, 6.4.A.2). On the AP exam you’ll be asked to calculate q for heating/cooling problems, infer direction of heat flow in calorimetry, and connect phase-change or reaction enthalpies to temperature changes (see the Topic 6.4 study guide for worked examples: https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN). For broader review, check the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6) and drill practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I determine the direction of energy flow in a dissolution experiment?
Look at the temperature change of the solution and use q = mcΔT plus energy conservation. Steps: measure Tinitial and Tfinal of the calorimeter mixture, find ΔT = Tfinal − Tinitial, then calculate q(solution) = msolution · c · ΔT (use cwater ≈ 4.18 J/g·°C if water is the solvent). If the calorimeter has a known heat capacity, add q(cal) = Ccal · ΔT and include that in the surroundings’ heat. By the first law, qsystem = −qsurroundings. So: - If Tfinal > Tinitial → ΔT positive → q(solution) positive → solution gained heat → dissolution released heat (exothermic; qsystem < 0). - If Tfinal < Tinitial → ΔT negative → q(solution) negative → solution lost heat → dissolution absorbed heat (endothermic; qsystem > 0). This aligns with CED 6.4.A (q = mcΔT, thermal energy flow, conservation of energy). For practice, see the Topic 6.4 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-capacity-calorimetry/study-guide/jShImkrhZMnPWxlEjdwN) and more problems (https://library.fiveable.me/practice/ap-chemistry).