This unit is all about molecular and ionic compound structure and properties! Now that we've discussed the types of bonds, intramolecular forces, and the structure of ionic solids, it is time to discuss metals and alloys!
Metallic Bonding
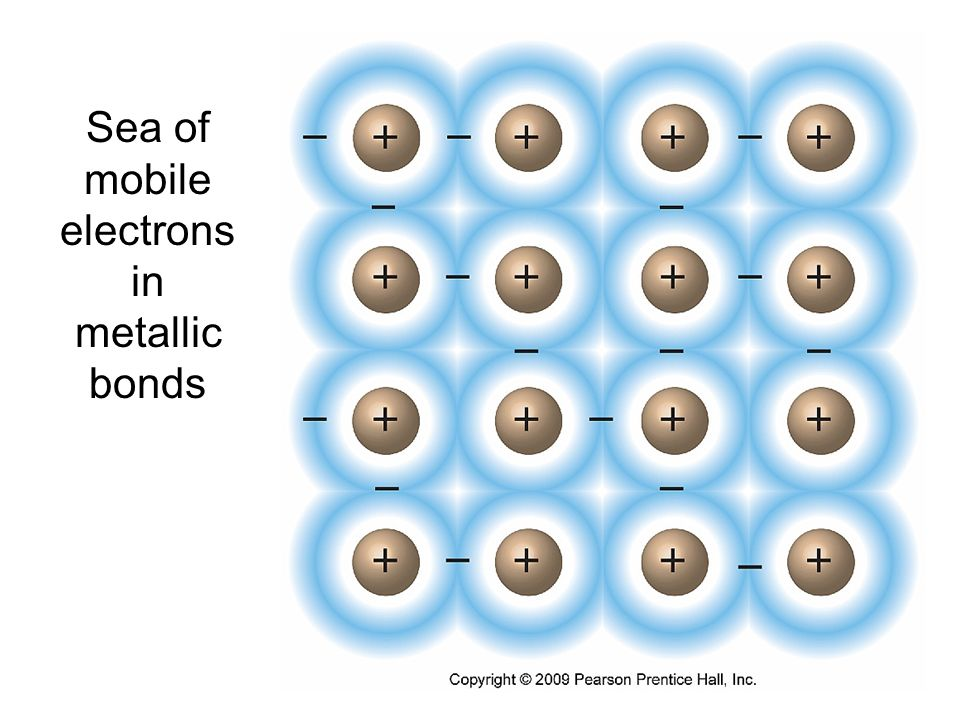
Although ionic and covalent substances are more common, you should be familiar with metallic substances as well and their lattice of cations surrounded by a ‘sea’ of valence electrons.

Sea of Electrons
When metals ionize, they lose a valence electron and become positive ions or cations. Metallic bonding can therefore be represented as an array of cations surrounded by this "sea" of valence electrons.
What this means is the nucleus and core electrons of the metal stay in place, but the valence electrons are very mobile, allowing them to be thought of as a sea.
Electrons usually belong to a certain atom but in metals, they move so much that they don't belong to one, single atom.
Since the valence electrons are free to move throughout the entire metallic structure, metallic substances have very unique properties:
- ⚡ Good conductors of electricity: The valence electrons in metals are delocalized. This simply means that they are mobile and can move freely throughout the entire structure. This allows metals to carry an electric current and conduct charge or, as we'll see in a future unit, be used in redox reactions.
- 🌡️ High melting and boiling points: These metallic bonds are very strong and require a lot of energy to be broken. Imagine boiling a metal such as gold or iron
- 🌟 Shiny appearance: The way light reflects off delocalized electrons in a metal causes many metals to appear shiny.
- 🔌 Malleability and ductility: Malleability reflects the metal's ability to be deformed and shaped without breaking. Ductility reflects a metal's ability to be bent and spun into a wire. These characteristics occur because the structure of metals is much less rigid than that of ionic solids. They can more easily be rearranged.
