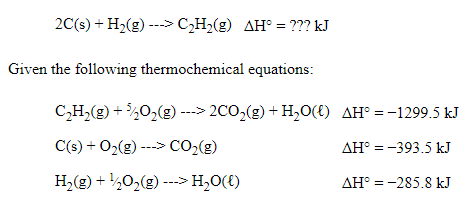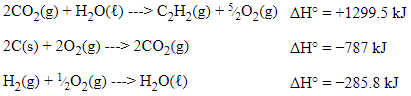Breaking Processes into Steps
Many chemical and physical processes can be broken down into a series of steps. Each step in the series has its own energy change (ΔH). This is a fundamental concept that allows us to use Hess's Law to calculate enthalpy changes for complex reactions.
For example, the formation of CO₂ from graphite can occur:
- Directly: C(s) + O₂(g) → CO₂(g)
- Or in steps:
- Step 1: C(s) + ½O₂(g) → CO(g)
- Step 2: CO(g) + ½O₂(g) → CO₂(g)

Energy Conservation and Hess's Law
The First Law of Thermodynamics states that energy is conserved - it cannot be created or destroyed, only transferred or converted. This principle is the foundation of Hess's Law.
Because total energy is conserved:
- Each individual reaction in a sequence transfers thermal energy to or from the surroundings
- The net thermal energy transferred in the sequence equals the sum of the thermal energy transfers in each step
- These thermal energy transfers result from potential energy changes among the species in the reaction sequence
- At constant pressure, the enthalpy change of the overall process equals the sum of the enthalpy changes of the individual steps
Hess's Law
Hess's Law states that the enthalpy change for a chemical reaction is the same regardless of the pathway taken from reactants to products. This means that if we know the enthalpy changes for different reactions, we can manipulate and combine them to find the enthalpy change for a new reaction. It's like taking different routes to the same destination - the total change in elevation is the same no matter which path you take! ✨
Rules of Hess's Law
- When a reaction is reversed, the enthalpy change stays constant in magnitude but becomes reversed in mathematical sign (Flipping the reaction flips the sign of ΔH).
- If an equation is multiplied by n, ΔH has to also be multiplied by n.
- When two (or more) reactions are added to obtain an overall reaction➕, the individual enthalpy changes of each reaction are added to obtain the net change of enthalpy of the overall reaction.
Representing Processes as a Sequence of Steps
When using Hess's Law, we represent a chemical process as a sequence of steps by:
- Identifying known reactions (steps) that contain the reactants and products of interest
- Manipulating these steps (reversing, multiplying) to match the target reaction
- Adding the steps together to obtain the overall process
Hess's Law Example Problem
Our general strategy is going to be manipulating these three simple reactions (our steps) and then adding them together to form the overall reaction. This demonstrates how we can represent a complex process as a sequence of simpler steps.
Thought Process
When I approach a Hess's Law question, I always find the reactants and products in the given thermochemical equations. This is what I would note:
- C (s) is a reactant in thermochemical equation #2 and in the overall equation, but there should be two atoms of carbon. Therefore, we have to manipulate the second equation and add a coefficient in front of the solid carbon. ❌
- H₂ (g) is a reactant in thermochemical equation #3 and the overall equation, and the coefficient is 1 in both equations. Therefore, we do not have to manipulate the third equation. ✔️
- C₂H₂ (g) is a reactant in thermochemical equation #1, but it is a product in the overall equation. This means we have to manipulate the first equation. ❌
Now that we have a general idea of what we are looking at, let's do the manipulations we know need to occur.
Steps to Take
Step 1: Flip reaction 1 to get C₂H₂ on the product side. If we flip the reaction, we also have to flip the sign on the enthalpy. Our manipulated first equation is as follows: 2CO₂ (g) + H₂O (l) → C₂H₂ (g) + 5/2O₂ (g) with ΔH° = +1299.5 kJ
Step 2: Multiply reaction 2 to get 2 solid carbon atoms. If we multiply the reaction, we have to multiply the enthalpy by 2 too✖️.
Whatever you do to the equation, you have to do the same to the enthalpy.
