Before we start learning about how to use mechanisms to find something out about the rate of a reaction, let's do a quick refresher on what exactly a mechanism is. A mechanism describes the steps of a reaction as it takes place from reactants to products. It's important to note that mechanisms tell us exactly what happens in a reaction, as the vast majority of reactions happen in many steps.
Review of Mechanisms
When you see a reaction A → B, there are typically multiple steps that lead to A yielding B. This could mean A reacting with an intermediate, or the reaction being catalyzed. Either way, each step is known as an elementary steps, and when you add all of the elementary steps of a mechanism together, you will get the overall balanced chemical equation.

Elementary Steps
Let's take a look at an example mechanism and break it down:
This is the mechanism for the reaction: O₃ + 2I⁻ + H₂O → O₂ + I₂ + 2OH⁻. How did we find that out? Well, if you add up all of the steps of a mechanism, it must and always will add up to the overall reaction.
In this mechanism, there are three elementary steps:
- In the first step, ozone and iodide slowly react to form molecular oxygen and iodite ions.
- In the second step, the formed iodite ions react with water molecules to form hypoiodous acid and hydroxide ions.
- In the last step, the formed hypoiodous acid from the previous elementary step reacts with iodide to form molecular iodine and more hydroxide ions.
Catalysts and Intermediates
Breaking this down helps us identify any possible intermediates or catalysts. Since there are no chemical substances that go into the mechanism and come out without being affected, there are no catalysts.
However, there are two intermediates in this mechanism. Can you identify them? Intermediates are species that are formed during a reaction and then go on to participate in further reactions. When comparing the first step to the second step, we can find that iodite is one of the intermediates. When comparing the second to the third, we can also see that hypoiodous acid is the second intermediate!
👉 If you'd like a review on any of this material, check out the previous guide that explained how to identify the components of a reaction mechanism.
Rate-Determining Steps
Now that we've been refreshed on what mechanisms are, let's start discussing rate laws and how to write the rate law using a mechanism. When writing the rate law of a mechanism, we need to identify the rate-determining step. The rate-determining step is the slowest step of a mechanism. This step essentially constrains how fast the reaction can go!
From here, we can use the stoichiometric coefficients of the slowest step in the mechanism to find the rate law. It is important to note that besides mechanisms, rate laws must be determined experimentally. In fact, problems often are formatted along the lines of, "The mechanism for a reaction is {mechanism}. If {rate law} is the experimentally determined rate law, is this mechanism valid?" Since the overall rate law of a reaction must be found experimentally, it can be used to verify mechanisms.
Example Free-Response Question
Let's break down this idea and put it into practice with an AP Chemistry free-response question (FRQ). The following question is #6 from the 2019 AP Chemistry exam posted online by College Board.
Understanding Basic Rate Laws
Nitrogen dioxide, NO₂ (g), is produced as a by-product of the combustion of fossil fuels in internal combustion engines. At elevated temperatures NO₂ (g) decomposes according to the equation below.
2 NO₂ (g) → 2 NO (g) + O₂ (g)
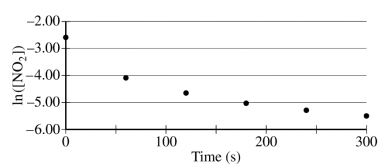
The concentration of a sample of NO₂ (g) is monitored as it decomposes and is recorded on the graph directly below. The two graphs that follow it are derived from the original data.
(a) Explain how the graphs indicate that the reaction is second order.
(b) Write the rate law for the decomposition of NO₂ (g).
These two parts were reviewed in earlier study guides within this unit, but let's break them down. For part a, all you have to do is identify that the graph plotting 1/[NO₂] versus time is linear. To review this concept, be sure to check out our study guide about concentration changes over time.
Part a sample response: The linear graph of 1/[NO₂] vs. time indicates a second-order reaction."
For part b, take a look at the chemical equation, and write the rate law for the decomposition of NO₂. To review this process, check out our study guide introducing the concept of the rate law.
Part b sample response: Rate = k[NO₂]²
Understanding Rate-Determining Steps
The second half of this 2019 FRQ tests your knowledge regarding how to verify a mechanism. Part (c) asks you to consider two possible mechanisms for the decomposition reaction:
(c.i) Is the rate law described by mechanism I shown below consistent with the rate law you wrote in part (b)? Justify your answer.
