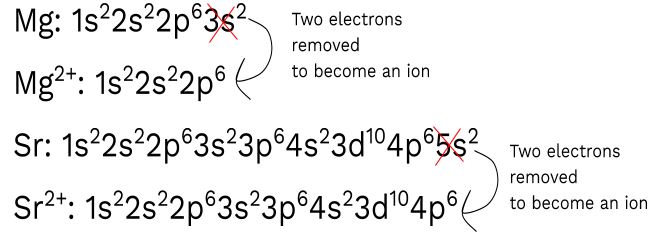Atoms come together to make molecules, right? What terms would you use for the two specific atoms that make up an ionic solid? The words you use can make or break your FRQ response, so make sure you are careful!
Ionic solids are made up of a positive cation and a negative anion. Ionic bonding typically occurs when a metal's valence electron is transferred to a nonmetal. Since the metal loses a negative electron, it becomes a cation. On the other hand, since the nonmetal gains a negative electron, it becomes an anion.
The positive cation and negative anion interact because of their opposite charges, but how much they interact depends, again, on Coulomb's Law.
Structure of Ionic Solids
As mentioned earlier, ionic interactions can produce brittle, hard solids that have high melting points. This is due to the ions being held in a 3-D array known as a crystal lattice.
The reason for this is that ions are attracted to their opposites, and so negative ions surround positive ones and vice versa. You can think of this arrangement as a way to maximize the attractive forces between the cations and anions while minimizing the repulsive forces.
Another characteristic of this arrangement is how the sizes of the ions generally fit well together. When metals ionize, they lose an electron, making them decrease in size. When nonmetals ionize, they gain an electron, making them increase in size. You could see this periodic trend above with a sodium and chlorine atom ionizing. This allows the small cations (Na+ ions) to fit between the larger anions (Cl- ions). This does not happen for all ionic solids, but it is something to note as it impacts the forces experienced.

Representation of Ionic Solids
It is important to note that particle diagrams for ionic substances look different from that of molecular substances (that have covalent bonds). Covalent substances are usually represented by a molecule (such as H2O) while ionic substances are represented by a network of positive 😊 and negative 😞 ions.
Image Courtesy of Principles of General Chemistry; We'll go into this further, but it is good to understand the difference between the two.Explaining the Lattice Structure
The lattice structure can be explained by the strong electrostatic forces that arise between cations and anions because of their opposing charges. Coulomb's law describes these exact forces, telling us that the electrostatic force between a cation and an anion is directly proportional to their charges and inversely proportional to the distance between them.
Image Courtesy of Wikipedia👉 There is no need to memorize the formula, but if you would like to break it down, check out our study guide on atomic structure and electron configuration.
It is rather important to understand that the strength of these electrostatic forces depends on two factors:
- Magnitude of charge: The greater the charge of the cation and anion, the stronger the attraction between the two.
- Distance between the nuclei of the ions: The closer the cation and anion, the stronger the attraction between the two. This is why size is great to note! If the small sodium cations fit between the chloride anions, there is a smaller distance between their nuclei. According to Coulomb's law, this increases the strength of attraction between the sodium and chloride ions!
Remember, attractive forces between cations and anions are maximized in the lattice structure while repulsive forces are minimized.
Properties of Ionic Substances
Ionic substances are typically solids at room temperature and are known for their very high melting and boiling points. Let's break down the general properties of ionic substances:
- 🌡️ High melting and boiling points: The strong electrostatic forces that make up an ionic solid require a lot of energy to overcome, which is why it takes a lot of heat to melt or boil an ionic substance.
- 🚫🔌Poor conductors of electricity as solids: In a lattice, electrons are stuck in place, or in other words, they are localized. Since there are no electrons moving around and carrying an electric current, ionic solids are generally poor conductors of heat and electricity.
- ⚡Good conductors of electricity in their liquid or aqueous states: When ionic solids melt into liquids, their ions are able to move around the liquid phase. In other words, the delocalization of electrons allows them to conduct heat and electricity. This applies to the aqueous phase as well.
- 💎 Hard and brittle: Ionic solids typically have these characteristics because of their strong electrostatic forces. These forces make the ionic solids very difficult to deform.
Lattice Energy
Lattice Energy is the energy released when ions bond to form an ionic solid. We're back to Coulomb's law! It actually relates to lattice energy too; it's kind of everywhere.