Comparing Solids
When comparing properties among the different solids, remember this chart:
| Type of Solid | Form of Unit Particles | Forces Between Particles | Properties | Examples |
|---|---|---|---|---|
| Molecular🧊 | Atoms or Molecules | LDFs, dipole-dipole, hydrogen bonds | fairly soft, low melting point, bad conductor | Argon, methane, sucrose, dry ice |
| Covalent-Network💎 | Atoms connected in a network of covalent bonds | Covalent bonds | Very hard, very high melting point, bad conductor | diamond, quartz |
| Ionic🧂 | Positive and negative ions | Electrostatic attractions | Hard and brittle, high melting point, bad conductor | salts (NaCl) |
| Metallic✨ | Atoms | Metallic bonds | varying hardness and melting points, good conductor, malleable, ductile | metals! Cu, Fe, Al |
Right now, you should only be very familiar with the two bolded rows. The others are covered in unit three in more depth when we discuss intermolecular forces!
Alloys
Metals can also bond with other elements and create alloys. Alloys can be formed when two or more elements, where at least one is a metal, are in their liquid form being mixed together. When this mixture cools, the alloy is created. In order to produce a certain alloy, pure metals and elements must be mixed in a specific ratio. Each combination of substances produces an alloy with very unique characteristics.
There are two types of alloys you should be familiar with.
🍳 Interstitial Alloys
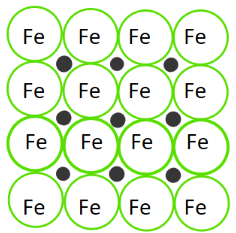
Interstitial alloys form when smaller atoms fill the interstitial spaces between larger atoms. The best example is steel, a substance that you probably haven't realized is an alloy!
Steel is made up of iron and carbon atoms. Iron is a common base element for interstitial alloys, and carbon is a common alloying element. These properties reflect their atomic radii. In steel, carbon fits into the interstices of iron.
Image Courtesy of Study; The little black dots represent carbon atoms.The properties of a sample of steel all depend on the ratio of carbon to iron. The amount of carbon that one uses to make steel can vary, allowing one to make different samples of steel.
Interstitial alloys, like steel, are known for their strength and hardness. This is because of the small size of the interstitial atoms (like carbon) that give the resulting alloy a high density. Think about it this way: you're stuffing small pieces into an array of atoms. This only makes the substance more dense and stronger!
🎺 Substitutional Alloys
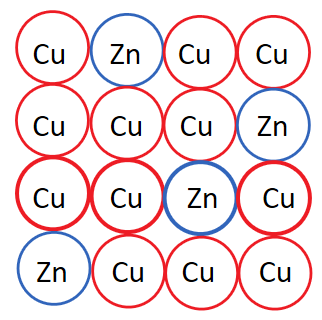
Substitutional alloys form when an atom of one element substitutes an atom of another element of similar size. This is different from interstitial because there are no additional atoms being added to the array, atoms are rather replaced.
The most well-known example of a substitutional alloy is brass. Copper is a common base element for substitutional alloys, and zinc is a common alloying element. In brass, zinc is substituted for copper.
Image Courtesy of StudySubstitutional alloys, like brass, are known for their good electrical and thermal conductivity. These two properties result from the presence of delocalized electrons in the crystal lattice.
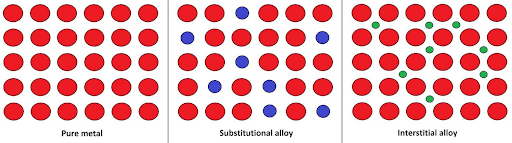
Interstitial vs. Substitutional Alloys
Image Courtesy of Chemistry LibreTexts/06%3A_Structures_and_energetics_of_metallic_and_ionic_solids/6.07%3A_Alloys_and_Intermetallic_Compounds/6.7A%3A_Substitutional_Alloys)/06%3A_Structures_and_energetics_of_metallic_and_ionic_solids/6.07%3A_Alloys_and_Intermetallic_Compounds/6.7A%3A_Substitutional_Alloys)The biggest difference is that with an interstitial alloy, you are adding smaller atoms, but with a substitutional alloy, you are replacing atoms with ones that are similar in size.