Part b gave us an experimentally determined rate law, so now we have to figure out what the rate law is of the mechanism above and explain if they are the same. Remember that when looking at a mechanism, we have to use the stoichiometric coefficients of the reactants in the slow step, or the rate-determining step. In this mechanism, the RDS is step 1.
Then, we find the rate law to be R = k[NO₂][NO₂], or Rate = k[NO₂]², which matches the experimentally determined rate law.
Part c.i Sample Response: Yes. Step 1 is slow, therefore it is the rate-determing step of this mechanism. The rate law of this elementary reaction is rate = k[NO₂][NO₂] = k[NO₂]², which is consistent with the second-order rate law in part (b).
Experimental Rate Laws vs. Mechanisms
The reason we look at verifying rate laws rather than finding rate laws directly from mechanisms is that the rate law for a reaction can only be determined experimentally as we saw in previous study guides. Rather, an experimentally determined rate law can help chemists verify a mechanism. If the proposed mechanism does not match the experimental rate law, we know that the mechanism cannot be true!
Using Keq to Find the Rate Law
The second half of part c has to do with intermediates. Let's break it down and take a look.
(c.ii) Is the rate law described by mechanism II shown below consistent with the rate law you wrote in part (b)? Justify your answer.
Using our rules from before, we find the slow step and write out our rate law as R = k[N₂O₄]. Boom, done. Wait... let's take a second look there. If we look at the overall reaction (this was given originally in the FRQ), it is 2NO₂ → 2NO + O₂. Therefore, R = k[N₂O₄] is not the rate law! We can rely on the experimentally derived rate law to be the correct one, especially since this question is asking if the mechanism is correctly representing that rate law.
This can be explained by seeing that N₂O₄ is an intermediate for this mechanism. Since intermediates are not reactants, you cannot have an intermediate in your rate law.
So then, how do we go about finding the rate law? This math involves a topic in chemistry that you most likely learned called equilibrium. The first step is a "fast equilibrium," meaning that the reactants constantly react to form the products, and the products constantly react to form the reactants.
We can use this concept to solve for Keq of step 1: Keq = [N₂O₄]/[NO₂]². Since we are trying to substitute for [N₂O₄] in the slow step's rate law, we should rearrange this Keq expression to equal [N₂O₄]. When doing this, you should get [N₂O₄] = Keq[NO₂]².
This may look confusing, but we are basically trying to find something to replace the intermediate with in the rate law. By setting up the fast equilibrium, we can place reactants into the rate law in place of the intermediate. Since [N₂O₄] = Keq[NO₂]², we can replace the [N₂O₄] in R = k[N₂O₄] with Keq[NO₂]².
This would give us the rate law of R = kKeq[NO₂]², which is consistent with the rate law before. You can kind of ignore the two different k values, as those aren't the point of emphasis in these questions. You can think of them as "k."
For reference, check out the given College Board sample response below:
Part c.ii sample response: Yes. Step 2 is slow; therefore, it is the rate-determining step of this mechanism. The rate law of this elementary reaction is rate = k[N₂O₄]. Because N₂O₄ is an intermediate, it cannot appear in the rate law of the overall reaction. Because Keq = [N₂O₄]/[NO₂]² in step 1, [N₂O₄] = Keq[NO₂]². Then, substituting Keq[NO₂]² for [N₂O₄] in the rate law of step 2 gives rate = (k Keq)[NO₂]², which is consistent with the rate law in part (b).
All given sample responses are provided by the College Board's Scoring Guidelines from the 2019 Exam Administration.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
elementary reaction | A single-step reaction that represents one molecular event in a reaction mechanism, with a specific rate law determined by its molecularity. |
irreversible | A reaction step that proceeds in only one direction and does not reverse under the reaction conditions. |
molecularity | The number of molecules that participate in an elementary step. |
rate law | A mathematical expression that relates the reaction rate to the concentrations of reactants, with each concentration raised to a power (order). |
rate-determining step | The slowest elementary step in a reaction mechanism that controls the overall rate of the reaction. |
reaction mechanism | The sequence of elementary steps that describes how a reaction proceeds at the molecular level. |
Frequently Asked Questions
What is a reaction mechanism and how is it different from just the overall chemical equation?
