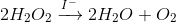What is a Catalyst?
Catalysts lower the activation energy of a reaction. Those who are proficient in biology may recognize catalysts as having a similar function as enzymes. Catalysts are defined as species that are consumed in one step in a reaction mechanism but appear again later. Essentially, they go in as a reactant but come out as a product COMPLETELY UNTOUCHED. They do not play a role in the actual reaction but rather modify the mechanism such that the energy regarding the reaction changes.
🎥 Watch: Catalysis
This is shown through the notation of a catalyst above the reaction arrow, as seen in the following image.
The equation can be read: "The decomposition of hydrogen peroxide into water and oxygen through catalyzation by iodide." The reaction is typically done with potassium iodide, though the iodide ion is the catalyst.

H2O2 actually decomposes slowly, but when we add a catalyst, the reaction speeds up—leading to the "Elephant's Toothpaste" reaction.
Catalysts and Energy
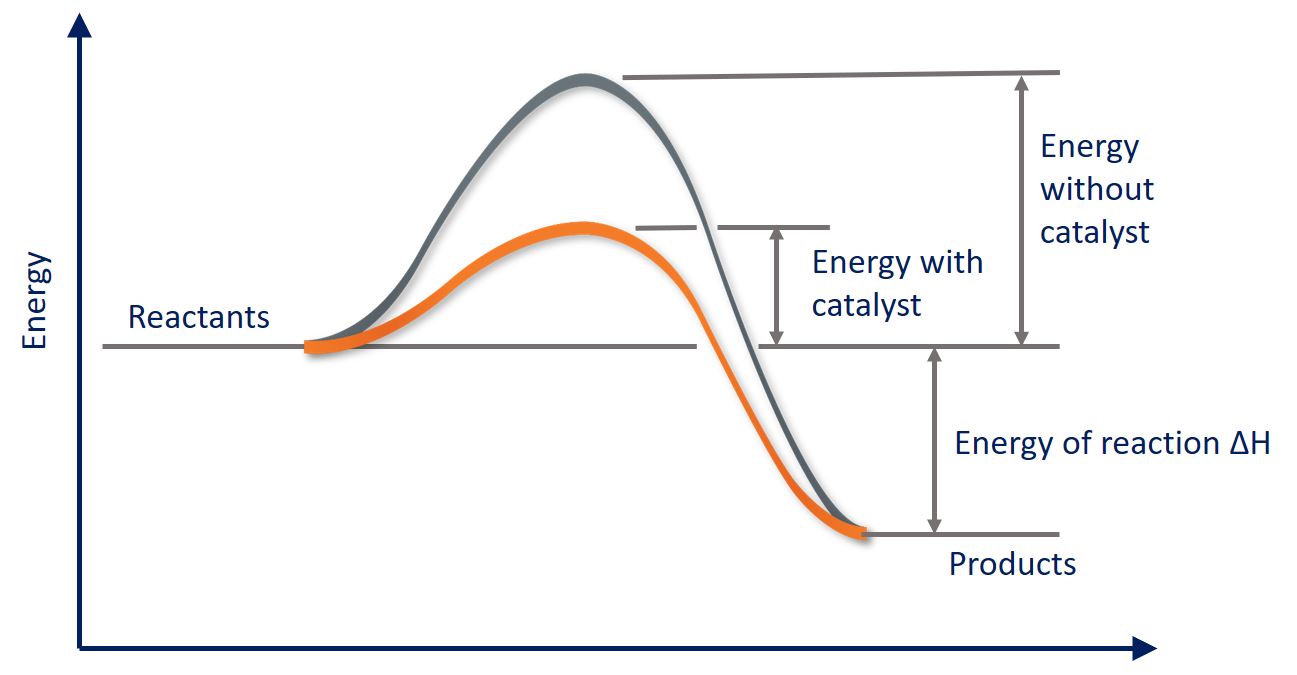
As we saw earlier, a catalyst functions by lowering the Ea (activation energy) for a reaction. Let's take a look at how this works graphically:
We know that the activation energy is the energy between the reactants and the activated complex (the hump). Then, we see that the addition of a catalyst brings down the curve to lower the activation energy. Catalysts may also split a one-step high Ea into a multi-step, low Ea reaction:
Images Courtesy of QuoraCatalysts and Mechanisms
Although it is less important to know than the thermodynamic effects, catalysts also change the way a mechanism works! We can see this in two mechanisms for the decomposition of H2O2, one being catalyzed and the other with no catalysis.