Alloys are generally harder and stronger than pure metals because the added elements distort the structure and properties. Alloys are also less malleable than pure metals.
Check your Understanding
The following practice question is based on one posted by the Advanced Placement Youtube channel, and it goes over content reviewed in this guide and the previous guide.
(1) A student ran an experiment to see if the following solids conduct electricity.
| Solids | Does it conduct electricity? |
|---|---|
| Fe (s) | yes |
| FeCl2 (s) | no |
(a) Explain the results the student saw.
(b) Is there anything that could have been different in this experiment to see the FeCl2 sample conduct electricity?
Practice Question Sample Responses
The following are sample responses for part a:
- This student found that a sample of iron conducted electricity since it is a metal. Metals have delocalized valence electrons, usually displayed by the sea of electrons diagram, allowing them to be good conductors of electricity.
- The student found that the sample of FeCl2 didn't conduct electricity because it is an ionic solid. Ionic solids have a lattice structure. Therefore, electrons cannot move freely and the sample didn't conduct electricity.
The following refers to part b:
Recall that as long as there are mobile valence electrons, the sample will conduct electricity. There are two ways to ensure mobile valence electrons are present. Either of the following responses is acceptable:
- Melt the FeCl2 solid and then test it for conductivity. The liquid FeCl2 would conduct electricity since the ions would be mobile and able to flow.
- Dissolve the FeCl2 solid in water. In an aqueous solution, the ions are able to flow and conduct electricity.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
delocalized valence electrons | Valence electrons in a metal that are not bound to specific atoms but move freely throughout the entire metallic structure, often described as a 'sea of electrons'. |
interstitial alloy | An alloy in which smaller atoms occupy the spaces between larger atoms in the crystal lattice, making the structure more rigid and decreasing malleability and ductility. |
interstitial spaces | The gaps or voids between atoms in a crystal lattice where smaller atoms can fit in an interstitial alloy. |
lattice | The regular, repeating three-dimensional arrangement of atoms or ions in a crystalline solid. |
metallic bonding | The type of chemical bonding in metals where valence electrons are delocalized throughout the structure, creating a flexible network of positive ions held together by a mobile electron sea. |
sea of electrons | A model representing the mobile, delocalized valence electrons that surround positive metal ions in a metallic solid. |
substitutional alloy | An alloy formed when atoms of comparable size replace or substitute for atoms in the original crystal lattice structure. |
Frequently Asked Questions
What is metallic bonding and how does it work?
Metallic bonding is the attraction between an array of positive metal ions and a “sea” of delocalized valence electrons. In metals, atoms lose valence electrons that become free to move through the lattice; the remaining cations sit in close-packed arrangements (FCC, BCC, or HCP) while the delocalized electrons hold the structure together. That sea of electrons explains high electrical and thermal conductivity, malleability and ductility (ions can slide past each other while electrons continue to bond them), and metallic luster. Alloying changes properties by either substituting similar-sized atoms into the lattice (substitutional alloys, e.g., brass) or placing much smaller atoms into gaps (interstitial alloys, e.g., carbon in steel). For the AP exam you should be able to represent a metallic solid with positive ions surrounded by delocalized electrons and explain how alloy type affects structure and properties (CED 2.4.A.1–A.3). Review the Topic 2.4 study guide on Fiveable for models and examples (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do metals conduct electricity so well compared to other materials?
Metals conduct electricity well because of metallic bonding: think of a lattice of positive metal ions immersed in a “sea of electrons” made from delocalized valence electrons (CED 2.4.A.1). Those electrons aren’t tied to one atom, so when an electric field is applied they can move freely through the metal lattice, carrying charge with very little resistance. By contrast, in ionic or covalent solids electrons are localized (in bonds or on ions), so they can’t flow easily. Crystal packing (FCC, BCC, HCP) also gives lots of overlap between metal orbitals, which creates continuous energy bands that allow electron flow. Small alloying changes (substitutional or interstitial) can scatter electrons and reduce conductivity, so alloys usually conduct less than pure metals (CED 2.4.A.2–3). For a quick topical review see the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about the sea of electrons model - can someone explain this in simple terms?
