Valence Shell Electron Pair Repulsion (VSEPR)
Lewis structures can determine properties such as geometry, bond orders, bond lengths, and dipoles for molecules. The Valence-Shell-Electron-Pair-Repulsion (VSEPR) theory can predict molecular geometry by minimizing electron-electron repulsion. It specifically uses the Coulombic repulsion between electrons as a basis for predicting electron arrangement.

What do I need to know about VSEPR?
You should definitely memorize the tables below for the AP Exam. They give you everything you need to know about VSEPR and will answer a lot of questions that require memorization on the AP. Once you practice, the questions that involve the VSEPR Theory become free points! 🥳
When determining molecular geometry, follow these steps:
- Draw the Lewis structure
- Count electron domains (both bonding and lone pairs)
- Determine the electron domain geometry
- Consider only the atoms (not lone pairs) to find the molecular geometry
Let's go over what each column means:
- Family - think of family as how many groups of atoms or molecules branch off the middle atom (number of x + number of e in the general formula).
- General Formula - made up of three parts:- M = middle atom- X = attached atoms- E = lone pairs
- Electron Domain Geography - gives you an idea of what the molecule looks like. It shows where the electrons or atoms are in relation to the middle atom, M.- The graph below also includes angle measures that you should be aware of in that specific molecular geometry.
- Shape - This is the main column that you should memorize and learn to associate with the general formula, electron domain geography, and hybridization.
- Hybridization - For the AP exam, focus on sp, sp², and sp³ hybridizations (families 2, 3, and 4), though sp³d and sp³d² exist for families 5 and 6.
VSEPR
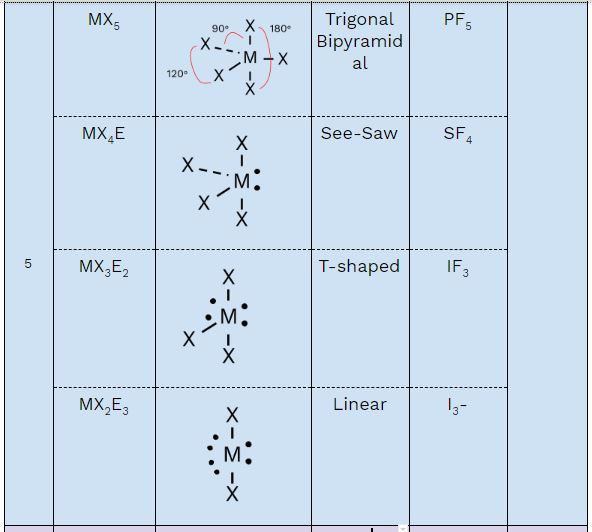
Family 5: Five Electron Domains
When a central atom has 5 electron domains, they arrange in a trigonal bipyramidal electron domain geometry to minimize repulsion. This creates two distinct positions:
- Equatorial positions: 3 positions in a plane, 120° apart
- Axial positions: 2 positions perpendicular to the plane, 180° apart from each other
| General Formula | Molecular Shape | Description | Bond Angles | Example |
|---|---|---|---|---|
| MX₅ | Trigonal bipyramidal | All 5 positions occupied by atoms | 90°, 120°, 180° | PF₅, PCl₅ |
| MX₄E | Seesaw | 1 lone pair in equatorial position | <90°, <120°, ~180° | SF₄ |
| MX₃E₂ | T-shaped | 2 lone pairs in equatorial positions | ~90°, <180° | ClF₃, BrF₃ |
| MX₂E₃ | Linear | 3 lone pairs in equatorial positions | 180° | XeF₂, I₃⁻ |
Lone pairs preferentially occupy equatorial positions because they have more space and minimize repulsion with other electron domains.
Family 6: Six Electron Domains
With 6 electron domains, the electron domain geometry is octahedral, with all positions equivalent at 90° angles:
| General Formula | Molecular Shape | Description | Bond Angles | Example |
|---|---|---|---|---|
| MX₆ | Octahedral | All 6 positions occupied by atoms | 90° | SF₆, PCl₆⁻ |
| MX₅E | Square pyramidal | 1 lone pair creates pyramid with square base | <90° | BrF₅, IF₅ |
| MX₄E₂ | Square planar | 2 lone pairs opposite each other | 90° | XeF₄, ICl₄⁻ |
Bonding
Sigma and Pi Bonds
Bond formation occurs through the overlap of atomic orbitals. The extent and type of overlap determines the bond strength and characteristics.