Non-catalyzed Mechanism Catalyzed MechanismThe catalyzed mechanism has two steps, whereas the uncatalyzed mechanism has three steps. By eliminating a step, the catalyzed reaction occurs more quickly.
In order for catalysts to increase the rate of a reaction, the addition of the catalyst must increase the number of effective collisions and/or provide a reaction path with a lower activation energy relative to the original reaction. How exactly do catalysts do this?
A common mechanism of catalysts to accelerate reactions is by binding to the reactants. The reactants are either oriented more favorably or react with lower activation energy in the presence of the catalyst, similar to the function of enzymes.
Acid-Base Catalysis
Acid-base catalysis involves a reactant or intermediate gaining or losing a proton, which fundamentally changes the reaction mechanism. This type of catalysis is crucial in many organic and biochemical reactions.
Key features of acid-base catalysis:
- Proton transfer creates new intermediates: When a reactant gains H⁺ (protonation) or loses H⁺ (deprotonation), it forms a new chemical species
- New elementary reactions are introduced: These new intermediates participate in different elementary steps than the uncatalyzed reaction
- The activation energy is lowered: The protonated/deprotonated intermediates are more reactive
- The catalyst is regenerated: The acid or base that donated/accepted the proton is restored at the end
Example: Acid-catalyzed ester hydrolysis mechanism
- Elementary reaction 1: H⁺ + RCOOR' → RCOO⁺HR' (protonation creates new intermediate)
- Elementary reaction 2: RCOO⁺HR' + H₂O → RC(OH)₂⁺OR' (nucleophilic attack on activated intermediate)
- Elementary reaction 3: RC(OH)₂⁺OR' → RCOOH + HOR' + H⁺ (products form and catalyst regenerated)
Notice how the protonated ester (RCOO⁺HR') is a new intermediate that doesn't exist in the uncatalyzed reaction. This intermediate is more electrophilic and reacts more readily with water, lowering the overall activation energy.
Surface Catalysis
In surface catalysis, reactions occur on the surface of solid catalysts through a process more complex than simple adsorption. Reactants or intermediates can form covalent bonds with the surface, creating entirely new chemical species.
Key mechanisms in surface catalysis:
- Covalent bond formation: Reactants don't just stick to the surface - they form actual chemical bonds with surface atoms
- New bound intermediates: These surface-bound species are new reaction intermediates with different reactivity
- New elementary reactions: The mechanism includes elementary steps involving these surface-bound intermediates
- Surface as active participant: The catalyst surface atoms are temporarily part of the reacting molecules
Example: Haber-Bosch ammonia synthesis on iron catalyst
- Elementary reaction 1: N₂(g) + 2Fe(surface) → 2N-Fe(surface) (N₂ forms covalent bonds with Fe atoms)
- Elementary reaction 2: H₂(g) + 2Fe(surface) → 2H-Fe(surface) (H₂ forms covalent bonds with Fe atoms)
- Elementary reaction 3: N-Fe(surface) + H-Fe(surface) → NH-Fe(surface) + Fe(surface)
- Elementary reaction 4: NH-Fe(surface) + H-Fe(surface) → NH₂-Fe(surface) + Fe(surface)
- Elementary reaction 5: NH₂-Fe(surface) + H-Fe(surface) → NH₃(g) + 2Fe(surface)
The N-Fe and H-Fe species are new intermediates where nitrogen and hydrogen atoms are covalently bonded to iron atoms on the catalyst surface. These surface-bound intermediates have different activation energies for reaction than free N₂ and H₂ molecules, enabling the reaction to proceed at reasonable temperatures.
