Atoms are the most basic unit of matter. When several atoms interact with each other, they can form molecules. Depending on how the electrons of these atoms interact, a covalent bond or an ionic bond could form.
Everything in chemistry strives to become the most stable possible. Atoms do the same thing! Atoms bond in order to reach a more stable, lower energy state.
Principles of Bonding

Valence Electrons
Valence electrons are typically involved in bonding, not core electrons! They play a crucial role in the bonding process and determine the chemical properties of the resulting molecule. Here is a quick review of everything we learned about valence electrons in unit one:
- Valence electrons are the outermost electrons in an atom.
- Valence electrons are found in the s and p orbital of the outermost occupied electron shell.
- A gap in ionization energies could tell us how many valence electrons an element has.
- Elements in the same group on the periodic table have the same number of valence electrons and therefore form similar molecules with other elements.
Electronegativity
As we discussed in unit one, electronegativity is one of the five periodic trends you should be familiar with. Here is what you should know before learning about bonding:
- Electronegativity of a measure of an atom's ability to attract electrons in a bond.
- Electronegativity values increase as you go from left to right across a period on the periodic table ➡️. This trend can be explained by the increasing atomic numbers across a period. The more protons there are in the nucleus of an atom, the stronger the nucleus' positive charge is. Nuclei with more protons are more effective at attracting electrons since opposite charges attract.
- Electronegativity values decrease as you go down a group on the periodic table ⬇️. This trend is explained by the increasing atomic radius of the elements down a group. The larger the atom, the more distance between the nucleus and the electrons, weakening the attraction.
✨ Coulomb's Law
Coulomb's Law comes in handy when you want to measure the attraction between two atoms. It states that the strength of forces that hold atoms together depends on two factors:
- Magnitude of charge: The greater the charge, the stronger the attraction.
- Distance between the nuclei of the particles: The closer the two particles, the stronger the attraction. This relates directly to the trend of electronegativity and basically explains it! As you go down a group, the distance between the valence electrons and the nucleus increases, which decreases the attractive force, and therefore, the atom's electronegativity. Think about it! 🙃
👉 Check out our study guide on valence electrons and ionic compounds to review more about bonding before getting into all the details!
Ionic Bonding
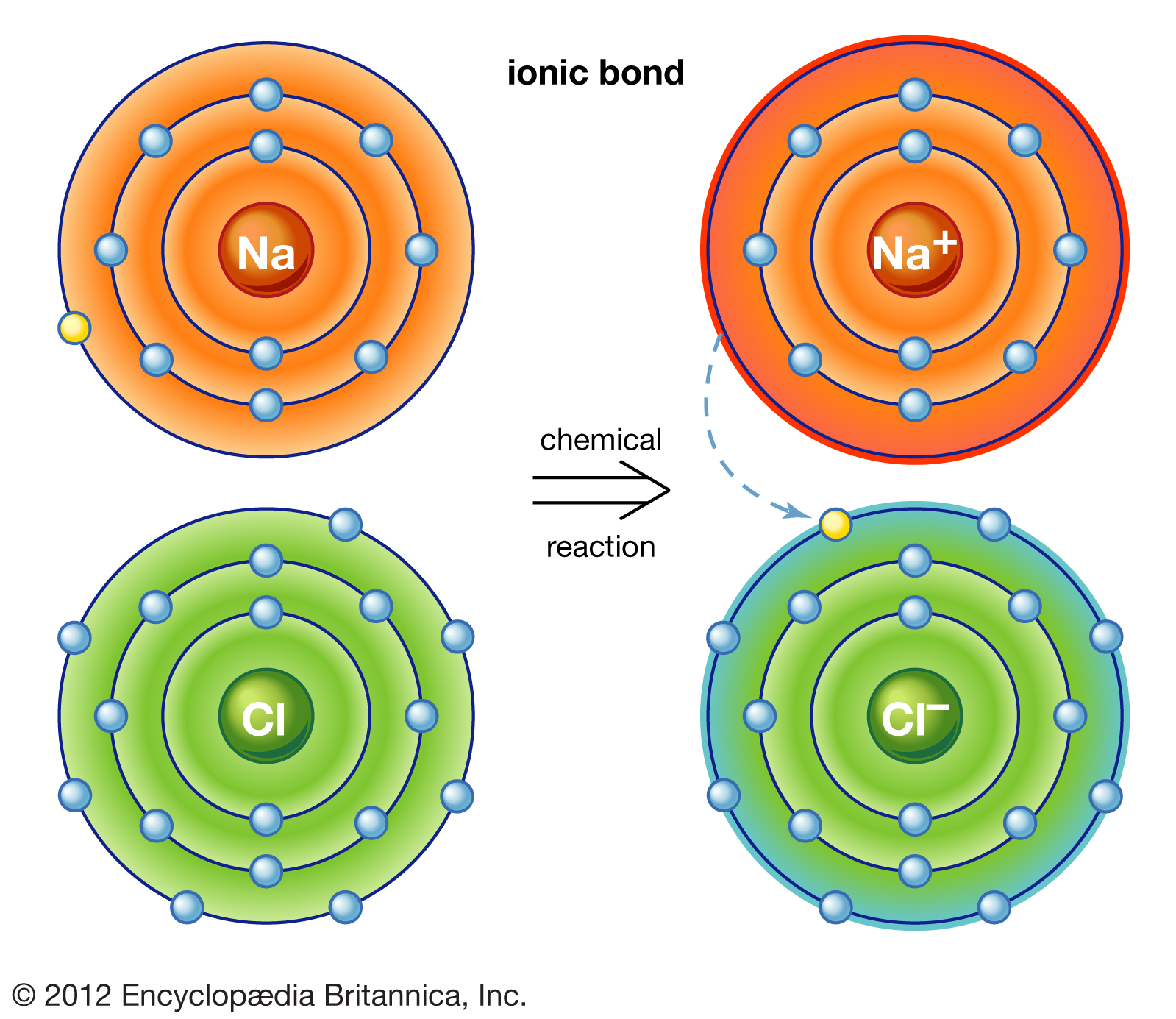
