You may be thinking, what are intramolecular forces? Intramolecular forces are the forces between two atoms in a molecule! This is very different from intermolecular forces, which we learn in unit three.
💡 Intramolecular and intermolecular forces are often confused, so here are some tips:
- Intermolecular forces are those between molecules. Think inter = between two groups
- Intramolecular forces are those between two groups in a molecule, so think intra = within a group.
So far, we learned about two intramolecular forces: covalent bonds and ionic bonds.
👉 For a review of what ionic bonds and covalent bonds are, as well as how to distinguish between the two, check out this study guide about the types of chemical bonds.
Potential Energy and Bonding
You may wonder where potential energy fits into bonding. Well, remember how chemistry favors and always strives to reach the highest stability? The lower the potential energy of a bond, the more stable it is! This is a fundamental rule you should try to keep in the back of your mind when learning about the strength and formation of bonds.
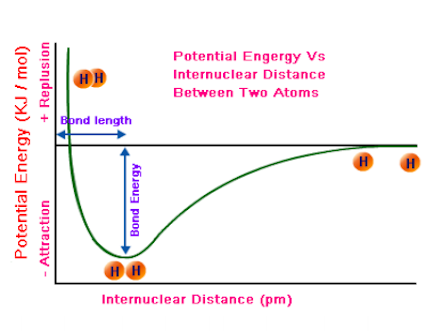
Because of this connection, physical or chemical processes can be described through energy diagrams. A graph of potential energy versus the distance between atoms is a useful tool for understanding the interactions between atoms. Taking a look at this graph, you can see several things:
-
The "equilibrium bond length" - basically another phrase for the distance between atoms where potential energy is at its lowest point. Let's simplify it even more: the distance at which the atoms are the most stable.
-
The bond energy - the amount of energy necessary to separate two atoms in a bond. This can be calculated or conceptually thought of as the difference in potential energy between the separated atoms and the atoms at their equilibrium bond length, or most stable phase.
-
The strength of the bond - this can be grasped from the bond energy. In general, bonds with higher bond energies are stronger and more stable, while bonds with lower bond energies are weaker and less stable.

Potential Energy and Covalent Bonds
In molecular compounds with covalent bonds, the bond length is influenced by both the size of the atoms and the bond order.
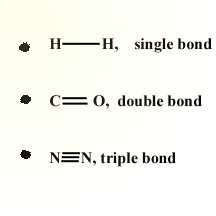
Bond order is another term for how bonds can either be classified as single, double, or triple bonds.
| Bond Order | Electrons Involved in Bond | Bond Length | Bond Energy |
|---|---|---|---|
| Single Bond (-) | Two electrons | Longest | Smallest |
| Double Bond (=) | Four electrons | Middle | Middle |
| Triple Bond (≡) | Six electrons | Shortest | Largest |
An easy way to remember the number of electrons involved in a bond is that each dash on a lewis dot diagram corresponds to two shared electrons.
Image Courtesy of ShodorSince bonds with higher bond energies are stronger and more stable, triple bonds are generally the most stable. This makes them the most difficult to break. However, it is important to note that stability also depends on other factors such as the size and charge of the atoms involved. Let's try and put some of this information together and take a look at a graph of potential energy.
👀 Breaking Down a Potential Energy Diagram
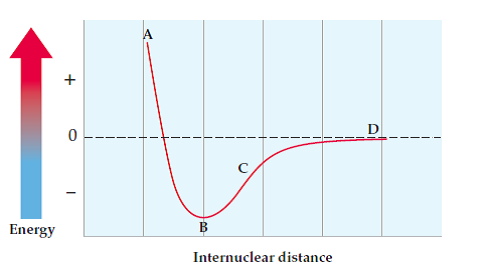
For covalent bonds, the bond length is influenced by the bond order (single, double, triple) and the balance between repulsive and attractive forces. Bond energy in the diagram shows how the greatest potential energy is the repulsion of two atoms.
Image Courtesy of SplainScienceLet's take a look at each of these stages:
- Repulsion: Since the atoms are very close together and the internuclear distance is very small, the atoms are experiencing lots of electron-electron repulsion. This causes the bond to be very unstable and leads to a potential energy of greater than zero.
