Molecular Collisions and Heat Transfer
Temperature reflects the average kinetic energy, or random motion, of particles. Kinetic energy is related to particle speed, so as temperature increases, molecules move faster and collide with the walls of the container more frequently and with greater force.

Key Principle: Temperature and Kinetic Energy
The particles in a warmer body have a greater average kinetic energy than those in a cooler body. This difference in average kinetic energy is what drives heat transfer between objects at different temperatures.
The Collision Theory
Collision theory is based on the following postulates:
- The rate of a reaction is proportional to the rate of the collisions 💨
- Orientation matters for the reactant molecules colliding ➡️⬇️
To understand how collision theory works, it's useful to think of an analogy to break down the molecular processes. Let's say you're making lemonade. To make lemonade, you need to mix lemon juice, sugar, and water together. But just having the ingredients in the same place isn't enough to make lemonade - they need to interact with each other in just the right way.
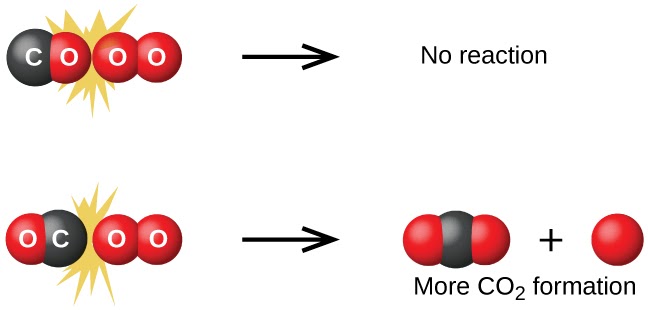
Collision theory is like the "rules" for how ingredients can mix together to make a reaction happen. It says that for two things to react and form a new substance, they need to bump into each other with enough energy, in the right orientation, and often enough. Think of it like two billiard balls - if they don't hit each other hard enough, or at just the right angle, they won't react.
And just like how making lemonade faster would require you to mix the ingredients more quickly, the theory also explains how increasing the temperature or concentration of reactants can make the reaction happen faster by increasing the number and energy of collisions.
Successful collisions would therefore collide with proper orientation to break bonds and have enough energy to overcome the reaction’s activation energy.
🧠 Just remember: proper orientation + enough energy = successful collision.
Heat Transfer Through Molecular Collisions
When objects at different temperatures come into thermal contact, collisions between particles result in the transfer of energy. This process is called:
- "Heat transfer"
- "Heat exchange"
- "Transfer of energy as heat"
Here's how it works:
- Faster-moving particles from the warmer body collide with slower-moving particles from the cooler body
- During these collisions, kinetic energy is transferred from the faster particles to the slower particles
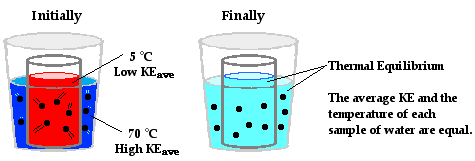
- The warmer body loses kinetic energy (cools down) while the cooler body gains kinetic energy (warms up)
- This continues until thermal equilibrium is reached
Transfer of Heat Between Objects
As we see in real life, heat travels from a hot object to a cold object until the two are in thermodynamic equilibrium (thermal equilibrium). Take for example, putting a pan up against hot grates on a stove that are heated up by a fire. The heat travels from the grates to the pan until the two are the same temperature.
Heat flows from the source (hot item) to the sink (cold item) through particle collisions, until thermal equilibrium is reached.
Thermal Equilibrium
Eventually, thermal equilibrium is reached as the particles continue to collide. At thermal equilibrium:
- The average kinetic energy of both bodies is the same
- Hence, their temperatures are the same
- There is no net transfer of energy between the bodies
- Particles still collide, but energy transfer in both directions is balanced
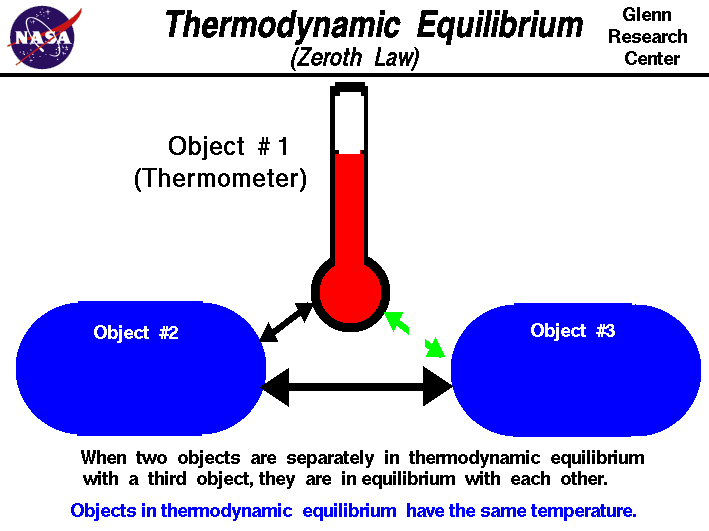
The Zeroth Law of Thermodynamics
The Zeroth Law of Thermodynamics is stated as "if a is in thermal equil. with b and b is in thermal equil. with c, then a is in thermal equilibrium with c" where a, b, and c are bodies.