A reaction mechanism is the step-by-step sequence of elementary reactions (each an elementary step with its own molecularity: unimolecular, bimolecular, termolecular) that shows how reactants become products, including short-lived intermediates and transition states. The overall chemical equation just summarizes starting materials and final products—it doesn’t show the individual collisions, intermediates, or which step is slow. For kinetics on the AP exam, remember: if the first step is rate-limiting (or each elementary step is irreversible), the observed rate law equals the molecularity of that slow elementary step (CED 5.8.A.1). So mechanisms let you derive a rate law (via the rate-determining step, pre-equilibrium or steady-state ideas) while the overall equation does not. For a quick review, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do you figure out the rate law from a reaction mechanism?
If the mechanism’s first step is rate limiting, the overall rate law comes straight from that slow elementary step: write rate = k [reactants in that step]^m where m = molecularity (unimolecular = 1st order, bimolecular = 2nd, termolecular = 3rd). This matches CED 5.8.A.1: the slow step sets the rate law. If the slow step involves an intermediate produced in a fast prior step, use the pre-equilibrium (or steady-state) approximation: write the fast equilibrium expression to solve for the intermediate in terms of stable species, substitute into the slow-step rate expression, and simplify to get the observable rate law. Mention rate constant k will combine constants from those substitutions. For AP exam practice, know to identify elementary steps, molecularity, intermediates, and rate-determining step (Topic 5.8). For extra worked examples, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6), the Unit 5 overview (https://library.fiveable.me/ap-chemistry/unit-5), and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why is the slowest step called the rate limiting step?
The slowest elementary step is called the rate-limiting step because it has the highest activation energy (largest barrier), so it controls how fast the overall reaction can proceed. Think of a multi-step mechanism as a highway with several toll booths: the slowest toll booth sets the traffic flow for the whole trip. Because elementary steps are fast or slow based on collision frequency, molecularity, and activation energy (collision theory/transition state), the overall rate depends on the concentration dependence (molecularity) of that slow step. For mechanisms where the first step is rate limiting, the reaction’s rate law equals the rate expression for that elementary step (CED 5.8.A.1). If earlier steps are fast equilibria, you may need pre-equilibrium or steady-state approximations to relate intermediates to reactants. For more practice and examples (including AP-style problems), see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and the Unit 5 overview (https://library.fiveable.me/ap-chemistry/unit-5).
What's the difference between molecularity and order in rate laws?
Molecularity is a property of a single elementary step: it’s the number of reactant molecules/particles that collide in that elementary step (unimolecular = 1, bimolecular = 2, termolecular = 3). Reaction order is an experimental property of the overall rate law (exponents on concentrations in rate = k[A]^m[B]^n); it can be non-integer and reflects how rate depends on concentrations. For mechanisms where the first step is rate-determining (or an irreversible slow step), the molecularity of that slow elementary step gives the reaction’s rate law and thus the overall order (CED 5.8.A.1). But for multi-step mechanisms with fast pre-equilibria or intermediates, the observed order may differ and must be derived using pre-equilibrium or steady-state approximations. For AP review, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about elementary steps - what makes a step elementary?
An elementary step is a single molecular event—one collision or one unimolecular change—that goes through a single transition state/activated complex and directly shows which species collide to form products or intermediates. You can tell a step is elementary if it’s written as a single-step equation (not a net overall equation) and its stoichiometric coefficients give the molecularity: unimolecular (A → products), bimolecular (A + B → products), or (rare) termolecular. Crucially for AP Chem (CED 5.8.A.1): the rate law for an elementary step is determined by its molecularity, so if the first step is rate-limiting the overall rate law equals that step’s molecularity-based law. Elementary steps produce/interconvert intermediates and can be reversible or irreversible; use pre-equilibrium or steady-state ideas when the slow step isn’t first. For more examples and practice, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and plenty of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain what rate limiting step means in simple terms?