Sigma (σ) bonds are formed by direct, head-on overlap of atomic orbitals along the internuclear axis (the imaginary line connecting two nuclei). This can occur between:
- Two s orbitals
- An s and a p orbital
- Two p orbitals overlapping end-to-end
- Hybrid orbitals (sp, sp², sp³)
The direct overlap in sigma bonds creates strong electron density between the nuclei, making them the strongest type of covalent bond.
Pi (π) bonds are formed by sideways overlap of p orbitals above and below the internuclear axis. The parallel p orbitals create two regions of electron density - one above and one below the bond axis. Because this sideways overlap is less effective than the head-on overlap in sigma bonds, pi bonds are weaker than sigma bonds. This explains why:
- Single bonds (1σ) can rotate freely
- Double bonds (1σ + 1π) are stronger but rigid
- Triple bonds (1σ + 2π) are the strongest but most rigid
You don't really have to know these definitions, but be aware of the following:
- A single bond is made up of 1 σ bond.
- A double bond is made up of 1 σ bond and 1 π bond.
- A triple bond is made up of 1 σ bond and 2 π bonds.
The more pi bonds in a molecule:
- The higher the bond energy
- The shorter the bond length
- The more restricted the rotation (single bonds can rotate freely, but double and triple bonds cannot rotate due to the pi bonds)
The restriction of rotation around double bonds has an important consequence: it can lead to geometric isomers (also called cis-trans isomers). Geometric isomers are a type of structural isomerism where compounds have the same molecular formula and connectivity but differ in the spatial arrangement of atoms due to restricted rotation.
Unlike single bonds, which can rotate freely, the pi bond in a double bond locks the geometry in place. This means that when different groups are attached to each carbon of a double bond, they can exist as distinct isomers that cannot interconvert at room temperature. For example, in 2-butene (CH3CH=CHCH3):
- Cis-2-butene: Both methyl groups are on the same side of the double bond
- Trans-2-butene: The methyl groups are on opposite sides of the double bond
These are completely different compounds with different physical properties (boiling points, melting points, dipole moments) even though they have the same molecular formula. The existence of geometric isomers is a direct result of the structural rigidity imposed by pi bonds.
Examples
Count the number of σ bonds and the number of π bonds in the following two structures:
Image Courtesy of BC Open textbooksIn the molecule on the left, there is 1 triple bond and 2 single bonds. 1 triple bond is made up of 1 σ bond and 2 π bonds, while the single bond is only made up of 1 σ bond. Therefore, in total, there are 3 σ bonds and 2 π bonds in this molecule.
In the molecule on the right, there are 3 double bonds and 9 single bonds. This means this molecule is made up of 12 σ bonds and 3 π bonds.
Hybridization
Hybridization is the idea that atomic orbitals fuse to form newly hybridized orbitals, which in turn, influences molecular geometry and bonding properties. Hybridization is also an expansion of the valence bond theory.
There are 5 main hybridizations, 3 of which you'll be tested on: sp3, sp2, sp, sp3d, sp3d2. For these hybridizations, electron orbitals fuse together to fill subshells and go to a lower energy state. It also allowed for things like CH4, since technically the way the electron pairs are organized, 4 sigma bonds would not be possible.
In the above example, carbon's 2p and 2s orbitals fuse into 4 half-filled sp3 orbitals that can make 4 sp3-orbital sigma bonds. The same principle applies to the other hybridizations.
Understanding how hybridization connects to molecular geometry is crucial - the type of hybridization directly determines the electron domain geometry and bond angles:
- sp hybridization: Linear geometry with bond angles of 180°
- Example: BeCl₂, CO₂, HC≡CH (around the triple-bonded carbons)
- sp² hybridization: Trigonal planar geometry with bond angles of 120°
- Example: BF₃, H₂C=CH₂ (around the double-bonded carbons)
- sp³ hybridization: Tetrahedral electron domain geometry with ideal bond angles of 109.5°
- Example: CH₄, NH₃ (though lone pairs reduce the H-N-H angle to ~107°)
Note that lone pairs occupy more space than bonding pairs, so they compress bond angles slightly below the ideal values. This relationship between hybridization, geometry, and bond angles helps you quickly analyze molecular structures.