💪 Review of Coulomb's Law
Let's ease into lattice energy...do you also remember how to find out which ionic compound would have a higher melting point? Let's review that, it'll help with lattice energy I promise!
- Out of NaF and NaCl, which one has the higher melting point? First, we would look at the charges of the ions. Here, they are both +1/-1, so charges can't have an effect on the differences in melting point.
- Then, we would look at which ions are smaller. Remembering the periodic trends, F- is much smaller than Cl-. Since it is smaller, it can be closer to the Na+ ion and increase the strength of attraction.
- Therefore, it takes more energy to break the bond, increasing the melting point.
If you still don't remember that process, you can review here!
Putting Everything Together
Lattice energy depends on the same two concepts that you used in that question: charge and distance. Coulomb's Law directly relates to melting point and lattice energy so just remember:
The smaller the size and the higher the charge, the higher the lattice energy. Therefore, the higher the melting point of an ionic solid, the higher the lattice energy.
Easy rule, right? Let's try a few out:
Which of the following compounds has a higher lattice energy?
- NaF or NaCl- Since we already thought this question through, it's easy to tell that NaF has the higher lattice energy.
- MgO or NaF- Charge first: Mg and O have a +2/-2 charge, while Na and F have a +1/-1 charge. You don't even have to check the distance, MgO must have the higher lattice energy.
- NaF or KCl- Since these ions are in the same groups, they have the same charge. We must remember the periodic trends and that the farther down in a group you go, the larger the ions get. Therefore, K+ and Cl- are larger than Na+ and F-. With the smaller size, NaF has a higher lattice energy.
- LiCl or NaCl- Last one! Since Li+ is smaller, LiCl must have a higher lattice energy.
Check your Understanding
The following question is from the Advanced Placement YT Channel. All credit to them.
Answer the following questions related to Mg and Sr.
- Write the complete ground state configuration for the ions Mg+2 and Sr+2.
- Do you predict that the ionic radius of Sr+2 is larger or smaller in size than the ionic radius of Mg+2? Justify your answer in terms of atomic structure and the electron configuration of each ion.
- The lattice energy of MgCl2(s) is equal to 2300 kJ/mol. Do you predict that the lattice energy of SrCl2(s) should be less than or greater than 2300 kJ/mol? Justify your answer in terms of Coulomb's law.
Sample Responses
(1) Looking at the periodic table and remembering electron configuration, you should get:
You cannot leave the electron configurations of Mg and Sr as your final answer! Make sure you always answer what they are asking.
I originally wrote down the electron configurations of Mg and Sr and then took off two valence electrons to get the final electron configurations of cations Mg+2 and Sr+2.
(2) This question goes back to periodic trends. Which ion has more electrons and electron shells? Sr2+ does, so it has a larger ionic radius.
Sample Response: Sr+2 has a larger ionic radius than Mg+2 because it has more occupied electron shells. The valence electrons in Sr2+ are in the 4th energy level whereas the valence electrons in Mg2+ are in the 2nd energy level. Electrons in the 4th energy level are generally farther away from the nucleus, making the ion larger.
(3) Charges are the same, so size must be accountable for the difference in lattice energy.
Sample Response: Coulomb's law states that the higher the charges of the ions and the smaller the distance between the ions, the stronger the attraction and the higher the lattice energy. Although the charges of Mg+2 and Sr+2 are the same, Sr is a much larger ion due to its greater amount of occupied energy shells. Since it is larger, the distance between Sr+2 and the chlorine ions is greater than the distance between Mg+2 and the chlorine ions. Therefore, the lattice energy of SrCl2 (s) must be less than 2300 kJ/mol.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
anion | A negatively charged ion formed when an atom gains one or more electrons. |
attractive forces | Electrostatic forces between oppositely charged ions that hold them together in an ionic solid. |
cation | A positively charged ion formed when an atom loses one or more electrons. |
Coulomb's law | The principle that the electrostatic force between charged particles is proportional to the product of their charges and inversely proportional to the square of the distance between them. |
ionic crystal | A solid structure in which cations and anions are arranged in a repeating three-dimensional pattern. |
ionic solid | A crystalline compound composed of cations and anions held together by electrostatic forces. |
particulate model | A representation of matter showing individual atoms, molecules, or ions and their interactions to describe chemical processes at the molecular level. |
repulsive forces | Electrostatic forces between ions of the same charge that push them apart in an ionic solid. |
Frequently Asked Questions
What is an ionic solid and how is it different from other types of solids?