Importantly, although the catalyst is frequently consumed by the rate-determining step, the net catalyst concentration remains constant. Therefore, the catalyst is always regenerated in catalysis mechanisms!
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
acid-base catalysis | A catalytic mechanism in which a catalyst facilitates a reaction by transferring a proton to or from a reactant or intermediate. |
activation energy | The minimum energy required for reactants to overcome the energy barrier and proceed to products in a chemical reaction. |
catalyst | A substance that increases the rate of a chemical reaction without being consumed in the reaction. |
covalent bonding | A chemical bond formed by the sharing of electrons between atoms. |
effective collisions | Collisions between reactant molecules that occur with sufficient energy and proper orientation to result in a reaction. |
elementary reaction | A single-step reaction that represents one molecular event in a reaction mechanism, with a specific rate law determined by its molecularity. |
enzyme | A biological catalyst that speeds up chemical reactions by binding to reactants and lowering the activation energy. |
rate-determining step | The slowest elementary step in a reaction mechanism that controls the overall rate of the reaction. |
reaction coordinate | A diagram or pathway showing the energy changes that occur as reactants are converted to products during a reaction. |
reaction intermediate | A species that is produced in one elementary step of a reaction mechanism and consumed in a subsequent step, not appearing in the overall reaction. |
reaction mechanism | The sequence of elementary steps that describes how a reaction proceeds at the molecular level. |
surface catalysis | A catalytic process in which reactants or intermediates bind to or form covalent bonds with a solid surface, creating new reaction pathways. |
Frequently Asked Questions
What is a catalyst and how does it actually make reactions go faster?
A catalyst is a substance that speeds a reaction without being consumed overall—it provides an alternative reaction mechanism with a lower activation energy or increases the number of effective collisions (CED 5.11.A.1–A.2). Practically, that means the catalyst often forms a new intermediate (binds reactants or donates/accepts a proton) so the rate-determining step needs less energy. Enzymes bind and orient substrates at an active site (transition-state stabilization), acid–base catalysts make or remove H+ (covalent steps), and surface (heterogeneous) catalysts adsorb reactants so they react more easily (CED 5.11.A.3–A.5). The catalyst’s net concentration stays constant because it’s regenerated later in the mechanism. For AP exam prep, be ready to explain how a catalyst changes the mechanism and lowers Ea (you might get that in a free-response or principle question). Review examples and practice problems on the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and more Unit 5 resources (https://library.fiveable.me/ap-chemistry/unit-5).
Why doesn't a catalyst get used up in a reaction if it's helping the reaction happen?
A catalyst speeds a reaction by providing a different mechanism with a lower activation energy or better reactant orientation—so more collisions become “effective” (CED 5.11.A.1). That doesn’t mean the catalyst vanishes: in catalytic mechanisms the catalyst may be consumed in one elementary step (even the rate-determining step) but is regenerated in a later step. So the net amount of catalyst stays constant over the overall reaction—you get a catalytic cycle (CED 5.11.A.2–A.5; keywords: catalyst regeneration, reaction intermediate, active site, turnover). Enzymes and surface catalysts work this way: they form intermediates or bind reactants temporarily, then release products and return to their original form. For AP exam answers, explicitly state the lowered activation energy or changed mechanism and that the catalyst is regenerated (tie to 5.11.A). For a quick refresher and practice problems on this topic, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and Unit 5 resources (https://library.fiveable.me/ap-chemistry/unit-5); thousands of practice questions are at https://library.fiveable.me/practice/ap-chemistry.
I'm confused about activation energy - how do catalysts lower it exactly?