- Some overlap/attraction: This is the most stable state and the bond length at this point is what we referred to as the "equilibrium bond length." There is a balance between the repulsive and attractive forces and a stable bond is formed. Hopefully, now you understand why potential energy is lowest when the bond is stable.- The potential energy at this stage is the amount required to break the bond or the bond energy.
- No overlap/attraction:Since the internuclear distance is so large, there are no interactions between the two atoms, and no bond is formed. This leads to a potential energy of almost zero.
Example with PE Diagrams
It is good to understand these properties because you may be asked to guess where an element falls on this graph.
Say the following image is a diagram of chlorine atoms bonded together (Cl-Cl). Where would Br-Br fall in comparison to chlorine's curve?
Image Courtesy of CheggTo answer this question, we have to think about periodic trends and take a look at the axes of this graph:
- Internuclear distance: is the Cl-Cl bond or Br-Br bond longer? Well, the bond between the atoms with larger atomic radii would have to be the longer one. As you go down in a group on the periodic table, the atomic radius increases. Since bromine is below chlorine on the periodic table, the Br-Br bond is longer than the Cl-Cl bond.
- This tells you where to draw the Br-Br curve in relation to the x-axis.
- Potential energy: which bond would be easier to break? Cl-Cl or Br-Br? This should automatically make you think of ionization energy. The lower the ionization energy, the easier it would be to break the bond. As you go down a group, ionization energy decreases because there are more occupied electron shells and the nucleus' attraction with the valence electrons is weakest. Therefore, bromine has a lower ionization energy and the Br-Br bond is much easier to break.
- This tells you where to draw the Br-Br curve in relation to the y-axis.
Knowing that the Br-Br bond is longer and easier to break, you would have to graph its curve up (less energy) and to the right more (larger internuclear distance).
This question is a very good way to test your knowledge about this key topic and periodic trends. Here is what the graph should look like:
Forces Within Ionic Bonds
Understanding the strength of ionic interactions involves the use of Coulomb's Law.
As we went over in the previous unit, you do not need to know the formula. However, you are expected to understand what Coulomb's Law conceptually means.
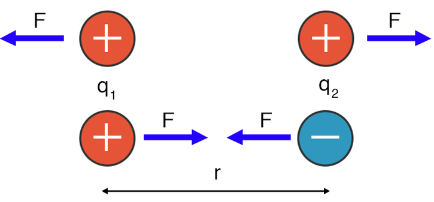
Essentially, the energy of two interacting charged particles (ions) depends on the magnitude of charge and the distance between the nuclei of the two particles.
- The greater the charge of the atoms, the stronger the attraction. This is because the more positively charged a nucleus is, the more strongly it can attract electrons toward it.
- The closer the two particles are to each other, the stronger the attraction. Think about it this way: magnets are not attracted to each other if they are placed 3 feet apart. They need to be close to feel the attraction.
Smaller and highly charged ions have the strongest interactions, according to Coulomb's Law.
Image Courtesy of Science FactsTake a look at this diagram. Don't worry too much about the variables, but F represents the force, q1 and q2 represent the magnitude of charges of the respective atoms, and r represents the distance between the nuclei of the atoms.
This image is basically showing you that Coulomb's law is the foundation behind "opposites attract." Coulomb's Law is everywhere!
Attraction occurs if the charges are opposite (positive and negative), while repulsion occurs if the charges are the same (positive and positive or negative and negative).
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
anion | A negatively charged ion formed when an atom gains one or more electrons. |
bond energy | The average energy required to break a chemical bond between two atoms. |
bond length | The distance between the nuclei of two bonded atoms, which is affected by bond order and atomic radius. |
bond order | The number of electron pairs shared between two atoms in a chemical bond, which affects bond energy and bond length. |
cation | A positively charged ion formed when an atom loses one or more electrons. |
Coulomb's law | The principle that the electrostatic force between charged particles is proportional to the product of their charges and inversely proportional to the square of the distance between them. |
covalent bond | A chemical bond typically formed between two nonmetals where valence electrons are shared between atoms. |
equilibrium bond length | The separation between atoms at which the potential energy is at its lowest point, representing the most stable bond distance. |
internuclear distance | The distance between the nuclei of two atoms, which influences the strength of the interaction between them. |
ionic interaction | The electrostatic attraction between cations and anions, whose strength depends on the charges of the ions and the distance between them. |
potential energy | The stored energy in chemical bonds and molecular structures that can be released or absorbed during a reaction. |
Frequently Asked Questions
What is intramolecular force and how is it different from intermolecular force?