Think of a metal as a lattice of positive metal ions (cations) sitting in fixed positions while their valence electrons aren’t tied to any one atom—they move freely through the whole solid like a “sea of electrons.” That’s exactly the CED picture: an array of positive metal ions surrounded by delocalized valence electrons (metallic bonding). Because electrons are mobile, metals conduct electricity and heat; because the ions can slide past each other while the electron sea still holds them together, metals are malleable and ductile. Alloying changes that lattice: substitutional alloys swap similar-size atoms into the lattice, while interstitial alloys pack much smaller atoms into gaps (e.g., C in Fe → steel), which can strengthen the metal. On the AP exam you may be asked to draw or explain this model (LO 2.4.A), so practice describing the positive-ion lattice + delocalized electrons. For a focused review, see the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and hit the practice problems page for more examples (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between interstitial and substitutional alloys?
Interstitial alloys: smaller atoms fit into the spaces (interstices) between the larger metal ions in the metal lattice. Because the added atoms are much smaller, they don’t replace metal atoms but occupy holes—e.g., carbon atoms in iron to make steel. That changes packing, reduces ductility a bit, and strengthens the metal by blocking dislocation motion. (CED: 2.4.A.2) Substitutional alloys: alloying atoms have similar radii to the host metal and replace host atoms in the lattice. The crystal structure stays similar but properties (like conductivity, malleability, corrosion resistance) shift depending on the substituent—e.g., zinc substituting for copper in brass. (CED: 2.4.A.3) For AP exam: be able to draw/describe a metallic lattice (sea of electrons + positive ions) and label which alloy type fits each example. Want practice? Review the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try practice problems at (https://library.fiveable.me/practice/ap-chemistry).
How do I know if an alloy is interstitial or substitutional just by looking at the elements?
Look at the sizes (radii) of the atoms. If the two elements have similar atomic radii (a common rule of thumb: within ~15%), one atom can replace the other in the metal lattice → substitutional alloy (e.g., Cu + Zn → brass). If one element is much smaller so it fits into the spaces between metal atoms (interstices), it forms an interstitial alloy (e.g., C in Fe → steel). Other helpful clues: similar valence/electronegativity and same crystal packing favor substitutional alloys; very small nonmetal atoms (C, H, N) almost always give interstitial alloys. On the AP CED this matches 2.4.A.2–2.4.A.3 (use “smaller atoms fill interstices” vs. “comparable radius → substitution”). Want practice deciding from element pairs? Check the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try problems at https://library.fiveable.me/practice/ap-chemistry.
Why does adding carbon to iron make steel stronger than pure iron?
Pure iron is a metal with a regular lattice of Fe+ ions in a “sea of electrons” so layers of atoms can slide past each other, which makes it relatively soft and ductile. When you add a small amount of carbon, the much-smaller C atoms fit into the interstitial spaces of the iron lattice (an interstitial alloy; CED EK 2.4.A.2). Those carbon interstitials distort the lattice and create local stress fields that block dislocation motion and make it harder for the metal layers to slip. The result: higher strength and hardness (steel), though usually a bit less ductile than pure iron. On the AP exam you might be asked to model this (show Fe ions + delocalized electrons and C in interstices) or explain how alloying changes mechanical properties—focus on lattice distortion and reduced dislocation mobility (LO 2.4.A). For a quick topic review see the Fiveable study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try related practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens to the electron sea when you mix different metals together?
When you mix metals, the “sea of electrons” picture still applies: positive metal ions sit in a lattice and valence electrons remain delocalized and shared across the whole solid. How the sea changes depends on the alloy type. In a substitutional alloy (similar atomic radii, e.g., brass), some host metal atoms are replaced by different metal atoms but the delocalized electrons remain mobile across the lattice—this can change electron density and bonding strength, altering conductivity, ductility, and hardness. In an interstitial alloy (much smaller atoms fit in holes, e.g., C in Fe → steel), extra atoms distort the lattice and impede dislocation motion; the electron sea is still delocalized but lattice distortion increases strength and reduces malleability. For AP Chem, you should be able to draw the model (array of positive ions + delocalized electrons) and name substitutional vs interstitial alloys (CED 2.4.A.1–3). For a quick topic review see the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and find practice problems at (https://library.fiveable.me/practice/ap-chemistry).