Ionic bonds are formed by the transfer of valence electrons from atom to atom, usually from a metal to a nonmetal. Let's take a look at an example of an ionic bond between a sodium and chloride atom!
Na(s) + ½ Cl2(g) → NaCl(s)
NaCl, a brittle salt with a high melting point was formed in this chemical reaction. Ionic bonds are held together not by shared electrons or a direct bond, but rather through electromagnetic forces that hold positive and negative ions together. These electromagnetic forces are so strong that it takes lots of energy to break them apart, hence the high boiling and melting points.
Ionic compounds also form a crystal lattice of ions, giving them their rigidity. In a crystal lattice, ions are arranged in a repeating, three-dimensional pattern. This crystal lattice is, again, held by strong electrostatic forces. This characteristic of ionic molecules makes them generally good electrical conductors. When an ionic compound is melted or dissolved in water, the ions can move freely within the crystal lattice structure, producing electricity!
Note that when sodium and chlorine interact to form an ionic bond, sodium gives a valence electron to chlorine. This gain and loss of an electron produces ions, hence the name "ionic bond." The atom that loses an electron, sodium, will gain a positive charge and is called a cation. The atom that gains an electron, chlorine, will gain a negative charge and is called an anion.
Applying Coulomb's Law
Remember that Coulomb's Law states that greater charges and smaller distances lead to the strongest attractions.
When asked which ionic compound would have a higher melting point, always look for differences in charge and size. The higher the charge of the ion, the stronger the negative-positive attraction is and the more energy it takes to break the bond. This increases both the melting and boiling point. Same goes for size!
TIP - Always look for differences in charge first; they have a greater impact on melting points.
Examples with Ionic Compounds and Melting Points
- Which compound would have a higher melting point: MgF2 or NaF?Looking at the charge first, you would notice that Mg has a +2 charge, while Na only has a +1 charge. This automatically means that MgF2 has a higher melting point than NaF.
- Which ionic compound would have a higher melting point: LiF or NaBr?Since both charges are the same (+1 / -1), the main difference would have to be the size of the ions. Keeping the periodic trends in mind, lithium and fluorine are much smaller ions than sodium and bromine. Therefore, LiF must have a higher melting point. Lithium and Fluorine are in period 2, while Na is in period 3 and Br is in period 4. Remember, when you go down on the periodic table, the atomic radii increase since there are more occupied electron shells. You may be asked to give a reason for this trend in free response questions!