Results of Steps
After doing these things, we are left with:
Now, the equations are set up properly to create the overall equation, but what happens with the extra compounds?
You could think of these as spectator compounds; they cancel out since they are present on both sides of the equation when all 3 equations are added together.
With this being said, the O₂s cancel out, H₂O cancels out, and the CO₂s cancel out.
👉 Since we added the reactions together, we can do the same with their enthalpies. This follows rule #3!
ΔH = +1299.5 kJ + (−787 kJ) + (−285.8 kJ) = +226.7 kJ.
Example Problem #2
Calculate the value of ΔHreaction for the following reaction using the listed thermochemical equations.
Thought Process
Let's note a few things:
- P₄O₁₀ is on the wrong side of the equation, so we have to manipulate equation 2. ❌
- PCl₅ is also on the wrong side of the equation and it doesn't have a coefficient of 6, so we have to manipulate equation 3.❌
- Cl₃PO is on the right side of the equation, but it doesn't have a coefficient of 10 so we have to manipulate equation 4. ❌
Steps to Take
This problem has a lot more steps! Let's go through the manipulations we have to make:
Step 1: Flip equation 2 to get P₄O₁₀ on the other side. If we flip the equation, we also have to flip the sign on the enthalpy.
Step 2: Flip equation 3 to get PCl₅ on the other side and multiply equation 3 by 6 so PCl₅ has a coefficient of 6. In doing this, we have to multiply the enthalpy by 6 and negate it (two steps in one!)✖️.
Step 3: Multiply equation 4 by 10 so that Cl₃PO has a coefficient of 10. In doing this, we also have to multiply the enthalpy by 10✖️.
Results of Steps
After doing these things, we are left with:
Now, we have everything on the right side of the equation, and they all have the proper coefficients. Our last step is to check if all the spectators cancel out, so let's count them up!
| Compound | Reactant Side | Product Side | Is Manipulation Necessary? |
|---|---|---|---|
| P₄ | 1/4 | 1 | ✔️ |
| Cl₂ | 3/2 | 6 | ✔️ |
| PCl₃ | 10 | 7 | ✔️ |
| O₂ | 5 | 5 | ❌ |
Luckily, all 3 compounds that we have to balance are in equation 1, meaning we now have to manipulate it in order for the equations to add up to the overall equation.
🤔 What should we multiply equation 1 by? In order to get the same amount of each compound, we have to multiply equation 1 by 4 and its enthalpy by 4!
Now everything cancels out and we are left with the overall equation! The last step is to add up all the enthalpies. 🥳
👉 ΔH = -1225.6 kJ + 2967.3 kJ + 505.2 kJ + (−2857 kJ) = -610.1 kJ.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
chemical process | A transformation in which substances are converted into different substances through the breaking and forming of chemical bonds. |
energy change | The difference in energy between the initial and final states of a system during a process or reaction step. |
enthalpy | The total heat content of a system; at constant pressure, the enthalpy change equals the thermal energy transferred to or from the surroundings during a chemical or physical process. |
enthalpy change | The difference in enthalpy between products and reactants in a chemical or physical process, representing the heat absorbed or released. |
first law of thermodynamics | The principle that energy is conserved in chemical and physical processes; energy cannot be created or destroyed, only transferred or transformed. |
Hess's law | The principle that the enthalpy change of an overall reaction equals the sum of the enthalpy changes of the individual steps in the reaction sequence. |
physical process | A change in the state or properties of matter that does not alter the identity of the substances involved. |
potential energy | The stored energy in chemical bonds and molecular structures that can be released or absorbed during a reaction. |
thermal energy transfer | The movement of heat energy to or from the surroundings during a chemical or physical process. |
Frequently Asked Questions
What is Hess's Law and why do we need to learn it?
Hess’s Law says the enthalpy change (ΔH) for an overall reaction equals the sum of the ΔH’s for each step that adds up to that reaction. Because energy is conserved (1st law), you can break a process into steps, reverse or scale steps (flip sign if reversed, multiply ΔH when you multiply an equation), and add their ΔH values to get the net ΔH. We learn it because many reactions are hard to measure directly; Hess’s Law lets you find ΔH using known formation or combustion steps (standard enthalpies) or construct enthalpy cycles/diagrams. On the AP exam you’ll use these additivity rules and the reversal/scaling rules (CED 6.9.B.2) in calculations and free-response problems. Practice applying Hess’s Law with the topic study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and more problems at (https://library.fiveable.me/practice/ap-chemistry).