Can you give me examples of interstitial alloys besides steel?
Yes—interstitial alloys aren’t limited to iron–carbon steel. “Interstitial” just means small atoms sit in the gaps of a metal lattice. Common interstitial atoms are H, C, N, B, O. Examples you can cite for AP Chem: - Palladium hydride (PdHx): hydrogen atoms occupy interstitial sites in Pd, changing electrical/mechanical properties (good AP example of H in metal). - Hydrogen in nickel or zirconium (hydrides): H in interstices causes embrittlement or hydride formation. - Nitriding/carburizing treatments (not a new alloy formula but a process): nitrogen or carbon diffuses into surfaces of steels and some titanium alloys, occupying interstitial sites and increasing hardness. - Boron-doped steels or tool alloys: tiny B atoms in interstices can improve hardness/toughness at low concentrations. Tie to the CED: these are interstitial solid solutions—small atoms in interstices of the metallic lattice, altering mechanical properties while the metal’s “sea of electrons” remains. For a quick Topic 2.4 review, see the Structure of Metals & Alloys study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p). For more practice on related ideas, check Fiveable’s AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
How do the sizes of atoms determine what type of alloy forms?
If the atoms you mix are about the same size (radii within ~15%), they can replace each other in the metal lattice → a substitutional alloy (one metal atom takes the place of another). If one atom is much smaller, it fits into the gaps (interstices) between the larger metal atoms → an interstitial alloy. The Course and Exam Description uses these exact terms: substitutional (e.g., brass: Zn substitutes for Cu) and interstitial (e.g., steel: C in iron interstices) (CED 2.4.A.2–2.4.A.3). Size-driven alloy type changes properties: interstitial atoms usually harden and reduce ductility; substitutional atoms change lattice spacing and can alter conductivity, malleability, and strength. On the AP exam you may be asked to draw or describe these models (positive metal ions + sea of electrons) and give examples—review Topic 2.4 study guide for diagrams and practice (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p). For more practice Qs, see the unit practice page (https://library.fiveable.me/practice/ap-chemistry).
I don't understand why smaller atoms go into spaces between larger atoms in interstitial alloys - why don't they just mix randomly?
Think of a metal lattice as a rigid array of positive metal ions in a “sea of electrons” (metallic bonding). The large metal atoms sit at fixed lattice points and leave specific empty spots (interstitial sites) between them. Tiny atoms—like carbon in steel—fit into those interstices with little rearrangement of the metal lattice, which lowers the system’s energy. If an atom is similar in size to the metal, it can substitute for a lattice atom (substitutional alloy), but a much smaller atom would cause less strain by occupying an interstitial site than by displacing a larger metal ion. Random mixing isn’t favored because the lattice geometry and size mismatch determine whether substitution or interstitial incorporation is energetically stable. For AP-style answers, describe the lattice + site idea and give carbon-in-iron as an example (CED 2.4.A.2). For more review and practice on metals and alloys, see the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
What are some real world examples of substitutional alloys that I should know for the AP exam?
Good examples to remember: brass (Cu–Zn)—classic substitutional alloy where Zn replaces some Cu in the lattice (given in the CED), bronze (Cu–Sn), sterling silver (Ag with some Cu), cupronickel (Cu–Ni, used in coins), and many aluminum structural alloys (Al with Mg or Cu substituting). Note: stainless “steel” alloys contain substitutional elements (Cr, Ni) substituting for Fe, but plain steel (Fe–C) is an interstitial alloy because carbon sits in Fe interstices—that distinction is one the exam likes (interstitial vs substitutional). Memorize brass as the go-to substitutional example and be able to describe substitutional alloys as atoms of comparable radii substituting in the metal lattice (CED 2.4.A.3). For a quick recap, see the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p). For more practice, try problems on the Unit 2 page (https://library.fiveable.me/ap-chemistry/unit-2) or the practice bank (https://library.fiveable.me/practice/ap-chemistry).