The rate-limiting step (aka rate-determining step) is simply the slowest elementary step in a multi-step reaction mechanism. Think of a reaction pathway like a multi-stage race: the slowest runner sets how fast the whole team finishes. Because that step is slow, it controls the overall reaction rate and (for mechanisms where the slow step is first or where steps are irreversible) the rate law comes from the molecularity of that slow step (unimolecular → first order, bimolecular → second order, etc.). Intermediates appear before/after the slow step but don’t show up in the rate law for a mechanism with a slow first step. On the AP exam, you’ll often be asked to write the rate law from a mechanism (CED 5.8.A.1), so identify the slowest elementary step and use its molecularity. For a deeper refresher, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6). More Unit 5 review and practice problems are at (https://library.fiveable.me/ap-chemistry/unit-5) and (https://library.fiveable.me/practice/ap-chemistry).
How do I identify which step is the slowest in a multi-step mechanism?
Look for the step that actually controls how fast product forms—the slow (rate-determining) step. Practically: - Compare the observed rate law (from experiment) to the molecularity of each elementary step. The slow step’s molecularity sets the rate law when it’s rate-limiting (CED 5.8.A). - On an energy diagram, the slow step has the largest activation energy (highest energy barrier, lowest peak-to-reactant drop). - Mechanistic clues: the slow step often produces or consumes a reaction intermediate that builds up; fast steps reach (quasi) equilibrium. If the first step is slow, rate = k[reactants of step1]. If not, use pre-equilibrium or steady-state approximations to express intermediates and get the overall rate law. - Use collision theory: the rarest/more complex collisions (higher molecularity) are often slower. For AP prep, practice matching mechanisms to rate laws and using pre-equilibrium examples (Fiveable’s Topic 5.8 study guide: https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6). Need more practice? Try the AP practice problems at https://library.fiveable.me/practice/ap-chemistry.
What happens if the first step isn't the rate limiting step?
If the first step isn’t the rate-limiting step, the overall rate is NOT simply given by the molecularity of step 1. Instead you must account for how intermediates form and are consumed: - If an early step is fast and reaches equilibrium before a slower step, use the pre-equilibrium approximation: write the intermediate’s concentration in terms of reactants using the equilibrium constant, then plug that into the slow step’s rate expression to get the overall rate law. - If an intermediate is produced and consumed at comparable rates, use the steady-state approximation: set the intermediate’s net rate of change ≈ 0, solve for its concentration in terms of reactants, then substitute into the slow step rate. AP tip: the CED explicitly covers the simple case where the first step is rate-limiting (5.8.A.1), but exam problems may demand pre-equilibrium or steady-state reasoning—so be comfortable deriving rate laws from mechanisms, not just reading the slow step. For a short refresher and worked examples, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6). For extra practice, check Fiveable’s AP Chem problem set library (https://library.fiveable.me/practice/ap-chemistry).
Why does only the rate limiting step determine the overall rate law?
Because an elementary step is where bonds break/form in one collision, its rate law follows directly from its molecularity (how many reactant particles collide). In a multi-step mechanism the slowest (rate-determining) step acts like a bottleneck: intermediates produced faster in prior steps pile up, but can only be consumed as fast as the slow step allows. So the overall reaction can’t proceed faster than that slow elementary step, and the measured rate law matches the molecularity and concentration dependence of that step. If the first step is slow (the common AP case), you read the rate law straight from that step (CED EK 5.8.A.1). For more complex mechanisms you may need the pre-equilibrium or steady-state approximations to express intermediates in terms of reactants before getting the rate law. For extra practice and examples, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and Unit 5 review (https://library.fiveable.me/ap-chemistry/unit-5).
I don't understand how to write a rate law from the slowest step - can you walk me through it?
Short version: if the mechanism’s slow (rate-determining) step is the first step, the overall rate law equals the rate law for that elementary step—use its molecularity. For an elementary step, write rate = k × concentrations of reactant species shown in that step, each to the first power for each molecule present (unimolecular → first order, bimolecular → second order, termolecular → third order). Example: if mechanism starts with A + B → I (slow), then rate = k[A][B]. If the slow step involves an intermediate (rare in AP CED first-step-limited cases), use the pre-equilibrium or steady-state idea to replace the intermediate with reactant concentrations before writing the final rate law. Remember AP language: “rate-determining step,” “elementary step,” “molecularity,” and don’t include spectators or products from other steps. For more worked examples and CED-aligned practice, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and the unit page (https://library.fiveable.me/ap-chemistry/unit-5).
What's molecularity and how do I find it for each elementary step?