Think of activation energy (Ea) as the energy hill reactants must climb to become products. A catalyst lowers Ea by giving the reaction a different route with smaller hills—usually by forming new intermediates or stabilizing the transition state. Mechanistically that means the catalyst participates in one or more elementary steps (often binding to reactants, donating/accepting H+ in acid–base catalysis, or adsorbing molecules on a surface), creating lower-energy transition states and then getting regenerated by the end (net catalyst concentration stays constant). Catalysts speed the approach to equilibrium but don’t change ΔH or the equilibrium constant. On the AP exam you should be able to explain this in terms of reaction mechanism, show an energy diagram with a lower peak for the catalyzed path, and mention catalyst regeneration (CED 5.11.A). For a focused review, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and more Unit 5 review (https://library.fiveable.me/ap-chemistry/unit-5). For practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between homogeneous and heterogeneous catalysts?
Homogeneous catalysts are in the same phase as the reactants (usually all in solution). They speed reactions by providing an alternate mechanism with lower activation energy—often forming a bound intermediate, participating in acid–base or covalent steps, and then being regenerated (net [catalyst] stays constant). Enzymes are a common biological example. Heterogeneous catalysts are in a different phase (usually a solid surface with gaseous or liquid reactants). They work by adsorption—reactants bind to the surface, are oriented or activated, form surface-bound intermediates, and then desorb as products. Surface catalysis introduces new elementary steps (Langmuir–Hinshelwood or Eley–Rideal models) and can suffer catalyst poisoning. Both types lower Ea and increase effective collisions; both follow catalytic cycles with regeneration. For an AP review, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and more unit resources (https://library.fiveable.me/ap-chemistry/unit-5). Practice problems are available at (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain enzyme catalysis in simple terms?
An enzyme is just a protein catalyst that speeds a reaction by giving reactants a different, easier path—usually lowering the activation energy (Ea) and stabilizing the transition state. Practically, the enzyme’s active site binds substrates so they’re held in the right orientation (or undergo “induced fit”), which increases effective collisions and/or lowers Ea. In a catalytic mechanism the enzyme may form a temporary intermediate (it can be “used” in a step) but is regenerated overall, so its net concentration stays constant (CED 5.11.A.1–A.3). Enzymes also can do acid–base or covalent catalysis and often show saturation behavior described by Michaelis–Menten kinetics (turnover number/frequency tell you how fast one enzyme cycles). For AP exam focus on: catalysts change mechanism and lower Ea, catalysts are regenerated, and enzymes often bind substrates to stabilize transition states (Topic 5.11). Review the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and get extra practice at (https://library.fiveable.me/practice/ap-chemistry).
How do catalysts change the reaction mechanism without changing the overall reaction?
A catalyst speeds a reaction by providing a different reaction mechanism—basically new elementary steps—that have lower activation energy or increase effective collisions, but it doesn’t change the overall reactants → products stoichiometry or ΔH. Practically that means: the catalyst is often temporarily bound (forming a new intermediate), may be consumed in a step that’s later reversed, and is regenerated by the end (net [catalyst] stays constant)—this is a catalytic cycle (CED 5.11.A.2–A.3). Types include homogeneous (acid–base or covalent catalysis), enzyme binding/transition-state stabilization, and heterogeneous surface catalysis (adsorption + surface reactions). Because the mechanism changes, the rate-determining step can change too, which is how the rate increases without changing overall reaction identity (CED 5.11.A.1, A.5). For AP study, be ready to explain mechanism changes, catalyst regeneration, and how activation energy is lowered (see the Topic 5.11 study guide: https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when they say a catalyst is "regenerated" in the mechanism?
“Regenerated” means the catalyst appears as a reactant in one elementary step (it’s used up or bound to an intermediate) but is produced again in a later step so its net concentration doesn’t change over the whole mechanism. In other words, the catalyst participates in a catalytic cycle: it forms new intermediates or lowers the activation energy (so more effective collisions / lower Ea per 5.11.A.1) but is returned to its original form at the end (5.11.A.2). That’s why catalysts speed up how fast equilibrium is reached without changing the equilibrium position. Watch for elementary steps that show the catalyst bonded to reactants (enzyme-style or surface adsorption) and then freed in a later step—that’s regeneration. For more examples and AP-aligned practice, check the Topic 5 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and tons of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to know about surface catalysis for the AP exam?