Dipole Moments
A molecule has a dipole moment when there is an uneven distribution of electron density, creating a separation of positive and negative charge. To determine if a molecule has a dipole moment:
-
Check if the molecule contains polar bonds (bonds between atoms with different electronegativities)
-
Consider the molecular geometry - even with polar bonds, symmetrical molecules can have no net dipole
-
Draw dipole arrows pointing from δ+ to δ- for each polar bond
-
Add the vectors - if they cancel out, the molecule is nonpolar; if not, it has a dipole moment
For example, CO2 has two polar C=O bonds, but its linear geometry means the dipoles cancel out, making it nonpolar. In contrast, H2O has two polar O-H bonds in a bent geometry, so the dipoles don't cancel, creating a net dipole moment.
Bond Length and Atomic Radius
Bond length isn't just affected by bond order (single, double, triple bonds) - atomic radius plays a crucial role too. Larger atoms form longer bonds because their valence electrons are farther from the nucleus. When comparing similar bonds:
- C-C bonds are shorter than Si-Si bonds
- C-F bonds are shorter than C-Cl bonds
- H-F bonds are shorter than H-I bonds
This trend follows the periodic trend of atomic radius: as you go down a group, atoms get larger, leading to longer bonds. When analyzing bond lengths, always consider both the bond order (more bonds = shorter distance) and the sizes of the atoms involved.
AP Free-Response Questions
The following questions are from past AP Chemistry exams that were posted online by College Board.
2007 Examination - #6a-d
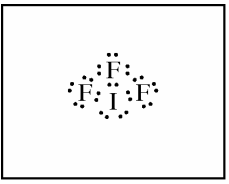
(a) In the box provided, draw a complete Lewis electron-dot diagram for the IF3 molecule.
(b) On the basis of the Lewis electron-dot diagram that you drew in part (a), predict the molecular geometry of the IF3 molecule.
(c) In the SO2 molecule, both of the bonds between sulfur and oxygen have the same length. Explain this observation, supporting your explanation by drawing in the box below a Lewis electron-dot diagram (or diagrams) for the SO2 molecule.
(d) On the basis of your Lewis electron-dot diagram(s) in part (c), identify the hybridization of the sulfur atom in the SO2 molecule.
2010 Examination - #5a-c
Use the information in the table below to respond to the statements and questions that follow. Your answers should be in terms of principles of molecular structure.
(a) Draw the complete Lewis electron-dot diagram for ethyne in the appropriate cell in the table above.
(b) Which of the four molecules contains the shortest carbon-to-carbon bond? Explain.
(c) A Lewis electron-dot diagram of a molecule of ethanoic acid is given below. The carbon atoms in the molecule are labeled x and y, respectively.
Identify the geometry of the arrangement of atoms bonded to each of the following.
(i) Carbon x
(ii) Carbon y
AP FRQ Scoring Guidelines
2007 Examination - #6a-d
For part a, one point is earned for a correct Lewis diagram, such as the one below. This example is done with solely dots, but you can also represent the bonds with lines.
The correct answer for part b is "T-shaped." One point is earned for the correct molecular geometry of the diagram drawn in part a. If you drew the Lewis diagram incorrectly in part a, you may still earn credit in part b as long as your answer is consistent with your drawing.
Two points can be earned in part c. One point is earned with the correct Lewis structure and the other is earned for stating that both sulfur-oxygen bonds are double bonds.
One point is earned in part d for listing the correct hybridization: sp2. If you drew the Lewis structure incorrectly in part c, but your answer in part d was consistent with it, you would get credit in this part.
2010 Examination - #5a-c
One point is earned in part a for the correct Lewis structure drawn in the table.
Two points can be earned in part b: one for the correct choice and one for the correct explanation. The following is a sample response that would earn both points: "Ethyne, which contains a triple bond, has the shortest C-to-C bond. The other molecules have single C-to-C bonds, and triple bonds are shorter than single bonds."