Intramolecular forces are the attractions that hold atoms together inside a molecule or ion—think covalent bonds (shared electrons) and ionic bonds (electrostatic attraction between cations and anions). They show up on a potential-energy vs. internuclear-distance curve: the lowest point is the equilibrium bond length and the depth is the bond energy (how much energy you must add to break the bond). Bond order, atomic size, Pauli repulsion, and Coulomb’s law (for ionic bonds) control intramolecular strength—higher bond order = shorter, stronger bond; smaller ions and larger charges → stronger ionic interactions. Intermolecular forces are weaker attractions between separate molecules (dispersion, dipole–dipole, hydrogen bonding). They determine boiling/melting points and solubility but don’t change molecular connectivity. On the AP exam, you should be able to relate these ideas to potential-energy curves, bond dissociation energy, and trends from Coulomb’s law (CED 2.2.A). For a focused study guide, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC). For more unit review and practice problems, visit the Unit 2 page (https://library.fiveable.me/ap-chemistry/unit-2) and AP Chem practice bank (https://library.fiveable.me/practice/ap-chemistry).
How do you read a potential energy vs distance graph for atoms?
Read the graph like a map of attraction vs. repulsion. The x-axis is internuclear distance (r); the y-axis is potential energy (PE). Key things to pick out: - The minimum point = equilibrium bond length (r_e). At r_e PE is lowest so the bond is most stable (CED 2.2.A.1). - Depth of the well below the zero-asymptote = bond energy / bond dissociation energy (energy you must add to separate atoms). Deeper well = stronger bond. - Left of r_e PE rises steeply (Pauli repulsion from overlapping cores). Right of r_e PE approaches zero (atoms separated, no bond). - Steeper, deeper wells = shorter, stronger bonds (higher bond order: single < double < triple). Morse-type curves show this shape. - For ionic pairs, use Coulomb’s law: higher charges or smaller ionic radii → deeper well (stronger interaction). On the AP exam you may be asked to identify r_e, bond energy, or compare curves. Review this Topic 2.2 study guide for examples (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the equilibrium bond length and why does it matter?
Equilibrium bond length is the internuclear distance where the potential energy curve reaches its minimum—the separation at which attraction (electron–nucleus) and repulsion (Pauli repulsion between cores/electrons) balance, so the bonded atoms are most stable. On a PE vs. distance graph it’s the x-value of the lowest point; the depth of that minimum is the bond energy (bond dissociation energy). It matters because shorter, higher-order bonds (double, triple) have smaller equilibrium lengths and larger bond energies; atomic size and charge also shift the minimum (Coulomb’s law explains stronger attraction for higher charges or smaller ionic radii). On the AP exam you should be able to read and explain these graphs, link minima to bond length/energy, and predict trends with bond order or atomic radius. For a quick review, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about bond energy - is it the energy needed to break a bond or make one?
Bond energy (bond dissociation energy) is the amount of energy required to break a bond—that is, to separate bonded atoms from the equilibrium bond length to infinite separation. On a potential energy vs. internuclear distance graph (CED 2.2.A.1), the bond energy equals the depth of the energy well relative to separated atoms. Breaking a bond requires you to add that positive amount of energy; forming the same bond releases that same magnitude of energy (negative ΔE). Higher bond order (double, triple) gives a deeper well (larger bond energy) and a shorter equilibrium bond length (CED 2.2.A.2). This concept shows up on the AP exam in questions about potential-energy curves, bond strengths, and bond dissociation energies. For a concise review, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC). For extra practice, Fiveable has tons of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why are triple bonds shorter than single bonds?
Short answer: triple bonds are shorter because they have a higher bond order—more shared electron density (one σ + two π bonds) is concentrated between the two nuclei, increasing the electrostatic attraction and pulling the nuclei closer until the potential energy is minimized. On a potential-energy vs. internuclear-distance curve, that stronger attraction shifts the equilibrium bond length to a smaller distance and raises the bond dissociation energy (so triple bonds are both shorter and stronger)—exactly what the CED says in 2.2.A.1–2.2.A.2. Pauli repulsion (between core electrons) still limits how close the nuclei get, so the bond length is the balance point where attraction and repulsion give the lowest potential energy. For more on potential-energy curves, bond order, and AP-style connections, check the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC). For extra practice, try AP-style problems at (https://library.fiveable.me/practice/ap-chemistry).