You need to know surface catalysis because the AP exam asks you to explain how catalysts change a reaction’s mechanism and rate (CED 5.11.A). Surface (heterogeneous) catalysts bind reactants to a surface (adsorption), form new bound intermediates, introduce new elementary steps, and usually lower the activation energy or increase effective collisions—so the rate increases even though the net catalyst concentration stays constant (keywords: activation energy, reaction intermediate, catalyst regeneration, heterogeneous catalysis). Be ready to describe how a catalyst can be consumed and then regenerated in a mechanism and to name simple surface mechanisms (Langmuir–Hinshelwood vs. Eley–Rideal) or show that binding/orientation stabilizes the transition state. Unit 5 is tested on the exam (7–9% of MCQs), so practice explaining these ideas clearly in words and with short mechanism steps. For a focused review, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and try related practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how acid-base catalysis works - what's happening with the protons?
Acid–base catalysis just means protons (H+) are moved around to make a faster pathway—the catalyst donates or removes a proton to form a new intermediate with a lower activation energy, then is regenerated. In general acid catalysis, an acid donates H+ to a reactant or transition state, stabilizing negative charge (or making a leaving group easier to lose). In general base catalysis, a base removes an H+, making a nucleophile stronger or stabilizing a positive charge in the transition state. Mechanistically that introduces extra elementary steps (you’ll often see a protonation step, a bond-making/breaking step that’s faster, then a deprotonation step that regenerates the catalyst). This fits the CED: the catalyst changes the mechanism, creates an intermediate, and is present at constant net concentration (5.11.A.2–5.11.A.4). For worked explanations and practice, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and Unit 5 resources (https://library.fiveable.me/ap-chemistry/unit-5). Practice more with problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between a catalyst binding to reactants versus forming covalent bonds with them?
Binding (noncovalent) vs forming covalent bonds is mostly about how the catalyst interacts with reactants and what new intermediates appear in the mechanism. When a catalyst binds to reactants noncovalently (e.g., enzymes using H-bonds, ion-dipole, van der Waals), it orients/stabilizes the transition state and lowers Ea without making a new chemical bond to the substrate—you usually get a reversible bound intermediate and the catalyst is regenerated (CED 5.11.A.1, 5.11.A.3). When the catalyst forms a covalent bond (including acid–base catalysis or a reactant forming a bond to a surface), the catalyst actually becomes part of a new chemical intermediate; that intermediate undergoes further steps and the catalyst is restored later (CED 5.11.A.4–5). Both lower activation energy and change the mechanism; both keep net [catalyst] constant over the full catalytic cycle. For more examples and AP-style practice, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and Unit 5 overview (https://library.fiveable.me/ap-chemistry/unit-5). You can practice problems at (https://library.fiveable.me/practice/ap-chemistry).
How do I identify the catalyst in a reaction mechanism on a multiple choice question?
Look for any species that appears in the mechanism steps but is present unchanged in the overall (net) reaction—that’s your catalyst. Key clues from the CED: a catalyst may be consumed in an intermediate (often in the RDS) but must be regenerated later, so its net concentration is unchanged (5.11.A.2). Practically on an MCQ: - Scan the elementary steps: if X is used in step 1 and formed again in step 3, X is likely the catalyst. - Compare the mechanism to the overall equation: species that appears in the mechanism but cancels out in the net equation = catalyst. - Watch for new bound intermediates (X–A) or proton transfers—those often signal homogeneous or acid/base catalysis (5.11.A.3–A.4). - For heterogeneous catalysis, look for species binding to a surface or “adsorbed” label (5.11.A.5). On the exam, they’ll test that the catalyst lowers activation energy by providing an alternate pathway and is regenerated (Topic 5.11). For more examples, review the Topic 5 catalysts study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and try practice questions (https://library.fiveable.me/practice/ap-chemistry).
What are reaction intermediates and how are they different from catalysts?