Image Courtesy of NASAVocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
heat transfer | The process by which thermal energy is transferred between particles in thermal contact through collisions. |
kinetic energy | The energy possessed by particles due to their motion; greater in warmer bodies and lower in cooler bodies. |
molecular collisions | Interactions between particles that can result in the transfer of energy between them. |
temperature | A factor that influences reaction rate by affecting the kinetic energy and collision frequency of reactant molecules. |
thermal energy | The total kinetic energy of particles in a substance due to their random motion. |
thermal equilibrium | The state reached when two bodies in thermal contact have the same average kinetic energy and temperature, with no net transfer of thermal energy. |
Frequently Asked Questions
What is thermal equilibrium and how do you know when it happens?
Thermal equilibrium is the state two (or more) objects in thermal contact reach when they stop exchanging net heat: their particles have the same average kinetic energy and therefore the same temperature (CED EK 6.3.A.3). Microscopically this happens because collisions transfer energy from faster (hotter) particles to slower (cooler) ones until the Maxwell–Boltzmann distributions match. Practically you know it’s happened when thermometers read the same temperature (no temperature gradient) and no net heat flow occurs. The zeroth law formalizes this: if A is in thermal equilibrium with B, and B with C, then A and C are in equilibrium. On the exam you might be asked to explain heat transfer direction, identify when ΔT = 0, or relate average kinetic energy to temperature (learning objective 6.3.A). For a quick review, see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and try practice questions at (https://library.fiveable.me/practice/ap-chemistry).
Why do hot things cool down and cold things warm up when you put them together?
When you put a hot object next to a cold one, the faster-moving particles in the hot object collide with the slower ones in the cold object and transfer energy. Temperature measures the average kinetic energy of particles (CED 6.3.A.1), so these collisions move energy as heat from higher to lower temperature (CED 6.3.A.2). As collisions continue the average kinetic energies equalize, and you reach thermal equilibrium—the two things end up at the same temperature (CED 6.3.A.3). Mechanisms: conduction (direct particle collisions), convection (bulk fluid motion), and radiation (electromagnetic emission) all move heat; the direction is always from hotter to colder until equilibrium. Think of the Maxwell–Boltzmann spread: hot samples have a higher average and more high-energy particles, so net energy flows out until distributions match. For AP review, study Topic 6.3 in Fiveable’s guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between heat and temperature - I'm so confused about this?
Think of temperature as the measure, and heat as the energy that moves. Temperature tells you the average kinetic energy of particles in a system—higher T means particles move faster (CED EK 6.3.A.1). Heat (q) is the transfer of thermal energy between systems because of a temperature difference; it happens by particle collisions (conduction), bulk motion (convection), or radiation (CED EK 6.3.A.2). Heat has units of energy (J); temperature has units like °C or K and is an intensive property (doesn’t depend on amount). Heat is extensive (depends on mass): q = m·c·ΔT in calorimetry, so different masses or specific heats change how much heat flows for the same ΔT. Heat flows spontaneously from hot to cold until thermal equilibrium (same average kinetic energy, same T—zeroth law / CED 6.3.A.3). For AP-style problems, practice identifying sign of q (lost vs gained), using q = m c ΔT, and reasoning about direction of heat flow. Review the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and try practice problems (https://library.fiveable.me/practice/ap-chemistry) to solidify this.
How does heat transfer actually work at the molecular level?
At the molecular level, heat transfer is just energy moving from faster-moving particles to slower ones through collisions. Particles in a warmer object have a higher average kinetic energy (from the Maxwell–Boltzmann distribution), so when they collide with cooler particles they transfer some kinetic energy; that exchange is what the CED calls “transfer of energy as heat” (6.3.A). In solids this happens mainly by conduction (direct collisions and phonon/vibrational coupling), in fluids by convection (bulk motion carrying energy), and across space by radiation (electromagnetic waves). As collisions keep redistributing energy, the average kinetic energies of both bodies equalize—that’s thermal equilibrium and reflects the zeroth law: same temperature means same average kinetic energy. Thermal conductivity and temperature gradients control how fast equilibrium is reached. For AP review, connect this to 6.3.A and check the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain why particles with higher kinetic energy means higher temperature?