An ionic solid is a 3-D periodic array of cations and anions held together mainly by electrostatic (Coulombic) attraction—think of a repeating ionic lattice/unit cell where each ion sits in lattice sites with a characteristic coordination number and ionic radius. That strong, long-range attraction gives ionic solids high lattice energies, high melting points, and brittleness (cleavage along planes) and makes them nonconductive as solids but conductive when molten or dissolved. How it’s different: metallic solids have delocalized electrons (metallic bonding) and are malleable and conductive as solids; covalent-network solids (like diamond) are a continuous network of covalent bonds with very high melting points; molecular solids (like I2) are held together by intermolecular forces (much weaker, low melting/boiling). You don’t need to memorize specific crystal structures for the AP exam (CED exclusion), but understand ionic packing, nearest-neighbor electrostatics, lattice energy, and how Coulomb’s law controls stability. For a concise topic review see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do ionic compounds form crystals instead of just random arrangements?
Ionic compounds form crystals because a regular 3-D lattice maximizes electrostatic attraction and minimizes repulsion. Coulomb’s law tells you opposite charges attract strongly and like charges repel; arranging cations and anions in a periodic array places each ion next to as many oppositely charged neighbors as possible (high coordination number, e.g., 6 or 8) and keeps like charges farther apart. That arrangement lowers the system’s potential energy and gives a large negative lattice energy (stabilizing the solid). Random arrangements don’t achieve the same net attractive interactions, so they’re higher in energy and unstable. In AP terms: ionic lattices (unit cells) balance ionic radius, effective charge, and nearest-neighbor interactions to maximize attraction and minimize repulsion (see Topic 2.3 in the CED). For a quick review, check the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about Coulomb's law - how does it relate to ionic solids?
Coulomb’s law (F = k·q1·q2/r^2) explains why ionic solids form regular 3-D lattices: opposite charges attract more strongly when charges (q) are larger and when ions are closer (smaller r), so the crystal arranges cations and anions to maximize attractive q+·q− interactions and minimize same-charge repulsions. That’s why coordination number and ionic radius matter—bigger ions pack differently and change r, which alters lattice energy (the stronger the net electrostatic attraction, the more negative the lattice energy). On the AP CED this is exactly Topic 2.3.A: you should be able to represent a particulate model showing ions in a periodic array consistent with Coulomb’s law and explain properties (hard, high melting point) from electrostatic attraction. For more examples and practice, review the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between cations and anions in crystal structures?
Cations are positively charged ions; anions are negatively charged. In an ionic crystal they occupy different lattice sites so the 3-D array maximizes electrostatic attraction (cation–anion) and minimizes repulsion (cation–cation, anion–anion)—that’s Coulomb’s law in action. Cations are usually smaller (lost electrons → smaller ionic radius) and fit into holes formed by the larger anions; the coordination number tells you how many opposite ions surround each ion. Those size and charge differences affect packing, lattice energy, and properties like high melting point and solubility. You don’t need to memorize specific unit cells for the AP exam—just be able to use ideas like ionic radius, coordination number, and Coulomb’s law to explain macroscopic properties (CED 2.3.A.1). For a focused review, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and try practice sets at (https://library.fiveable.me/practice/ap-chemistry).
How do ionic solids arrange themselves to be most stable?
Ionic solids arrange into regular 3-D lattices that maximize cation–anion attraction and minimize like-charge repulsion—basically following Coulomb’s law. That means each ion sits in a repeating unit cell with a coordination number that balances size (ionic radii) and charge so oppositely charged neighbors are as close as possible while same-charge ions stay farther apart. The result: a periodic ionic lattice with high lattice energy (strong overall electrostatic stabilization), determined by nearest-neighbor interactions and factors like charge density and the Madelung constant. Bigger charge or smaller radii → stronger attraction and tighter packing; high polarizability can distort ideal packing. You don’t need to memorize specific structures for the AP exam—just be able to explain that ionic crystals form systematic arrays to maximize attraction / minimize repulsion (CED 2.3.A). For a focused study, see the Topic 2.3 guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and more practice at the Unit 2 page (https://library.fiveable.me/ap-chemistry/unit-2) or the practice problems site (https://library.fiveable.me/practice/ap-chemistry).
Why do opposite charges attract but like charges repel in ionic crystals?