Two points can also be earned in part c, one for naming the correct molecular geometry for carbon x and the other for carbon y:
- Carbon x - trigonal planar
- Carbon y - tetrahedral
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
atomic radius | The size of an atom, typically measured as the distance from the nucleus to the outermost electrons. |
bond angles | The angle formed between two bonds that share a common central atom. |
bond energy | The average energy required to break a chemical bond between two atoms. |
bond length | The distance between the nuclei of two bonded atoms, which is affected by bond order and atomic radius. |
bond order | The number of electron pairs shared between two atoms in a chemical bond, which affects bond energy and bond length. |
bond polarity | The unequal distribution of electron density in a chemical bond due to differences in electronegativity between atoms. |
Coulombic repulsion | The electrostatic repulsion between negatively charged electron pairs that determines their spatial arrangement around a central atom. |
dipole moment | A measure of the separation of positive and negative charge in a polar molecule. |
electron pair | Two electrons occupying the same orbital, including bonding pairs and lone pairs around a central atom. |
geometric isomers | Molecules with the same molecular formula but different spatial arrangements of atoms due to restricted rotation around pi bonds. |
hybrid atomic orbital | An orbital formed by the combination of atomic orbitals on a central atom, used to explain molecular geometry and bonding. |
hybridization | The mixing of atomic orbitals to form new hybrid orbitals that describe the arrangement of electrons around a central atom. |
Lewis diagram | A structural representation of a molecule showing the arrangement of valence electrons as dots and bonds between atoms. |
molecular geometry | The three-dimensional arrangement of atoms around a central atom in a molecule, determined by the positions of bonding and lone pairs. |
multiple bond | Chemical bonds consisting of more than one electron pair shared between two atoms, such as double or triple bonds. |
pi bond | A covalent bond formed by sideways overlap of p orbitals, which prevents rotation and is weaker than a sigma bond. |
polyatomic ion | Charged species composed of two or more atoms bonded together. |
sigma bond | A covalent bond formed by direct overlap of atomic orbitals along the internuclear axis, allowing rotation around the bond. |
sp hybridization | The mixing of one s orbital and one p orbital to form two hybrid orbitals with ideal bond angles of 180°. |
sp2 hybridization | The mixing of one s orbital and two p orbitals to form three hybrid orbitals with ideal bond angles of 120°. |
sp3 hybridization | The mixing of one s orbital and three p orbitals to form four hybrid orbitals with ideal bond angles of 109.5°. |
valence orbital | The outermost electron orbitals of an atom that participate in chemical bonding. |
VSEPR theory | A theory that uses Coulombic repulsion between electron pairs to predict the three-dimensional arrangement of electron pairs and molecular geometry around a central atom. |
Frequently Asked Questions
What is VSEPR theory and how does it work?
VSEPR (valence shell electron pair repulsion) predicts molecular shape by assuming electron pairs (bonding and lone) around a central atom repel each other and arrange to minimize Coulombic repulsion (use a Lewis structure first). Count electron regions to get electron-pair geometry (linear, trigonal planar, tetrahedral, trigonal bipyramidal, octahedral), then remove lone pairs to get molecular geometry (e.g., tetrahedral electron geometry → trigonal pyramidal or bent if lone pairs present). Lone pairs repel more than bonding pairs, so they compress bond angles (ideal angles: sp = 180°, sp2 = 120°, sp3 = 109.5°). Use VSEPR plus Lewis diagrams to predict bond angles, presence of a dipole, relative bond lengths/energies (higher bond order → shorter/stronger), and hybridization (sp, sp2, sp3). Remember multiple bonds have sigma + pi components (sigma stronger), and AP won’t test d-orbital hybridization or detailed hybrid derivations. For a clear walkthrough and practice, see the Fiveable Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do electrons repel each other and how does that affect molecular shape?
Electrons repel because they carry like charges and interact via Coulomb’s law: like charges push away so the system lowers its energy by maximizing distances between electron pairs. VSEPR uses that repulsion to predict how electron pairs (bonding and lone pairs) arrange around a central atom to minimize repulsion. Practically, that means lone pairs occupy more space than bonding pairs (lone pair–lone pair > lone pair–bonding pair > bonding pair–bonding pair), so lone pairs compress bond angles (e.g., NH3 ≈ 107° vs. tetrahedral 109.5°). Combine Lewis structures with VSEPR to get electron-pair geometry and molecular shape (linear, trigonal planar, tetrahedral, bent, trigonal pyramidal, etc.). Also connect shape to hybridization: sp = 180°, sp2 ≈ 120°, sp3 ≈ 109.5° (CED 2.7.A.1–3). For more examples and AP-style practice, check the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and try practice sets at (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between molecular geometry and electron geometry?