How does Hess's Law actually work in chemistry problems?
Hess’s Law just says enthalpy changes add—because energy is conserved. To use it: write the target reaction, then pick given thermochemical equations you can combine to make that overall equation. Apply three rules: if you reverse an equation, flip the sign of ΔH; if you multiply an equation by c, multiply ΔH by c; when you add equations, add their ΔH values. Manipulate the given steps (reverse and/or scale) until all intermediate species cancel and you’re left with the target reaction, then sum the adjusted ΔH values to get ΔH of the overall process. Practically: label each step’s ΔH, show algebraic cancellations of species, and write the final ΔH with units (kJ or kJ·mol⁻¹ as given). Hess problems on the AP expect clear step-by-step algebra and correct use of the reversal/scaling rules (CED 6.9.B). For guided examples, see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and more practice (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about enthalpy changes - do they add up or cancel out when you combine reactions?
They add algebraically—enthalpy changes follow Hess’s Law. If you combine steps, the overall ΔH is the sum of each step’s ΔH (CED 6.9.B.1, 6.9.B.2c). Two quick rules to use every time: if you reverse a reaction, flip the sign of ΔH; if you multiply a reaction by c, multiply ΔH by c (6.9.B.2a,b). So when you manipulate equations to make intermediates cancel, do the same math to their ΔH values and then add them to get the net ΔH. This works because energy is conserved (first law) and enthalpy is additive for a constant-pressure sequence. For worked examples and step-by-step practice, see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and try problems at Fiveable practice (https://library.fiveable.me/practice/ap-chemistry).
What happens to the enthalpy when you reverse a chemical reaction?
If you reverse a chemical reaction, the enthalpy change (ΔH) keeps the same magnitude but switches sign. So if the forward reaction has ΔH = −150 kJ (exothermic), the reverse reaction has ΔH = +150 kJ (endothermic). This is one of Hess’s law rules: reversing a step reverses the sign of its ΔH while conserving energy (first law of thermodynamics). Remember also that if you multiply an equation by a factor, you multiply ΔH by that factor, and when you add reactions you add their ΔH values. These ideas are tested on the AP exam under Topic 6.9 (Hess’s Law)—see the Topic 6.9 study guide for examples (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT). For more unit review and lots of practice problems, check the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6) and the AP Chem practice bank (https://library.fiveable.me/practice/ap-chemistry).
Why does flipping a reaction change the sign of delta H?
Because enthalpy change (ΔH) measures heat transferred when reactants become products at constant pressure, the sign tells you the direction of heat flow. If a forward reaction releases heat (exothermic, ΔH negative), running the exact same process backward would absorb that same amount of heat (endothermic, ΔH positive). Energy is conserved, so reversing the arrow simply reverses the direction of energy flow—same magnitude, opposite sign. Hess’s law formalizes this: when you reverse a reaction, you reverse the sign of its ΔH (CED principle 6.9.B.2a). That lets you add and subtract known ΔH values to build new reactions and get overall ΔH. This is a core AP Chem skill for Topic 6.9 and appears in free-response tasks: practice combining enthalpies and using the reversal rule (see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6)). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain the difference between individual reaction steps and the overall enthalpy change?
Think of individual steps as pieces of a story and the overall ΔH as the story’s total change in heat. Each step has its own enthalpy change (ΔH1, ΔH2, …)—you can reverse a step (flip the sign) or scale it (multiply ΔH by the same factor). Because energy is conserved (CED 6.9.B.1), the net thermal energy transferred for the whole sequence equals the sum of the step ΔHs. So the overall ΔH is just ΔH1 + ΔH2 + … (that’s Hess’s law and the reversal/scale rules from 6.9.B.2). You don’t need to know microscopic paths or mechanisms—only the initial and final states matter, so breaking a process into convenient steps makes calculating ΔH easier. For extra practice and diagrams (H vs. reaction progress), check the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6); there are lots of practice problems too (https://library.fiveable.me/practice/ap-chemistry).