Temperature is a measure of the average kinetic energy of particles—not how fast one particular particle is moving, but the average across many particles. In a warmer object the molecules have a higher average speed, so their translational kinetic energy (½mv²) is larger. The Maxwell–Boltzmann distribution describes how particle speeds are spread out; raising temperature shifts that distribution to higher speeds and higher average KE. When two bodies touch, collisions transfer energy from faster (higher-KE) particles to slower ones—that transfer is heat—and after enough collisions they reach thermal equilibrium, meaning their average kinetic energies (and thus temperatures) are equal (CED 6.3.A.1–6.3.A.3). For AP Chem, remember temperature is in kelvin and tied to average KE (equipartition principle); this idea shows up in explanations of heat flow and equilibrium on free-response and multiple-choice items. For a short study refresher, see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
What happens during molecular collisions when two objects at different temperatures touch?
When two objects at different temperatures touch, their particles—those in the warmer object—have a higher average kinetic energy. When particles collide at the contact surface, faster (higher-energy) particles tend to transfer some kinetic energy to slower particles. Those collisions are the microscopic mechanism of heat transfer by conduction: energy flows from the hotter object to the colder one until their particles have the same average kinetic energy (thermal equilibrium) and thus the same temperature. The rate of that energy flow depends on the temperature gradient and the materials’ thermal conductivity; the Maxwell–Boltzmann distribution explains that only a fraction of particles carry the higher energies at any moment. This is exactly what AP CED learning objective 6.3.A describes—know that collisions transfer energy as heat and that equilibrium equalizes average kinetic energy. For a short study recap, see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt). For lots of practice problems, try Fiveable’s practice set (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how energy gets transferred between particles - can someone break this down?
Think of energy transfer as tiny billiard-ball collisions. Particles in a warmer object have a higher average kinetic energy (speed) than particles in a cooler one (CED 6.3.A.1). When those particles touch or interact, faster particles collide with slower ones and on average give some kinetic energy away—this microscopic exchange is heat transfer (CED 6.3.A.2). Repeated collisions change the particles’ speeds until both bodies share the same average kinetic energy (same temperature)—thermal equilibrium (CED 6.3.A.3). The Maxwell–Boltzmann distribution explains why not every collision transfers energy the same way, but net flow is from hot → cold. Modes: conduction = direct particle collisions, convection = bulk fluid motion moving energetic particles, radiation = photons (no contact). This idea appears on AP free-response and multiple-choice (use these terms on the exam). For a quick review and practice problems, see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and the unit page (https://library.fiveable.me/ap-chemistry/unit-6).
Why does thermal equilibrium always happen eventually and not just sometimes?
Thermal equilibrium always happens eventually because energy flows from faster (hotter) particles to slower (cooler) ones through collisions until their average kinetic energies match. Particles in the warmer body have a higher average kinetic energy (CED 6.3.A.1); when two systems are in thermal contact, collisions and interactions transfer energy (CED 6.3.A.2), shifting the Maxwell–Boltzmann distributions. Each collision has a chance to move a bit of energy from a faster particle to a slower one, so statistically the hot side loses energy and the cold side gains it. As this process repeats, temperature differences shrink and the net heat flow goes to zero—that’s thermal equilibrium (CED 6.3.A.3 and the zeroth law). Randomness guarantees that, on average, energy keeps redistributing until both systems share the same average kinetic energy. For a quick AP-aligned review, see the Topic 6.3 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I explain heat transfer in terms of particle collisions for the FRQ?
For an FRQ: describe heat transfer as energy moving by particle collisions. Say warmer-body particles have a higher average kinetic energy (6.3.A.1). When particles from the hot and cold objects collide, faster particles transfer some kinetic energy to slower ones—those collisions are heat transfer (6.3.A.2). State the direction: net energy flows from higher average KE (higher T) to lower until no net transfer. Explain thermal equilibrium: after many collisions the average kinetic energies equalize and temperatures are the same (6.3.A.3). A strong answer links particle behavior to temperature and uses phrases from the CED (average kinetic energy, thermal contact, thermal equilibrium, heat flow direction). For FRQ scoring, focus on clear causal language (“because,” “so”) and mention collisions and equalization of average KE. For a quick review, check the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and try practice FRQs on Fiveable (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between average kinetic energy and temperature of particles?
Temperature is a measure of the average translational kinetic energy of particles: as temperature (in kelvin) increases, the average kinetic energy of particles increases (equipartition theorem). So a warmer object has particles with a greater average KE than a cooler one (CED EK 6.3.A.1). When two bodies are in thermal contact, collisions let faster (higher-KE) particles transfer energy to slower ones—that transfer is heat—and after enough collisions both reach thermal equilibrium (CED EK 6.3.A.2–3). At equilibrium the average kinetic energies (and therefore temperatures) are equal even though individual particle speeds still vary (Maxwell–Boltzmann distribution). For AP-style answers, use kelvin for temperature and mention thermal equilibrium/heat flow direction; these ideas appear in Topic 6.3 (see the Topic 6.3 study guide: https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt). For extra practice, check Unit 6 materials (https://library.fiveable.me/ap-chemistry/unit-6) and AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do particles keep colliding even after thermal equilibrium is reached?
