Ocean Chemistry and Carbon Cycle
The ocean absorbs roughly 25% of the humans emit each year, making it Earth's largest active carbon reservoir. That absorption helps slow atmospheric warming, but it comes at a cost: as dissolves in seawater, it triggers a series of chemical reactions that lower the ocean's pH. This process is ocean acidification, and it has serious consequences for marine life, ecosystems, and economies that depend on the sea.
Process of Ocean Acidification
Ocean acidification is the gradual decrease in seawater pH caused by the ocean absorbing excess atmospheric . Here's how it works, step by step:
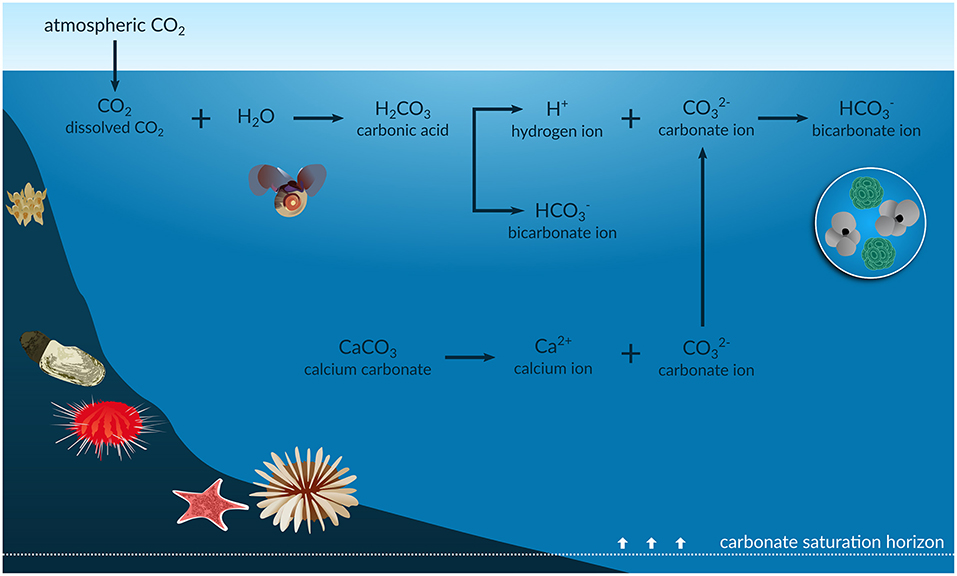
- from the atmosphere dissolves into surface seawater.
- That dissolved reacts with water to form carbonic acid ().
- Carbonic acid quickly dissociates, releasing hydrogen ions () and bicarbonate ions ().
- The increase in concentration is what lowers the pH, making the water more acidic.
Since the Industrial Revolution, ocean surface pH has dropped from about 8.2 to 8.1. That sounds small, but the pH scale is logarithmic, so this represents roughly a 26% increase in hydrogen ion concentration.
The main anthropogenic drivers are:
- Fossil fuel combustion (the dominant source of excess )
- Deforestation, which removes trees that would otherwise absorb
- Land-use changes like agriculture and urbanization, which release stored carbon from soils
Ocean's Role in the Carbon Cycle
Carbon moves between the atmosphere and the ocean through three main pathways:
- Air-sea gas exchange: diffuses across the ocean surface. Cold water absorbs more than warm water because gas solubility increases at lower temperatures.
- Biological pump: Phytoplankton take up during photosynthesis near the surface. When they die or are consumed, carbon sinks to deeper water as falling organic matter (sometimes called marine snow).
- Solubility pump: Cold, dense water at high latitudes absorbs and sinks during thermohaline circulation, carrying dissolved carbon into the deep ocean where it can be stored for centuries.
Once in the ocean, carbon is stored in three main forms:
- Dissolved inorganic carbon (DIC): The largest pool, including dissolved , bicarbonate, and carbonate ions
- Particulate organic carbon (POC): Dead organisms and organic debris sinking through the water column
- Dissolved organic carbon (DOC): Organic molecules dissolved in seawater from biological processes
Several factors affect how much the ocean can absorb. Warmer water holds less dissolved gas, so rising sea surface temperatures actually reduce the ocean's capacity as a carbon sink. Higher salinity also slightly decreases solubility. Biological activity matters too: large algal blooms can temporarily draw down significant amounts of surface .

Chemistry of the Ocean Carbonate System
The carbonate system is the set of chemical equilibria that controls seawater pH. It involves four species:
- Dissolved
- Carbonic acid ()
- Bicarbonate ion ()
- Carbonate ion ()
These are linked by three equilibrium reactions:
In normal seawater (pH ~8.1), bicarbonate () is by far the most abundant form, making up about 90% of dissolved inorganic carbon. Carbonate ions () account for most of the rest, while dissolved and together make up less than 1%.
pH buffering is a critical concept here. The carbonate and bicarbonate ions act as a buffer, meaning they resist changes in pH by absorbing or releasing ions. This is why the ocean doesn't acidify as fast as you might expect. But this buffering capacity has limits, and the current rate of absorption is pushing the system beyond what the buffer can easily handle.
The calcium carbonate saturation state (often written as ) describes whether seawater favors the formation or dissolution of calcium carbonate (). When , conditions favor shell and skeleton formation. When , calcium carbonate dissolves. As ocean acidification reduces the concentration of ions, drops, making it harder for organisms to build their shells. This affects both aragonite (the more soluble form used by corals and pteropods) and calcite (the more stable form used by coccolithophores and some foraminifera).
Temperature, pressure, and biological activity all shift these equilibria. For example, photosynthesis removes and raises local pH, while respiration releases and lowers it.
Impacts on Marine Ecosystems
Ocean acidification doesn't just change water chemistry in the abstract. It has real, measurable effects on marine organisms at multiple levels.
Calcifying organisms are hit hardest. Corals, mollusks, sea urchins, and some plankton all build structures from calcium carbonate. As the saturation state drops, these organisms must spend more energy to form and maintain their shells or skeletons. In severely undersaturated water, existing shells can actually begin to dissolve.
Physiological stress extends beyond shell-building. Many marine animals struggle to maintain their internal acid-base balance when surrounding water becomes more acidic. This can alter metabolic rates, reduce energy available for growth and reproduction, and impair sensory functions. Some fish species, for example, show disrupted behavior in laboratory experiments at elevated levels.
Ecosystem-level changes follow from these individual effects. When key species decline or shift their ranges, food web dynamics change. Species composition and biodiversity can shift as acid-tolerant species replace sensitive ones.
Coral reefs face a double threat. Acidification slows coral growth and calcification rates, while simultaneously making reefs more vulnerable to bleaching from warming waters. The Great Barrier Reef has already shown measurable declines in calcification rates over recent decades.
Fisheries and aquaculture are economically vulnerable. Commercially important shellfish like oysters and clams are highly sensitive to acidification. The U.S. Pacific Northwest oyster industry experienced major larval die-offs in the mid-2000s linked to upwelling of corrosive, low-pH water. Coastal communities that depend on these industries face real economic consequences as conditions worsen.