Covalent Bonding
In covalent bonds, electrons are shared between two or more atoms (typically nonmetals). There are two different types of covalent bonds based on the electronegativities of the atoms involved:
- If a polar covalent bond is formed, there is an unequal distribution of charge.
- If a nonpolar covalent bond is formed, there is an equal distribution of charge.
We'll learn more about polarity when we get into molecular geometry later in this unit.
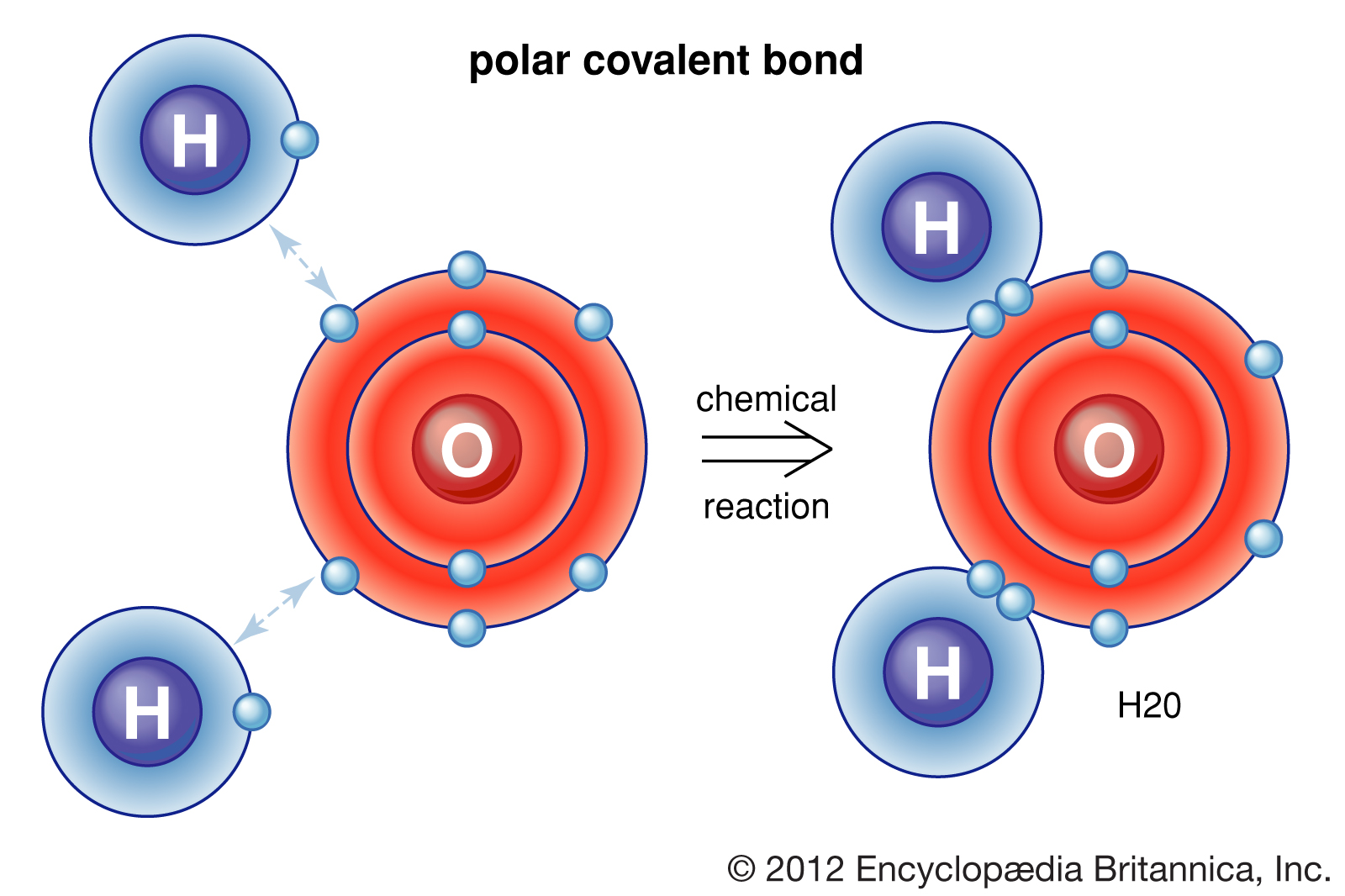
Example of Polar Covalent Bond - H2O
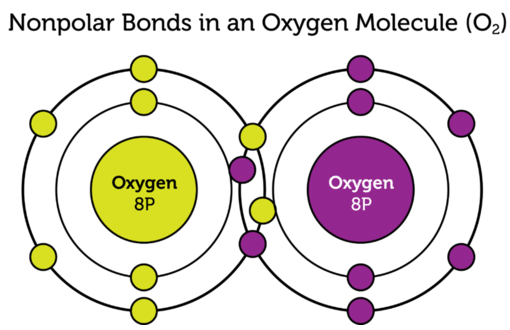
We could see two O-H polar covalent bonds here! These attractions are very strong and form water which, as you know, is essential for life on Earth.Example of Nonpolar Covalent Bond - O2
Image Courtesy of cK-12; In this oxygen molecule, there is a nonpolar covalent bond between the two oxygen atoms!Distinguishing Between Nonpolar & Polar Covalent Bonds
To analyze the difference between the two types of covalent bonds, we would need to look at electronegativity. Valence electrons shared between atoms of similar electronegativity constitute a nonpolar covalent bond. This is why the two oxygen atoms above form a nonpolar covalent bond. Since the electronegativity is the same, the oxygen nuclei pull on the other oxygen atom's electrons with the same strength. Think "nonpolar = balance!"
Valence electrons shared between atoms of unequal electronegativity constitute a polar covalent bond, like in a water molecule. Hydrogen has an electronegativity of 2.2 while oxygen has an electronegativity of 3.44. Therefore, oxygen attracts electrons more strongly and will pull the electrons towards it.
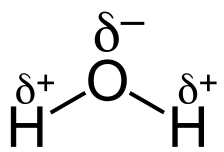