How do I know when to multiply the enthalpy by a coefficient in Hess's Law problems?
Multiply the enthalpy when you change the amount of a reaction—Hess’s Law treats ΔH like any other quantity that scales with stoichiometry. If you multiply a thermochemical equation by a factor c (for example, 2 H2 + O2 → 2 H2O multiplied by 2), you must multiply ΔH by the same factor (rule 6.9.B.2.b in the CED). If you reverse a reaction, flip the sign of ΔH (rule 6.9.B.2.a). Then add the ΔH values of your adjusted steps to get the overall ΔH (rule 6.9.B.2.c). Quick check: if you need ΔH for forming 2 moles of a product and you only have ΔH for 1 mole, multiply ΔH by 2. If you’re combining given equations, first reverse/scale those equations to match the target overall equation, carrying the same sign/scale changes to each ΔH, then add. For extra practice and examples, see the Hess’s Law study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and more problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the connection between Hess's Law and the first law of thermodynamics?
Hess’s Law is basically a direct application of the first law of thermodynamics (energy conservation). The CED puts it simply: because total energy is conserved, the thermal energy transferred in a multi-step process equals the sum of the transfers for each step. At constant pressure those thermal transfers show up as enthalpy changes, so ΔH for an overall reaction = sum of the ΔH’s of the steps. That’s why you can reverse a thermochemical equation (flip the sign of ΔH), scale an equation (scale ΔH), and add equations (add ΔH’s) to get a target ΔH—all follow from energy conservation. This is exactly what you should be able to use on the AP: represent a process as steps (LO 6.9.A) and add enthalpies to get the net ΔH (LO 6.9.B). For practice problems and worked examples, see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and the Unit 6 overview (https://library.fiveable.me/ap-chemistry/unit-6).
I don't understand how breaking reactions into steps helps us calculate enthalpy - can someone explain?
Think of enthalpy like money: energy changes for simple steps add up to the total change for the whole process. By splitting a hard reaction into known steps (each with its ΔH), you can sum those ΔH values to get the overall ΔH because energy is conserved (first law of thermodynamics). That’s Hess’s law in practice. Use the three rules: reverse a step → flip the sign of ΔH; multiply a step → multiply ΔH by the same factor; add steps → add their ΔH’s. Breaking into steps is especially useful when the overall reaction’s ΔH isn’t measured directly but standard formation or combustion ΔH’s for the parts are available. This method is exactly what AP asks you to do on exams for Topic 6.9 (represent processes as sequences and use additivity of enthalpy). See the Topic 6.9 study guide for examples and worked problems (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT). For extra practice, check the AP Chem practice problem bank (https://library.fiveable.me/practice/ap-chemistry).
How do you add multiple reactions together to get the target equation using Hess's Law?
Think of Hess’s Law like algebra with chemical equations: you can reverse and scale given reactions so unwanted species cancel, and the ΔH values follow the same operations. Steps: 1. Write the target equation at top. 2. For each given reaction, decide if you need to reverse it (if you reverse, change sign of ΔH) or multiply it by a factor (multiply ΔH by same factor). 3. Add the adjusted reactions term-by-term; species that appear on both sides cancel. 4. The sum of the adjusted ΔH values equals ΔH of the target (conservation of energy). Quick example: if you need A → C but have A → B (ΔH1) and C → B (ΔH2), reverse the second to get B → C (ΔH = −ΔH2), then add: A → B + B → C = A → C, ΔH = ΔH1 + (−ΔH2). This matches the CED rules (reversal sign, scaling factor, additivity). For a focused review, see the Hess’s Law study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT). For more practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
What are the three main rules I need to remember for Hess's Law calculations?