Why do alloys often have different properties than the pure metals they're made from?
Alloys have different properties because adding other atoms changes the metal’s lattice and the “sea of electrons” that holds it together. In a pure metal, identical positive ions sit in a regular lattice with delocalized valence electrons (metallic bonding). When you make an alloy, alloying atoms either substitute for metal atoms (substitutional) or sit in gaps between them (interstitial). Those extra atoms distort the lattice, blocking dislocation motion and making the metal harder and stronger (that’s why small C interstitials in iron make steel less ductile). Alloying can also change electrical and thermal conductivity by scattering the delocalized electrons, and it can alter malleability and corrosion resistance by changing bonding and crystal packing. For AP review, be ready to draw the metallic “sea of electrons” model and label substitutional vs. interstitial alloys (see Topic 2.4 study guide on Fiveable: https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p). For extra practice, check unit problems (https://library.fiveable.me/practice/ap-chemistry).
How do I draw or represent the structure of an alloy on the AP test?
On the AP exam, draw a simple model that highlights the CED points: positive metal ions in a regular lattice plus delocalized valence electrons (a “sea of electrons”), and then show how alloying changes that lattice (learning objective 2.4.A). How to draw it quickly: - Sketch a portion of a close-packed lattice (dots or circles for metal cations in FCC/BCC rows). - Around/behind the lattice draw a shaded/ dotted cloud or a few free electrons (e−) to show the delocalized “sea of electrons” and label both. - For a substitutional alloy, replace some metal ions with differently labeled circles (e.g., Zn in Cu lattice) and note “substitutional: similar radii.” - For an interstitial alloy, draw small atoms (e.g., C) in the gaps between metal ions and label “interstitial: small atoms in voids (steel: C in Fe).” Keep it schematic—clear labels score. The CED expects you to represent these core ideas (2.4.A.1–3). For a quick review, see the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the deal with brass - is zinc just replacing copper atoms in the structure?
Short answer: yes—brass is mainly a substitutional alloy, so Zn atoms replace some Cu atoms in the copper metal lattice rather than just sitting in gaps. Why that works: copper and zinc have similar atomic sizes and both form positive metal ions surrounded by a “sea of electrons” (metallic bonding, CED 2.4.A.1). When Zn substitutes for Cu (an α-brass solid solution up to ≈35% Zn), the delocalized electrons still conduct and allow malleability, but changes in electron count and slight lattice distortion change hardness, strength, and color. At very different sizes or higher Zn content you can get multiple phases or intermetallics instead of a single substitutional solid solution. For the AP exam you should be able to draw the metallic lattice + sea of electrons and label substitutional vs. interstitial alloys (CED 2.4.A.2–3). See the Topic 2.4 study guide for diagrams and practice (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p). For more practice questions, check Fiveable’s AP Chem practice page (https://library.fiveable.me/practice/ap-chemistry).
Do I need to memorize specific examples of alloys or just understand the concepts?
You don’t need to memorize a long list of alloys—focus on the concepts and a few canonical examples the CED gives. For AP Chem you should be able to: - Describe metallic bonding as positive metal ions in a “sea of delocalized valence electrons” and relate that to malleability and ductility (2.4.A.1). - Distinguish interstitial vs. substitutional alloys: interstitial = much smaller atoms in lattice holes (carbon in iron → steel, 2.4.A.2); substitutional = similar-sized atoms replace host atoms (zinc in copper → brass, 2.4.A.3). - Be able to represent a metallic solid or alloy with a model showing ions + delocalized electrons and explain how alloying changes packing/strength. Remember the exam tests models and reasoning more than rote lists (practice representing structures on both multiple-choice and free-response). If you want a quick refresher and the two example alloys the CED uses, check the Topic 2.4 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-metals-alloys/study-guide/yTmM9j22ZoTxtr6TCC3p) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).