This unequal distribution of charge leads to oxygen developing a partial negative charge. In fact, all polar bonds have some ionic character - the electrons are partially transferred from one atom to another. The difference between ionic and covalent bonding isn't black and white but exists on a continuum. As the electronegativity difference increases, bonds become more ionic in character. This difference in electronegativity leads to bond dipoles, which are covered more in the next unit.
For now, just remember that greater differences in electronegativity within a bond lead to greater bond dipoles and more ionic character!
Image Courtesy of Socratic; δ, or lowercase delta, represents a partial positive or partial negative charge.Metallic Bonding
So far we've covered ionic bonds (electron transfer) and covalent bonds (electron sharing), but there's a third type of bonding that occurs in metals: metallic bonding. In metallic bonds, valence electrons are not transferred or shared between specific atoms. Instead, they form a "sea" of delocalized electrons that can move freely throughout the metal structure.
Think of it this way: metal atoms release their valence electrons into a shared pool, creating positively charged metal cations surrounded by a mobile "electron sea." These delocalized electrons aren't stuck between any two atoms - they can flow throughout the entire metal sample! This unique arrangement explains many metallic properties:
- Electrical conductivity: Those mobile electrons can carry electric current easily
- Malleability and ductility: Metal atoms can slide past each other without breaking bonds because the electron sea adjusts
- Metallic luster: Free electrons can absorb and re-emit light, creating that characteristic shine
- Thermal conductivity: Mobile electrons transfer kinetic energy efficiently
The strength of metallic bonds varies with the number of valence electrons contributed and the size of the metal atoms. Transition metals often have particularly strong metallic bonds because they can contribute more electrons to the delocalized "sea."
Which Chemical Bond will Form?