Three rules to always use for Hess’s law problems (from the CED): 1) Reversal: if you flip a thermochemical equation, change the sign of ΔH (same magnitude, opposite sign). 2) Scaling: if you multiply an equation by a factor c, multiply ΔH by the same factor c. 3) Additivity: when you add two or more equations to get an overall reaction, add their ΔH values to get the net ΔH. Remember why: total energy is conserved (first law), so enthalpy changes for steps add up to the overall ΔH (6.9.B.1 and 6.9.B.2 in the CED). Use those three moves—reverse, scale, add—to manipulate given equations until their sum matches the target reaction, then combine the ΔH values. Want extra practice? Work through the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and tons of practice questions (https://library.fiveable.me/practice/ap-chemistry).
Why is Hess's Law useful in real chemistry applications?
Hess’s Law is super useful because it lets you get the enthalpy change (ΔH) for reactions you can’t measure directly by adding known ΔH values for related steps—exactly what the CED calls the additivity of enthalpy (6.9.B.1 & 6.9.B.2). In real chemistry that means you can: use standard enthalpies of formation or combustion to build an enthalpy cycle, combine or reverse thermochemical equations (flip the sign if reversed, scale ΔH when you multiply an equation) and find ΔH for hard-to-run or dangerous reactions. That’s how chemists estimate energy costs for industrial processes, check calorimetry results, or predict if a reaction will release/absorb a lot of heat. For an AP-focused refresher (rules, sample problems, and diagrams), see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT). For more practice, Fiveable has hundreds of AP Chem problems (https://library.fiveable.me/practice/ap-chemistry).
I missed the lab on Hess's Law - what's the basic concept I need to understand for the test?
Hess’s Law: total ΔH for a process = sum of the ΔH values for the steps. It comes from energy conservation (first law): enthalpy is additive at constant pressure. Key rules to use on the test: if you reverse a reaction, flip the sign of ΔH; if you multiply a reaction by c, multiply ΔH by c; if you add reactions to get an overall reaction, add the ΔH values. Practically, you’ll be given thermochemical equations or formation/combustion ΔH° values and asked to combine, reverse, and scale them to get the target reaction (enthalpy cycle / H vs. reaction-progress diagrams may help visualize). Problems often require algebraic manipulation and unit labels—show the steps you used to flip/scale each ΔH. Review the Topic 6.9 study guide for worked examples (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and practice lots of problems (https://library.fiveable.me/practice/ap-chemistry)—that repetition is what makes this stick.
How do I set up a Hess's Law problem step by step?
Step-by-step for solving a Hess’s Law problem: 1. Write the target (overall) reaction clearly. 2. List the given thermochemical equations and their ΔH values. 3. Use the CED rules: if you reverse an equation, flip the sign of ΔH; if you multiply an equation by c, multiply ΔH by c. 4. Algebraically combine (add/subtract) the modified given equations so that all reactants and products cancel except those in the target reaction. Keep track of ΔH for each modified step. 5. Add the modified ΔH values—the sum is ΔH for the target (additivity of enthalpy; conservation of energy). 6. Check atoms and charge balance and re-check sign arithmetic. 7. If needed, convert per-mole vs. given-amounts (scale ΔH to moles of interest). Remember AP specifics: show the manipulations (reversals/scaling) and the ΔH adjustments—graders expect those steps (CED 6.9.B). For a quick refresher and examples, see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and more practice at (https://library.fiveable.me/practice/ap-chemistry).
What's the relationship between potential energy changes and enthalpy in multi-step reactions?
Potential energy changes of species in each step show up as heat transferred to/from the surroundings; at constant pressure those transfers are enthalpy changes. So for a multi-step process the overall ΔH equals the sum of the ΔH values for the individual steps—energy is conserved (first law). That’s Hess’s law: reverse a step and flip the sign of ΔH; multiply a step and scale ΔH the same; add steps and add their ΔH’s. In practice you can build an enthalpy cycle or H vs. reaction-progress diagram and just add vertical changes to get the net ΔH. This is exactly what AP Topic 6.9 tests (represent sequences and use additivity rules from the CED). For more worked examples and an H-diagram review, see the Topic 6.9 study guide (https://library.fiveable.me/ap-chemistry/unit-6/hess-law/study-guide/p9ryCGfaOvpZj0Qye5eT) and related Unit 6 resources (https://library.fiveable.me/ap-chemistry/unit-6). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).