Opposite charges attract and like charges repel because of electrostatic forces described by Coulomb’s law: the force between two charges is proportional to the product of their charges and inversely proportional to the square of the distance between them. In an ionic crystal that means + and − ions pull each other together (large negative potential energy) while same-sign ions push each other apart. Ionic lattices arrange cations and anions in a 3-D periodic array to maximize attractive nearest-neighbor interactions and minimize repulsive same-charge contacts—that arrangement gives the largest (most negative) lattice energy and makes the solid stable. Factors like ionic charge, ionic radius (charge density), and packing affect how strong those attractions/repulsions are (Madelung constant and nearest-neighbor interactions appear in lattice-energy calculations). For AP Chem, you should be able to represent an ionic solid particle model consistent with Coulomb’s law (see Topic 2.3 study guide on Fiveable: https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u). For extra practice, try problems at https://library.fiveable.me/practice/ap-chemistry.
Can someone explain what a 3D array means when talking about ionic solids?
A "3-D array" just means the ions in an ionic solid are arranged in a repeating pattern in all three spatial directions (x, y and z)—think a 3-D checkerboard of cations and anions. Each ion sits at a lattice site in an ionic lattice (the repeating pattern), and the smallest repeating piece is the unit cell. That arrangement maximizes cation–anion attractions and minimizes like-charge repulsions, consistent with Coulomb’s law. Coordination number tells you how many opposite-charged neighbors each ion has in that 3-D network; ionic radius and charge affect how tightly they pack and the lattice energy. You don’t need to memorize specific crystal types for the AP exam—just be able to use a particulate model and explain how 3-D periodicity explains properties like high melting point and brittleness (Topic 2.3 in the CED). For a clear visual and review, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and extra practice (https://library.fiveable.me/practice/ap-chemistry).
What forces are involved in holding ionic solids together?
Ionic solids are held together mainly by strong electrostatic forces between oppositely charged ions—Coulombic attraction between cations and anions—which organize ions into a periodic 3-D lattice that maximizes attraction and minimizes repulsion (CED keywords: Coulomb’s law, ionic lattice, coordination number, nearest-neighbor interactions). Short-range ionic repulsion (from overlapping electron clouds) prevents collapse, so the balance of attraction and repulsion sets the equilibrium ion spacing and coordination. Lattice energy (and the Madelung constant) quantifies the total electrostatic stabilization of the crystal: larger charges and shorter distances → higher lattice energy → stronger ionic solid. Other modifiers: charge density, polarizability, and ionic radius affect packing and strength. For a focused review tied to the AP framework, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how ionic solids maximize attractions and minimize repulsions at the same time?
Think of an ionic crystal as a 3-D checkerboard that places oppositely charged ions next to each other so Coulomb attraction (–k q1q2/r) is as large as possible while keeping like charges separated to reduce repulsion. Practically that means: cations and anions sit in a regular lattice with each ion surrounded by as many opposite neighbors as fits (coordination number) so nearest-neighbor attractions dominate the energy. The packing also depends on ionic radii and charge—bigger or higher-charged ions pull neighbors closer (larger lattice energy) but if same-charged ions get too close strong short-range repulsion increases, so the stable lattice is the arrangement that balances those opposing forces (this is captured by lattice energy and the Madelung constant, which sum all neighbor interactions). You won’t need to memorize specific unit cells on the AP, but you should be able to explain this balance with Coulomb’s law (CED 2.3.A.1). For a focused read, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do you draw a particulate model for an ionic solid?
Draw a particulate model that shows a regular 3-D array of alternating cations and anions, labels for size/charge, and a repeating unit cell. Steps: 1) Sketch a square (or cubic) box to represent one unit cell. 2) Place smaller, positively charged spheres (cations) and larger, negatively charged spheres (anions) at lattice points so neighbors are opposite charge (this maximizes attraction, minimizes like-charge repulsion per Coulomb’s law). 3) Use different colors/sizes and label +/− and approximate ionic radii. 4) Indicate a coordination number (e.g., “coordination = 6” for a 6:6 nearest-neighbor arrangement) and draw arrows showing nearest-neighbor attractions. 5) Add “repeat in x, y, z” arrows to show the infinite lattice. Keep the model qualitative—not a specific crystal type (specific structures aren’t required on the AP). For a quick walkthrough, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u). For extra practice problems, try Fiveable’s AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
Why don't we need to memorize specific crystal structures for the AP exam?