Thermal equilibrium means the average kinetic energy (temperature) of two bodies is the same, but particles still move and collide constantly. Collisions keep happening because molecules always have kinetic energy (they don’t stop at equilibrium). Each collision can transfer energy, but at equilibrium the amount of energy transferred one way equals the amount transferred the other way on average, so there’s no net heat flow. Think of the Maxwell–Boltzmann distribution: individual particle speeds fluctuate, but the distribution (and therefore the average kinetic energy/temperature) stays constant. This is exactly what the CED calls out in 6.3.A: collisions cause heat transfer, and at equilibrium collisions continue but with zero net energy exchange. If you want a quick refresher on these ideas for the AP, check the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and try practice questions (https://library.fiveable.me/practice/ap-chemistry).
How long does it take for two objects to reach thermal equilibrium?
There’s no single fixed time—how long until thermal equilibrium depends on the masses, specific heats, contact area, and how effectively heat transfers between them (conduction/convection/radiation). For many AP-level problems you can use a lumped-capacitance (Newton’s law of cooling) model: the temperature difference decays exponentially toward Teq, T(t) = Teq + [T(0) − Teq] e^(−t/τ), where τ (the time constant) ≈ (m c)/(h A). m = mass, c = specific heat, A = contact area, h = heat-transfer coefficient (depends on conduction/convection). Smaller m or c, larger A or h → shorter τ → faster equilibration. If you need an exact time for two finite objects in contact, you’d set up coupled energy balances (two ODEs) and solve; the same exponential approach appears but with a combined τ that depends on both m·c values and hA. For derivations and practice problems, see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and the AP Chemistry practice set (https://library.fiveable.me/practice/ap-chemistry).
What are some real world examples of heat transfer through molecular collisions?
Heat moves by particle collisions when faster (hotter) molecules bump into slower (cooler) ones, transferring kinetic energy until thermal equilibrium is reached. Real-world examples: - A metal spoon in hot coffee: vibrating metal atoms collide and conduct heat from coffee into the spoon (conduction). - A frying pan on a gas stove: hot atoms in the pan’s base transfer energy to cooler metal above and to the food by collisions at contact points. - Touching a warm radiator: skin molecules collide with the radiator surface, raising your skin’s average kinetic energy (you feel hot). - Warm air from a heater contacting a cold window: gas molecules hit the window surface, transferring heat and cooling the air near the glass (convection aids overall flow). These all rely on collisions and the Maxwell–Boltzmann idea that hotter bodies have higher average kinetic energy (CED 6.3.A). For more examples and AP-aligned notes, see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt). Need practice applying this on FRQs? Try problems at (https://library.fiveable.me/practice/ap-chemistry).
I missed the lab on thermal equilibrium - what were we supposed to observe?
In the lab you should’ve seen that when a hot object and a cold object are put in thermal contact (or stirred together), the hot one cools and the cold one warms until both reach the same temperature—thermal equilibrium. On a temp vs. time graph the hot sample’s temperature falls, the cold sample’s rises, and the two curves meet at a single equilibrium temperature. Microscopically, particles in the warmer body have a higher average kinetic energy and, through collisions at the interface (conduction, plus convection/radiation if applicable), energy transfers from hotter to cooler until their average kinetic energies—hence temperatures—are equal (CED 6.3.A, zeroth law). You should also note the direction of heat flow is always from higher to lower temperature and stops at equilibrium (no net heat flow). Review the topic study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and practice problems (https://library.fiveable.me/practice/ap-chemistry) to prep for AP-style questions.
How do I know if two substances have reached thermal equilibrium in an experiment?
You know two substances reached thermal equilibrium when their temperatures are the same and there’s no net heat flow between them. Microscopically that means particle collisions have redistributed energy so both bodies have the same average kinetic energy (CED 6.3.A.3). In the lab you confirm this by: (1) putting them in thermal contact (insulated), (2) measuring both temperatures with thermometers or probes, and (3) waiting until both readings become constant and equal. For calorimetry problems use qlost = qgain (mcΔT lost = mcΔT gained) to check the same final temperature. Watch for different heat capacities or masses—they change how much each shifts temperature. This is exactly the kind of reasoning AP asks for in Topic 6.3 (explain heat transfer/particle collisions). For a quick review see the Topic 6.3 study guide (https://library.fiveable.me/ap-chemistry/unit-6/heat-transfer-thermal-equilibrium/study-guide/WJd1kCvPeSS08sVOaDPt) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).