Electron geometry describes how all electron pairs (bonding + lone pairs) arrange around a central atom to minimize Coulombic repulsion (VSEPR). Molecular geometry describes the positions of the atoms only—i.e., the shape you’d see if you ignore lone pairs. Example: O in H2O is sp3 (electron geometry = tetrahedral because there are 4 electron regions) but molecular geometry = bent because two of those regions are lone pairs (result: ≈104.5° H–O–H). NH3: electron = tetrahedral, molecular = trigonal pyramidal. CO2: 2 regions → both electron and molecular geometry = linear. For AP exam tasks, use Lewis structures + VSEPR to get electron geometry, then remove lone pairs to report molecular geometry and predict bond angles/hybridization (sp, sp2, sp3) as in the CED (Topic 2.7). For a quick review, see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I figure out if a molecule is sp, sp2, or sp3 hybridized?
Think: how many regions of electron density (bonds—single or multiple count as one region—plus lone pairs) sit on the central atom in your Lewis structure. That number gives the hybridization you use on the AP exam: - 2 regions → sp hybridized → linear, ideal angle 180°. - 3 regions → sp2 hybridized → trigonal planar, ~120°. - 4 regions → sp3 hybridized → tetrahedral, ~109.5°. Remember: a double or triple bond counts as one region (it has one sigma + one or two pi bonds, but still one region for VSEPR/hybridization). Use the full Lewis diagram first, include lone pairs, then count regions. AP won’t ask you to derive hybrids or use d-orbitals (those are excluded in Topic 2.7). Hybridization also connects to sigma vs pi bonding: sigma comes from hybrid overlap, pi from unhybridized p orbitals. Want more examples and practice? Check the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and thousands of practice questions (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about bond angles - why is tetrahedral 109.5 degrees but trigonal planar is 120 degrees?
Think of VSEPR: electron pairs (bonding or lone) repel, so they spread out to maximize distance. With three electron regions around a central atom they lie in one plane 120° apart—that’s trigonal planar (sp2 hybridization, ideal angle 120° per the CED). With four regions they arrange in 3D at the corners of a tetrahedron so every region is as far from the others as possible; the angle between those bonds is about 109.5° (sp3 hybridization, CED gives 109.5°). A quick physical reason: three points on a circle are spaced 120°; four points in space minimize repulsion by forming a tetrahedron, and the math/geometry gives arccos(−1/3) ≈ 109.5°. Remember: lone pairs take up a bit more space than bonding pairs, so they can compress bond angles slightly from these ideal values. For AP review, the CED lists the ideal angles for sp, sp2, sp3 (180°, 120°, 109.5°). For a concise study guide and extra practice problems, see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2).
What's the difference between sigma and pi bonds and which one is stronger?
Sigma (σ) bonds form when orbitals overlap head-on along the internuclear axis (e.g., s–s, s–p, or sp/sp2/sp3 hybrids overlapping). Pi (π) bonds form from side-to-side overlap of unhybridized p orbitals above and below that axis and occur in addition to a σ bond in double/triple bonds. Because head-on overlap gives greater orbital overlap, σ bonds are stronger (higher bond energy) and shorter than π bonds—so the CED says overlap is stronger in σ than π bonds. A single bond = one σ; a double = one σ + one π (π prevents free rotation); a triple = one σ + two π. For AP exam focus: use Lewis/VSEPR + hybridization to explain geometry and relative bond energies (Topic 2.7). For a quick review see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I predict the shape of a molecule using Lewis structures and VSEPR?