What does bond order mean and how does it affect bond length?
Bond order is how many shared electron pairs connect two atoms (single = 1, double = 2, triple = 3). Higher bond order means more electron density between the nuclei, so the nuclei are pulled closer together. On a potential-energy vs. internuclear-distance graph, that shows up as a smaller equilibrium bond length and a deeper energy minimum (larger bond energy) for higher bond orders (CED 2.2.A.1–2). So triples are shortest and strongest, doubles are intermediate, singles are longest and weakest. If you want to visualize this or practice graph/term questions for the AP exam (they expect you to interpret potential-energy curves and relate bond order to bond length and bond energy), check the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and more unit review at (https://library.fiveable.me/ap-chemistry/unit-2). For extra practice problems, see (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain Coulomb's law in simple terms for ionic bonds?
Coulomb’s law says the electric force (and therefore the interaction energy) between two charged particles depends on their charges and how far apart they are: F ∝ q1·q2 / r^2 (and potential energy U ∝ q1·q2 / r). For ionic bonds that means two simple things: (1) bigger charges (like 2+ and 2– vs. 1+ and 1–) make much stronger attractions, so higher bond energy; (2) smaller ions let nuclei get closer (smaller r), so the attraction is stronger and the equilibrium bond length is shorter. On a potential-energy vs. internuclear-distance graph, stronger Coulomb attraction pushes the minimum (lowest U) deeper and at a shorter r—that’s your larger bond energy and shorter bond length (CED 2.2.A.1 and 2.2.A.3). For more practice and AP-style explanations, check the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and solve related practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why do smaller ions have stronger interactions than bigger ones?
Smaller ions interact more strongly because their centers (nuclei) are closer together, so the electrostatic attraction is larger. Coulomb’s law shows force ∝ (q1·q2)/r^2 (and potential energy ∝ (q1·q2)/r), so for the same charges smaller r → bigger attraction and a deeper (more negative) potential-energy well. Practically that means shorter equilibrium bond/ion separation, higher bond (or lattice) energy, and a steeper potential-energy curve (stronger, harder-to-separate interaction). Atomic/ionic radius and effective nuclear charge control r; smaller radius (or higher charge) increases interaction strength. This idea is exactly what Topic 2.2 connects to potential-energy vs. internuclear distance and ionic bond strength in the CED. For more AP-aligned review, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and extra practice (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between covalent bond length and ionic bond strength?
Covalent bond length is the equilibrium internuclear distance where the potential energy of two atoms is lowest on a PE vs. distance curve (think Morse potential). It depends on atomic size (core radius) and bond order: larger atoms give longer bonds; double/triple bonds are shorter and have higher bond energies (harder to break). Ionic bond strength isn’t a “length” only—it’s the Coulombic attraction between cation and anion and is described by Coulomb’s law: strength ∝ (charge1 × charge2)/distance. So higher charges and smaller ionic radii → stronger ionic interactions (higher lattice or attraction energy). On the AP, expect to relate PE curves, equilibrium bond length, and bond energy for covalent bonds (CED 2.2.A.1–2) and use Coulomb’s law to explain ionic strength (CED 2.2.A.3). For a quick review, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How does the charge on ions affect how strongly they attract each other?
Charge matters a lot—Coulomb’s law tells you the electrostatic force (and thus attraction) is proportional to the product of the charges and inversely proportional to the square of the distance. So ions with larger magnitudes (e.g., +2 and −2) attract each other much more strongly than +1 and −1 pairs. On a potential-energy curve that means a deeper (more negative) well and typically a shorter equilibrium distance and larger bond (lattice) energy for higher charges. Practically: MgO (Mg2+ & O2−) has a much stronger ionic attraction and higher melting point than NaCl (Na+ & Cl−). Remember size matters too—smaller ionic radii let centers get closer, increasing attraction (CED 2.2.A.3). For more review on potential-energy curves and Coulomb’s law in AP terms, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2). Practice problems: (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why the potential energy graph has that curve shape - what causes it?