Here are some principles of ionic and covalent bonds that you should look out for when deciding what chemical bond will form:
- Ionic Bonds
- Form between two elements that have an electronegativity difference of >1.7
- Usually form between a metal and a nonmetal
- Join a cation (positive ion) and an anion (negative ion)
- Covalent Bonds
- Usually form between two nonmetals
- Polar covalent bonds form between two elements that have an electronegativity difference of 0.4-1.7
- Nonpolar covalent bonds form between two elements that are usually the same, but the electronegativity difference is usually between 0-0.4. Think about the nonpolar covalent bond between C-H. The electronegativity difference falls within this range.
- Metallic Bonds
- Form between metal atoms of the same or different elements
- Involve delocalized valence electrons
- Create the characteristic properties of metals
It's important to remember that while electronegativity differences provide useful guidelines, they're not the only factor in determining bond type. The best way to characterize the type of bonding is by examining the properties of the compound itself. For instance, a compound that conducts electricity when dissolved in water but not as a solid likely has ionic bonding, while a compound that never conducts electricity probably has covalent bonds.
Characteristics of Different Bond Types
If a solid has a high melting point and is a good conductor of heat and electricity when dissolved in water, it is most likely an ionic compound. This is because of the concept of a free-flowing ion that generates electricity.
If a solid has a low melting point and doesn't conduct electricity in any state, it is most likely a molecular compound (which has covalent bonds).
If a solid conducts electricity in its solid state, has a shiny appearance, and can be hammered into sheets or drawn into wires, it's a metal with metallic bonding. The delocalized electrons allow for conductivity even when the atoms are in fixed positions!
There is one more circumstance: If a solid has a high melting point and doesn't conduct electricity in any state, it is a network solid made up of covalent bonds. Don't worry about this yet, it's covered in future units :).
Remember that these categories represent idealized bonding types. Real compounds often exhibit characteristics that fall somewhere between these extremes, reflecting the continuous nature of chemical bonding rather than distinct categories.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
bond dipole | The separation of partial positive and negative charges in a polar bond; greater electronegativity differences lead to greater bond dipoles. |
covalent bond | A chemical bond typically formed between two nonmetals where valence electrons are shared between atoms. |
delocalized electrons | Valence electrons in a metallic solid that are not associated with any individual atom but are distributed throughout the structure. |
electronegativity | A measure of an atom's ability to attract valence electrons in a chemical bond; increases across a period and decreases down a group in the periodic table. |
ionic bond | A chemical bond typically formed between a metal and nonmetal where electrons are transferred from one atom to another. |
ionic character | The degree to which a bond exhibits properties of an ionic bond; all polar bonds possess some ionic character. |
metallic solid | A solid composed of metal atoms where valence electrons are delocalized and not associated with any individual atom. |
nonpolar covalent bond | A covalent bond between atoms of similar electronegativity where valence electrons are shared relatively equally. |
partial negative charge | The negative charge that develops on the more electronegative atom in a polar covalent bond. |
polar covalent bond | A covalent bond between atoms of unequal electronegativity where valence electrons are shared unequally, resulting in partial charges on the atoms. |
valence electrons | Electrons in the outermost shell of an atom that participate in bonding and determine many properties of substances. |
Frequently Asked Questions
What is electronegativity and how does it affect chemical bonding?
Electronegativity is an atom’s tendency to attract shared valence electrons in a bond. On the periodic table it increases left → right and decreases top → bottom (explainable with the shell model and Coulomb’s law: more protons and smaller radius → stronger attraction). Electronegativity differences determine bond character: when atoms have very similar values you get nonpolar covalent bonds (e.g., C–H is effectively nonpolar); moderate differences give polar covalent bonds where the more electronegative atom gains a partial negative charge and a bond dipole; very large differences push the bond toward ionic character. Remember the AP CED idea that bonding is a continuum, not a strict split, and that metal–nonmetal pairs are generally ionic while nonmetal–nonmetal pairs are covalent (LO 2.1.A). For metallic solids valence electrons are delocalized (metallic bonding). For more AP-aligned review, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and lots of practice questions at (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about the difference between ionic, covalent, and metallic bonds - can someone explain?