Start with a correct Lewis structure: place atoms, count valence electrons, draw bonds, add lone pairs, and check formal charges. Count the regions of electron density (bonding pairs and lone pairs) around the central atom—that gives the electron-pair geometry via VSEPR (2 → linear, 3 → trigonal planar, 4 → tetrahedral, 5 → trigonal bipyramidal, 6 → octahedral). Then get molecular (shape) by removing lone pairs from the electron geometry (e.g., tetrahedral electron geometry with one lone pair → trigonal pyramidal; two lone pairs → bent). Remember lone pairs repel more than bonding pairs, so bond angles shrink (approx. 109.5° for sp3, 120° for sp2, 180° for sp). Assign hybridization from the electron regions (sp, sp2, sp3 for 2, 3, 4 regions). Finally, use bond polarities and molecular geometry to decide if dipole moments cancel. This procedure matches AP CED expectations for predicting geometry, bond angles, hybridization, and dipoles. For a focused walkthrough and practice problems, see the VSEPR & hybridization study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2).
Why can't double bonds rotate but single bonds can?
Single (sigma) bonds form by head-on overlap of orbitals along the internuclear axis, so the electron density is cylindrically symmetrical about that axis—you can rotate the bonded atoms without changing that overlap. Double bonds consist of one sigma plus one pi bond. The pi bond comes from side-by-side overlap of unhybridized p orbitals above and below the bond axis. Rotating around the bond breaks that side-by-side overlap and thus breaks the pi bond, which costs energy. Because the pi bond prevents free rotation, double bonds lock the relative positions of substituents and allow geometric isomers (CED 2.7.A.4). Sigma bonds are stronger (more overlap) than pi bonds, so breaking a pi bond to rotate is unfavorable. For more on sigma vs. pi bonding, VSEPR shapes, and why that gives 120° for sp2 centers (like in C2H4), see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu). Practice related problems at (https://library.fiveable.me/practice/ap-chemistry).
What does hybridization actually mean and why do we need to know about it?
Hybridization is a model that says an atom’s valence atomic orbitals (s and p) mix to form new “hybrid” orbitals that point in directions matching the molecular shape predicted by VSEPR. Practically, it explains why a central C in CH4 uses four equivalent sp3 orbitals (tetrahedral, 109.5°), C in C2H4 uses sp2 (trigonal planar, 120°) and in C≡C uses sp (linear, 180°). We need it because Lewis + VSEPR tell you shape and lone-pair counts, but hybridization connects that shape to which orbitals overlap to form sigma and pi bonds, helps predict bond angles, bond rotation (pi bonds restrict rotation), and where electron density sits—all things the AP CED lists (2.7.A.1–4). Note: AP won’t ask you to derive hybrids or use d-orbital hybrids; just know sp, sp2, sp3 nomenclature and how it ties to geometry. For a quick refresher, check the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do lone pairs affect the shape of molecules compared to bonding pairs?
Lone pairs push harder than bonding pairs because their electron density sits closer to the central atom and isn’t shared between two nuclei. VSEPR uses that stronger Coulombic repulsion to predict shapes and bond angles. So: lone pairs compress bond angles and change molecular geometry relative to the electron-pair (ideal) geometry. Examples you should memorize for the AP: CH4 (sp3)—tetrahedral, 109.5°; NH3 (one lone pair)—trigonal pyramidal, ~107°; H2O (two lone pairs)—bent, ~104.5°. In trigonal bipyramidal systems lone pairs occupy equatorial sites (less repulsion)—giving seesaw, T-shaped, or linear molecular shapes. In octahedral systems a lone pair gives square pyramidal or square planar outcomes. On the AP, always draw a Lewis structure first, count electron domains, then apply VSEPR and hybridization (sp, sp2, sp3) to get angles and polarity (CED 2.7.A). For a focused review, see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between bent and linear molecular geometries?
Linear and bent describe the positions of bonded atoms around a central atom predicted by VSEPR. Linear: two regions of electron density around the central atom (bonding pairs only) repel to 180°, giving an sp (or sometimes sp hybridized) central atom and a straight molecule (example: CO2, 180°). Bent: central atom has at least one lone pair plus bonded pairs; lone pairs repel more strongly than bonding pairs, compressing the bond angle below the ideal (for sp3 central atoms like H2O the angle ≈104.5°; for sp2 with one lone pair it’s ≈120° but slightly less). Key AP connections: use Lewis structures + VSEPR (CED 2.7.A.1–2) to predict shape, bond angles, hybridization (sp, sp2, sp3 per 2.7.A.3), and whether the molecule is polar (bent molecules often have a net dipole; linear can be nonpolar if bond dipoles cancel). For practice and quick review see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and more problems at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how to determine if a molecule has a dipole moment - can someone explain?