Molecularity = the number of particles (atoms, molecules, or ions) that collide in a single elementary step. For each elementary step, just count the reactant species in that step: - 1 particle → unimolecular (rate ∝ [A]) - 2 particles → bimolecular (rate ∝ [A][B] or ∝ [A]^2) - 3 particles → termolecular (rare; rate ∝ product of three concentrations) Important: molecularity applies only to elementary (single-step) reactions because those show the actual collision event. If a step is written with two reactant molecules, the elementary-step rate law will include those concentrations. Per the AP CED, when the first step is rate-limiting (or steps are irreversible), the overall rate law equals the molecularity of the slowest elementary step (5.8.A.1). If the slow step involves an intermediate or a pre-equilibrium, you may need the pre-equilibrium/steady-state approach. For more practice and examples, check the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and Unit 5 resources (https://library.fiveable.me/ap-chemistry/unit-5). For lots of practice problems, see (https://library.fiveable.me/practice/ap-chemistry).
How do you know if a reaction mechanism is correct or not?
You check a proposed mechanism by seeing if it’s consistent with three things: the experimental rate law, the overall stoichiometry, and any observed intermediates/catalysts. For AP-style mechanisms (CED 5.8.A), if the first step is rate-limiting (slow), the rate law must match the molecularity of that slow elementary step (e.g., bimolecular slow step → rate ∝ [A][B]). If the mechanism uses a fast pre-equilibrium or steady-state for intermediates, you must algebraically eliminate intermediates and show the derived rate law equals the experimental one. Also confirm the sum of elementary steps gives the overall reaction and that predicted intermediates or catalysts make chemical sense. If anything disagrees with the experimental rate law or observed species, the mechanism’s wrong. For practice, see the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and grab more problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do we assume elementary steps are irreversible in these problems?
We assume elementary steps are irreversible in many AP problems because it makes the connection between mechanism and rate law direct: for an elementary, irreversible step the rate law equals its molecularity (e.g., bimolecular → rate ∝ [A][B]). The CED explicitly says if each elementary step is irreversible, or the first step is rate-limiting, the slow step sets the observed rate law (5.8.A.1). If a step is reversible you can’t just read off the rate law—you must use a pre-equilibrium or steady-state approximation to express intermediate concentrations, which adds algebra and extra assumptions. So for exam practice they give irreversible steps (or a clear rate-limiting first step) to test your ability to map molecularity → rate law without extra complexity. Want a quick refresher and examples (including pre-equilibrium cases) check the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and more practice problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the connection between reaction intermediates and rate laws?
Reaction intermediates are species formed in one elementary step and consumed in a later step. Their presence matters for rate laws because the overall rate is determined by the slowest elementary step (rate-determining step). If the slow step produces or consumes an intermediate, the rate law comes directly from that step’s molecularity (e.g., bimolecular slow step → rate ∝ [A][B])—which is exactly what the CED says for mechanisms where the first step is rate limiting (5.8.A). If the slow step isn’t first, you often need the pre-equilibrium or steady-state approximation to express the intermediate’s concentration in terms of reactants so you can write the observable rate law. So intermediates can appear in mechanism sketches but are eliminated when you derive the final rate expression used on the exam. For a focused review, check the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I remember which step controls the rate when there are multiple steps in the mechanism?
Short rule: the slowest (rate-determining) elementary step controls the overall rate—the rate law follows that step’s molecularity. So if a mechanism lists steps and one is labeled “slow,” write the rate law from the reactants of that slow elementary step (unimolecular → first order, bimolecular → second order). Memory tips: - Circle the slow step in the mechanism. That step’s reactant stoichiometry = the rate law (when the slow step is first rate-limiting, per CED 5.8.A). - If no step is marked slow, ask which step has the highest activation energy or produces an intermediate—that’s likely rate-limiting. - If a mechanism gives a fast pre-equilibrium, use the pre-equilibrium approximation to express intermediate concentration in terms of stable species (or use the steady-state approximation). On the AP exam they may explicitly say “first step is rate limiting”—then just use that step’s molecularity (CED 5.8.A). For extra practice, check the Topic 5.8 study guide (https://library.fiveable.me/ap-chemistry/unit-5/reaction-mechanism-rate-law/study-guide/4v22KrcnuKsP5QYwBUu6) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).