You don't need to memorize specific crystal lattices because the AP expectation is about understanding the principles that make ionic solids behave the way they do—not naming every structure. The CED explicitly excludes “knowledge of specific crystal structures” from assessment. Focus on particulate models that reflect Coulomb’s law, how cation/anion size and charge set coordination number and packing, and how those factors control lattice energy, nearest-neighbor interactions, and properties like melting point and solubility (keywords: ionic lattice, unit cell, coordination number, ionic radius, lattice energy, Madelung constant). On the exam you’ll be asked to apply those ideas, interpret models, or predict trends—not recall lattice names. For targeted review, use the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u), the Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What properties of ions affect how they arrange in a crystal?
Ions arrange in a crystal to maximize attraction and minimize repulsion (Coulomb’s law). The main ion properties that control that arrangement are: - Charge (magnitude): higher charges → stronger attraction, larger lattice energy, often higher coordination numbers to balance neighbors. - Ionic radius (size): smaller ions pack closer, changing coordination number and nearest-neighbor distances. - Charge density / effective ionic charge: small, highly charged ions have high charge density → stronger attraction and shorter ion–ion distances. - Polarizability (how easily an ion’s electron cloud distorts): large, highly polarizable ions increase covalent character and can shift preferred packing. - Relative sizes of cation vs anion: size ratio influences coordination number and which lattice sites fit (packing). - Local electrostatic environment (nearest-neighbor interactions, Madelung constant): affects lattice energy and stability. Remember: you don’t need to memorize specific crystal structures for the AP exam—just be able to explain arrangements using Coulomb’s law, coordination number, ionic radius, lattice energy, and related keywords. For a focused review, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and more unit practice at (https://library.fiveable.me/ap-chemistry/unit-2). For extra problems, check (https://library.fiveable.me/practice/ap-chemistry).
How does the size of ions affect the structure of ionic solids?
Ion size strongly controls how ions pack and how strongly they attract one another. Larger cations or anions increase interionic distance, so Coulomb’s law predicts weaker electrostatic attraction and lower lattice energy; smaller ions sit closer, giving stronger attraction and higher lattice energy. Size also sets coordination number: small cations fit into fewer anion sites (lower coordination), while larger cations can be surrounded by more anions (higher coordination). Big, highly polarizable anions (like I−) distort electron clouds more, increasing covalent character and changing packing. Overall an ionic lattice arranges ions to maximize cation–anion attraction and minimize anion–anion and cation–cation repulsion; that’s why ionic radius, charge density, and nearest-neighbor distances determine unit-cell geometry and stability. (Specific crystal types aren’t required for the AP exam.) For a clear AP-aligned summary and practice, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) or the Unit 2 overview (https://library.fiveable.me/ap-chemistry/unit-2).
Why are ionic solids so different from molecular solids in their arrangement?
Ionic solids pack very differently from molecular solids because their arrangement is driven by Coulomb’s law: opposite charges attract and like charges repel. In an ionic lattice cations and anions form a periodic 3-D array that maximizes cation–anion attractions and minimizes cation–cation and anion–anion repulsions—so ions sit at lattice sites with high coordination numbers determined by ionic radii and charge (this gives the regular unit cell structures and large lattice energies). By contrast, molecular solids are held together by weaker intermolecular forces (dipole, dispersion, H-bonding), so their packing reflects molecule shape and directional interactions, not strong long-range electrostatics. For AP exam framing, represent ionic solids with particulate models consistent with Coulomb’s law and mention concepts like coordination number, ionic radius, and lattice energy (CED 2.3.A.1). For a quick review see the Topic 2.3 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens to the forces between ions when you change the distance between them?
When you change the distance between ions, the electrostatic forces change a lot because of Coulomb’s law. The magnitude of the attractive (or repulsive) force goes as F ∝ q1·q2 / r^2, so if you double the distance between ions the force falls to one-quarter; if you halve the distance the force quadruples. The electrostatic potential energy (what matters for lattice stability) varies as E ∝ q1·q2 / r, so smaller r gives a much more negative (more stable) energy for oppositely charged ions. At very short distances, strong repulsion (from electron cloud overlap/Pauli exclusion) rises sharply and prevents collapse of the lattice. These distance-dependent attractions and repulsions explain why ionic crystals adopt arrangements that maximize nearest-neighbor opposite-charge contacts and minimize same-charge repulsions (CED Topic 2.3, Coulomb’s law, lattice energy, nearest-neighbor interactions). For more review, see the Topic 2.3 study guide (https://library.fiveable.me/ap-chemistry/unit-2/structure-ionic-solids/study-guide/3khaTI6A3tdnaMTMJy2u) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).