Start with a Lewis structure and VSEPR to get the molecular shape (electron pair geometry + molecular geometry). Next, decide each bond’s polarity using electronegativity (ΔEN): if ΔEN ≠ 0 the bond is polar and has a dipole vector pointing toward the more electronegative atom. Finally, add those bond dipole vectors as vectors. If the vector sum ≠ 0 the molecule has a net dipole (polar); if the vectors cancel, it’s nonpolar. Quick rules/examples: - Linear CO2: two equal C=O dipoles opposite → cancel → no net dipole. - Bent H2O: OH dipoles don’t cancel → net dipole (polar). - Trigonal planar BF3: three equal B–F dipoles symmetrically cancel → nonpolar. - Trigonal pyramidal NH3: dipoles don’t cancel → polar. This is exactly the CED skill in Topic 2.7 (predict presence of a dipole using Lewis + VSEPR + bond polarity). For more practice and examples, see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and thousands of practice questions at (https://library.fiveable.me/practice/ap-chemistry).
Why do some molecules with the same number of electron pairs have different shapes?
Because VSEPR predicts arrangements of electron pairs, two molecules can have the same number of electron pair regions but different molecular shapes depending on what those regions are. Key reasons: - Lone pairs vs bonding pairs: lone pairs repel more strongly (LP–LP > LP–BP > BP–BP), so lone pairs compress bond angles and change the visible shape (e.g., tetrahedral electron geometry → trigonal pyramidal or bent molecular geometry). - Multiple bonds: double/triple bonds count as one electron region but have higher electron density and repel more than a single bond, slightly changing angles. - Different placements around non-equivalent positions: in trigonal bipyramidal electron geometry, equatorial vs axial positions give different molecular shapes (seesaw, T-shaped, linear) depending on where lone pairs sit. Use Lewis structures first, then VSEPR and hybridization (sp, sp2, sp3) to predict electron geometry, molecular geometry, and bond angles. AP note: don’t worry about d-orbital hybridization on the exam. For a focused review, see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I remember all the different molecular geometries like trigonal bipyramidal and octahedral?
Think in steric number (electron domains) and AXE notation—that’s the fastest shortcut. Memorize these core cases: - SN = 2: linear (AX2, 180°, sp) - SN = 3: trigonal planar (AX3, 120°, sp2) or bent (AX2E) - SN = 4: tetrahedral (AX4, 109.5°, sp3), trigonal pyramidal (AX3E), bent (AX2E2) - SN = 5: trigonal bipyramidal (AX5) → remember lone pairs go equatorial; seesaw (AX4E), T-shaped (AX3E2), linear (AX2E3) - SN = 6: octahedral (AX6) → square pyramidal (AX5E), square planar (AX4E2) Key tips: count all bonding + lone pairs on the central atom; place lone pairs to minimize repulsion (lone pair > lone-bonding pair); equatorial positions in trigonal bipyramidal have 120° neighbors so they’re preferred for lone pairs. Link geometry to hybridization: sp, sp2, sp3 (AP only expects these; d-orbital hybrids aren’t required). For quick practice and reminders see the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and extra problems at (https://library.fiveable.me/practice/ap-chemistry).
What happens to bond length and bond energy when you have single vs double vs triple bonds?
Short answer: as bond order increases (single → double → triple) bond length decreases and bond energy (strength) increases. That’s because multiple bonds have more electron density between the nuclei: a single bond = one sigma; a double = one sigma + one pi; a triple = one sigma + two pi. Sigma overlap is stronger than pi, but adding pi bonds increases overall attraction, pulls atoms closer (shorter bonds) and raises bond energy (harder to break). Typical trends: triple bonds are shortest and strongest, singles are longest and weakest. Also, pi bonds restrict rotation and contribute to bond order, which AP asks you to use when comparing relative bond lengths and energies (CED 2.7.A.2.c–d and 2.7.A.4). Want examples and practice? See the Topic 2.7 study guide (https://library.fiveable.me/ap-chemistry/unit-2/vsepr-bond-hybridization/study-guide/OslsAmh8LcVoqbpnjPAu) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).