Think of two atoms approaching: two forces compete, and their potential energy curve is the result. - At long distances PE ≈ 0 because atoms don’t interact much. - As they get closer, attractive interactions (electrostatic attraction between nuclei and the other atom’s electrons—think Coulomb’s law) lower the PE, so the curve falls. That creates the well. - At very short distances electron clouds start to overlap and Pauli repulsion plus nucleus–nucleus repulsion make PE rise very steeply. The lowest point of the well is the equilibrium bond length (minimum PE) and the depth is the bond (dissociation) energy. The smooth asymmetric shape is captured by the Morse potential: gradual approach to zero on the right (dissociation) and a sharp rise on the left (strong repulsion). This is exactly what the CED expects you to interpret on a PE vs. internuclear distance graph (equilibrium bond length and bond energy). For a quick refresher, check the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC). For more practice problems, see Fiveable’s AP Chem practice page (https://library.fiveable.me/practice/ap-chemistry).
What happens to bond energy when you go from single to double to triple bonds?
As bond order increases (single → double → triple) bond energy increases and bond length decreases. That means a double bond is shorter and stronger than a single bond, and a triple bond is shorter and stronger than a double bond. On a potential-energy vs. internuclear-distance curve the equilibrium bond length shifts left (smaller r) and the well depth (bond dissociation energy) gets deeper as bond order increases—so it takes more energy to separate atoms. This follows CED 2.2.A.1–2.2.A.2: higher bond order → larger bond energies, shorter equilibrium bond lengths. You’ll see this on AP free-response or MCQ items that ask you to compare bond strengths or draw/interpret potential-energy curves. For a quick review of Topic 2.2 and practice, see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and try extra problems at (https://library.fiveable.me/practice/ap-chemistry).
How do I use Coulomb's law to predict which ionic compound will be stronger?
Use Coulomb’s law: F ∝ (q1·q2)/r^2 (and potential energy ∝ q1·q2/r). For ionic compounds that means two things predict stronger ionic attraction (stronger lattice energy, deeper potential-energy well, shorter equilibrium distance): - Larger charges → much stronger attraction. Example: MgO (Mg2+ and O2−, product = +4) will have a much stronger attraction than NaCl (Na+ and Cl−, product = +1) if ion sizes are similar. - Smaller ions (smaller r) → stronger attraction because the centers are closer. So LiF (small Li+, small F−) is stronger than KBr (larger ions) even though charges are the same. On AP questions, state both charge and ionic radius and then use Coulomb’s law to justify which lattice/potential-energy is larger (CED 2.2.A.3). For more examples and practice, check the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and AP Unit 2 review (https://library.fiveable.me/ap-chemistry/unit-2).
Why does atomic size affect both covalent and ionic bonding?
Atomic size changes the distance between nuclei and electrons, so it changes both bond length and interaction strength. - Covalent bonding: bigger atoms have larger electron clouds and cores, so valence orbitals overlap less. That increases the equilibrium internuclear distance (longer bond length on a potential-energy curve) and lowers bond energy—bonds are weaker and easier to break. (Also remember bond order: higher order → shorter, stronger bonds.) - Ionic bonding: Coulomb’s law (F ∝ q1q2/r^2, E ∝ q1q2/r) shows interaction strength increases as ion centers get closer. Smaller ions give smaller r, so stronger attraction and larger lattice energies; bigger ions => larger r, weaker ionic interactions. On the AP, you should be able to sketch potential-energy vs internuclear distance showing these effects and cite equilibrium bond length and bond energy (CED 2.2.A). For a quick review, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC), the Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between potential energy and the actual strength of a chemical bond?
Potential energy vs. bond strength: the deeper and wider the potential-energy well, the stronger the bond. On a PE vs. internuclear-distance graph the minimum is the equilibrium bond length (lowest PE) and the vertical depth from that minimum to the separated-atoms PE is the bond (dissociation) energy—the energy required to break the bond. So larger bond energy = stronger bond. Why bonds differ: - Bond order: single < double < triple → shorter equilibrium distance and larger bond energy (stronger). - Atomic size / Coulomb’s law: smaller atoms (shorter r) and larger charges give stronger attraction for ionic bonds. - Short distances show huge PE rise from Pauli repulsion (prevents nuclei from overlapping). On the AP exam you’ll be asked to read PE vs. distance graphs and relate minimum depth and position to bond energy and length. For a focused review, see the Topic 2.2 study guide (https://library.fiveable.me/ap-chemistry/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC) and practice questions (https://library.fiveable.me/practice/ap-chemistry).