Think of bonding as how valence electrons are shared or transferred between atoms. - Ionic: a (usually) metal + nonmetal where one atom gives electrons to another, forming ions. Large differences in electronegativity and strong Coulombic attraction make an ionic lattice. Properties: high melting points, brittle crystals, conduct electricity only when molten or dissolved (ions mobile). Use lattice energy and experimental properties to judge bonding (CED 2.1.A.4, keywords: lattice energy). - Covalent: two nonmetals share valence electrons. If electronegativities are almost equal, the bond is nonpolar; if unequal, it’s polar with partial charges and a bond dipole (CED 2.1.A.2–2.1.A.3). Properties: lower melting/boiling points (molecular), poor electrical conductors; polarity affects solubility and intermolecular forces. - Metallic: metal atoms contribute delocalized valence electrons to an electron “sea” (CED 2.1.A.5). Properties: electrical/thermal conductivity, malleability, variable melting points. Remember bonding is a continuum (ionic character vs covalent). For more AP-aligned review, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and the Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2). Practice tons of problems at (https://library.fiveable.me/practice/ap-chemistry)—exam questions often ask you to connect bonding type to observable properties.
Why does electronegativity increase across a period but decrease down a group?
Electronegativity rises across a period because atoms gain protons (higher nuclear charge) while adding electrons to the same shell. With the same principal energy level, the increased positive charge pulls valence electrons in more tightly (Coulomb’s law: force ∝ q1q2/r^2), so atoms more strongly attract bonding electrons (CED 2.1.A.1). Electronegativity decreases down a group because valence electrons occupy higher principal energy levels (larger r) and inner shells provide shielding. The increased distance and shielding reduce the effective nuclear attraction felt by bonding electrons, so atoms lower in a group attract bonding electrons less strongly. Why it matters: this trend helps predict bond polarity (nonpolar → polar → ionic continuum per CED 2.1.A.2–A.3). Review Topic 2.1 study guide for more examples (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I know if a bond is polar or nonpolar covalent?
Start with electronegativity. If two atoms have very similar EN, the shared electrons are equally shared → nonpolar covalent; if ENs differ, the more EN atom pulls electrons toward itself → polar covalent (it gets a partial negative δ–, the other a partial positive δ+). Use these quick steps: 1. Look up EN values and find ΔEN. Small ΔEN → nonpolar (C–H is treated as effectively nonpolar); larger ΔEN → polar (greater ΔEN → larger bond dipole). Remember AP treats bond character as a continuum and that all polar bonds have some ionic character (CED 2.1.A.2–2.1.A.3). 2. Draw dipole arrows (→ with + at tail) and check molecular geometry: bond dipoles can cancel in a symmetric molecule (CO2 is nonpolar overall despite polar C=O bonds). 3. Use the metal vs nonmetal rule: metal–nonmetal bonds are generally ionic; nonmetal–nonmetal are covalent (CED 2.1.A.4). For practice identifying bonds and net molecular polarity, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between a polar covalent bond and an ionic bond?
A polar covalent bond and an ionic bond differ mainly in how the valence electrons are distributed between atoms. - Polar covalent bond: two nonmetals with different electronegativities share electrons unequally. The more electronegative atom gets a partial negative charge (δ–) and the other gets a partial positive (δ+), creating a bond dipole. All polar bonds have some ionic character—bonding is a continuum (CED 2.1.A.3). Polar covalent bonds give molecules directional dipoles and influence solubility and intermolecular forces. - Ionic bond: typically a metal and a nonmetal where electrons are transferred so one becomes a cation and the other an anion; the attraction is strong Coulombic (electrostatic) between full charges (CED 2.1.A.4, 2.1.A.1). Ionic solids usually have high melting points and conduct electricity when molten or dissolved. On the AP exam you should explain bonding using electronegativity trends, partial charges/bond dipoles, and connect bond type to observable properties (melting point, conductivity). For a quick review see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why are carbon-hydrogen bonds considered nonpolar even though carbon is more electronegative than hydrogen?
Carbon is only slightly more electronegative than hydrogen (EN difference ≈ 0.35). That small difference produces a very tiny bond dipole and negligible partial charges, so C–H bonds are treated as nonpolar covalent in AP chemistry (CED EK 2.1.A.2). In many organic molecules the tiny C–H dipoles either cancel by symmetry or are too small to give significant polarity, so hydrocarbons behave like nonpolar substances (low solubility in water, weak permanent dipoles). Remember: bond polarity is a continuum—small EN differences give little ionic character. For more on how electronegativity trends and bond character link to properties (and AP-style practice), check the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and the unit page (https://library.fiveable.me/ap-chemistry/unit-2). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
How does Coulomb's law relate to electronegativity trends?