A reaction intermediate is a species that’s formed in one elementary step of a mechanism and then consumed in a later step—it appears in the mechanism but not in the overall balanced equation. A catalyst, by contrast, also appears in the mechanism but is consumed and then regenerated so its net concentration stays constant (it’s present before and after the reaction). Both can lower the activation energy by providing a different pathway (CED 5.11.A.1), and catalysts often create new intermediates by binding to reactants (enzymes, surface or acid–base catalysis; CED 5.11.A.3–5). Key differences: intermediates are transient and not returned to their original form; catalysts are returned (catalyst regeneration, CED 5.11.A.2). For more examples and AP-aligned practice, check the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru), the Unit 5 overview (https://library.fiveable.me/ap-chemistry/unit-5), and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do enzymes work so much better than regular catalysts?
Enzymes work way better than most “regular” catalysts because they change the mechanism in very specific, powerful ways (CED 5.11.A). Their active site binds substrates tightly so reactants are oriented correctly, held close, and put into a geometry that stabilizes the transition state—all of which lowers activation energy a lot (transition state stabilization, effective collisions). Many enzymes use acid–base or covalent steps and form short-lived intermediates, then regenerate the catalyst in the catalytic cycle ( catalyst regeneration, rate-determining step). Two big advantages: extreme specificity (only certain substrates fit) and very high turnover numbers/turnover frequency, so one enzyme processes thousands of substrate molecules per second. You’ll see this idea under enzyme catalysis and Michaelis–Menten kinetics in Topic 5.11 (see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and Unit 5 overview (https://library.fiveable.me/ap-chemistry/unit-5)). For extra practice on kinetics and catalysis, check the 1000+ practice problems (https://library.fiveable.me/practice/ap-chemistry).
I missed the lab on catalysis - what happens when you add a catalyst to hydrogen peroxide?
If you add a catalyst (like MnO2 or the enzyme catalase) to hydrogen peroxide, the decomposition reaction 2 H2O2 → 2 H2O + O2 speeds up dramatically. The catalyst provides an alternative mechanism with a lower activation energy (so more collisions are effective), often forming short-lived intermediates or binding the reactant at an active site, and then is regenerated (net catalyst concentration stays the same). You’ll see rapid bubbling as O2 is released. Important: a catalyst changes the rate and mechanism but does not change ΔH or the reaction’s equilibrium position. This is exactly the Topic 5.11 idea—transition-state stabilization, catalyst regeneration, and different elementary steps (heterogeneous surface catalysis for MnO2; enzyme catalysis for catalase). For a quick recap of the AP concepts and keywords (activation energy, mechanism, catalyst regeneration, surface vs. enzyme catalysis), check the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru). For extra practice on kinetics and mechanisms, use the AP Chemistry practice set (https://library.fiveable.me/practice/ap-chemistry).
How do I draw energy diagrams showing the effect of a catalyst on activation energy?
Draw a reaction coordinate (x-axis = reaction progress, y-axis = potential energy). Sketch two curves from same reactants to same products so products are lower or higher depending on ΔH you want. - Uncatalyzed: one high peak = transition state; label Ea (energy difference from reactants to peak). - Catalyzed: draw a lower peak (or two smaller peaks if the catalyst creates a new intermediate/step). Label the new, smaller Ea(s). Always label: reactants, products, ΔH (same vertical difference for both curves), and “catalyst regenerated” if you show an intermediate. Add a note: catalyst lowers activation energy by stabilizing the transition state or forming a bound intermediate (mechanism change), but it doesn’t change ΔH or equilibrium. That matches CED EK 5.11.A.1–A.3 and catalyst regeneration A.2. For more examples and practice drawing multi-step catalytic cycles, see the Topic 5.11 study guide (https://library.fiveable.me/ap-chemistry/unit-5/catalysts/study-guide/bkTgdolcJRgD7fG434Ru) and Unit 5 overview (https://library.fiveable.me/ap-chemistry/unit-5). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).