Coulomb’s law (F = k q1 q2 / r^2) explains electronegativity trends: the stronger the attraction between the positively charged nucleus (more protons = larger q) and an electron (q2), and the smaller the distance r (electrons held closer), the greater the pull on shared electrons—so higher electronegativity. Across a period, nuclear charge increases while valence shell size stays similar, so F increases and electronegativity rises. Down a group, valence electrons are farther from the nucleus (larger r) because of added shells, so F decreases and electronegativity falls. That’s exactly what EK 2.1.A.1 says: use the shell model + Coulomb’s law to rationalize the periodic trend. This matters for bond type/polarity (2.1.A.2–A.3): bigger EN differences → bigger bond dipoles and more ionic character. For more review, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand what delocalized electrons mean in metallic bonding - help?
“Delocalized electrons” just means the valence electrons in a metal don’t belong to one atom or one bond—they’re free to move through the whole metal lattice. In the metallic bond model, positive metal ions sit in a regular array and the valence electrons form an electron “sea” that flows around them. Because electrons are delocalized, metals conduct electricity (electrons move under a voltage) and heat, and the lattice can slide without breaking directional bonds, so metals are malleable and ductile. This idea is exactly what the CED calls out (Essential Knowledge 2.1.A.5). When you explain metallic bonding on the AP exam, link delocalization to properties (conductivity, malleability, luster)—that’s the connection they want. For a quick recap, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and try practice questions (https://library.fiveable.me/practice/ap-chemistry).
Is there a clear cutoff between ionic and covalent bonds or is it more like a spectrum?
It's more of a spectrum, not a sharp cutoff. The CED explicitly says “the difference between ionic and covalent bonding is not distinct but rather a continuum” (2.1.A.3). Electronegativity differences give a helpful rule of thumb (often ~1.7 on the Pauling scale is cited for “mostly ionic”), but that number isn't a hard rule—bonds between a metal and nonmetal are generally ionic and between two nonmetals generally covalent (2.1.A.4). All polar bonds have some ionic character; use trends (EN across a period, down a group—2.1.A.1) plus evidence from a compound’s properties (lattice energy, melting point, solubility, electrical conductivity) to classify bonding. For AP exam work, explain using electronegativity, partial charges/bond dipoles, and the compound’s properties. For a quick review, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and do extra practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do bonds between metals and nonmetals tend to be ionic while bonds between nonmetals are usually covalent?
Metals and nonmetals behave differently because of their valence electrons and electronegativity (CED 2.1.A.1, 2.1.A.5). Metals have low ionization energy and low electronegativity, so they readily lose valence electrons; nonmetals have higher electronegativity and tend to gain electrons. When the difference in electronegativity is large (typical for a metal + nonmetal), electrons are effectively transferred and oppositely charged ions form—that’s ionic bonding, stabilized by strong Coulombic attraction and high lattice energy (CED 2.1.A.3–4). Between two nonmetals the electronegativity difference is small, so they share valence electrons instead—covalent bonds (nonpolar if nearly equal). Remember it’s a continuum: polar covalent bonds have some ionic character (CED 2.1.A.3.c). For AP focus, connect bonding type to element properties and to observable compound properties (melting point, conductivity). Review Topic 2.1 on Fiveable (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I determine bond polarity using electronegativity differences?
Find the two atoms’ electronegativities (from a table), subtract the smaller from the larger, and use that difference plus the bonding partners to judge polarity. Quick rules you can use on the AP: - ΔEN ≈ 0 → nonpolar covalent (electrons shared equally; e.g., C–H treated as effectively nonpolar). - ΔEN ≈ 0.5–1.7 → polar covalent (unequal sharing; the more EN atom gets a partial negative, δ–, the other δ+). - ΔEN ≳ 1.7 and/or metal + nonmetal → mostly ionic (remember AP: bonding is a continuum; check properties to be sure). Also remember: bond polarity is about each bond’s dipole. Use molecular geometry (VSEPR) to see if bond dipoles cancel—if they don’t, the molecule is polar overall. The CED emphasizes trends (EN increases across a period, decreases down a group) and that greater ΔEN → greater bond dipole and more ionic character. For practice and review, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and try problems at Fiveable (https://library.fiveable.me/practice/ap-chemistry).
What properties of a compound can tell me what type of bonding it has?
Look at a compound’s macroscopic properties—they’re the best clues to bond type (CED 2.1.A.4): - Melting/boiling points: very high → ionic or network covalent; low → molecular covalent. - Electrical conductivity: solids that conduct → metallic; molten or aqueous conductors → ionic (ions mobile); molecular solids/liquids usually don’t conduct. - Solubility: ionic/polar covalent dissolve in polar solvents (water); nonpolar covalent dissolve in nonpolar solvents. - Mechanical behavior: shiny, malleable, ductile → metallic (delocalized valence electrons, CED 2.1.A.5); hard and brittle → ionic lattice. - Other clues: volatility (molecular compounds volatile), lattice energy (high for strong ionic bonding), and element types—metal + nonmetal usually ionic; nonmetal + nonmetal usually covalent (but remember it’s a continuum of ionic character, CED 2.1.A.3). Use electronegativity differences plus these properties to decide. For a quick review, see the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and more practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do atoms with higher electronegativity develop partial negative charges in polar bonds?
Electronegativity is an atom’s tendency to attract shared valence electrons. In a polar covalent bond between atoms of different electronegativity, the more electronegative atom pulls the shared electron pair closer to its nucleus. Because electrons are negative, that increased electron density near the more EN atom gives it a partial negative charge (δ–), while the other atom becomes partially positive (δ+). Qualitatively this follows Coulomb’s law and the shell model: a nucleus with greater effective nuclear charge holds electrons more tightly (stronger attraction, smaller average electron–nucleus distance), so the bonding electrons spend more time near the higher-EN atom. Larger EN differences create larger bond dipoles and more ionic character (a continuum, not a sharp divide)—exactly what the CED describes for 2.1.A.3. If you want a short review or practice questions on this, check the Topic 2.1 study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) or AP Chem practice problems (https://library.fiveable.me/practice/ap-chemistry).
How does the shell model help explain electronegativity trends?
The shell model explains electronegativity with distance and shielding of valence electrons plus Coulomb’s law. Across a period, atoms have the same outer shell number but more protons → greater effective nuclear charge (Zeff). Valence electrons are pulled in closer (smaller atomic radius), so Coulomb attraction between nucleus and bonding electrons is stronger → higher electronegativity. Down a group, atoms have more electron shells and increased inner-electron shielding, so valence electrons are farther from the nucleus; Coulomb attraction to bonding electrons is weaker → lower electronegativity. This qualitative explanation matches CED 2.1.A.1 and helps you predict bond polarity (nonpolar vs. polar covalent) and ionic character from electronegativity differences. For a quick refresher tied to Topic 2.1, see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Can a bond have both ionic and covalent character at the same time?
Yes. Bonds lie on a continuum between purely covalent and purely ionic—so a single bond can have both covalent and ionic character. The CED even says all polar bonds have some ionic character and the ionic/covalent distinction isn’t sharp (2.1.A.3). Electronegativity differences drive this: when two atoms have similar EN, electrons are shared (nonpolar covalent); when EN differs moderately, the bond is polar covalent (partial charges, some ionic character); when EN differs a lot and a metal/nonmetal pair forms, the bond is largely ionic. You also see mixed character in real materials (for example, many “ionic” salts show some covalent bonding to explain properties like directionality or lower-than-expected lattice energies). For AP review, focus on electronegativity trends, bond dipoles/partial charges, and using properties (melting point, conductivity, solubility) to judge bonding (see Topic 2.1 study guide on Fiveable for quick examples) (https://library.fiveable.me/ap-chemistry/unit-2/types-chemical-bonds/study-guide/syV9vHl1BKGwM5tnaOt0). For extra practice, check the AP practice problems on Fiveable (https://library.fiveable.me/practice/ap-chemistry).