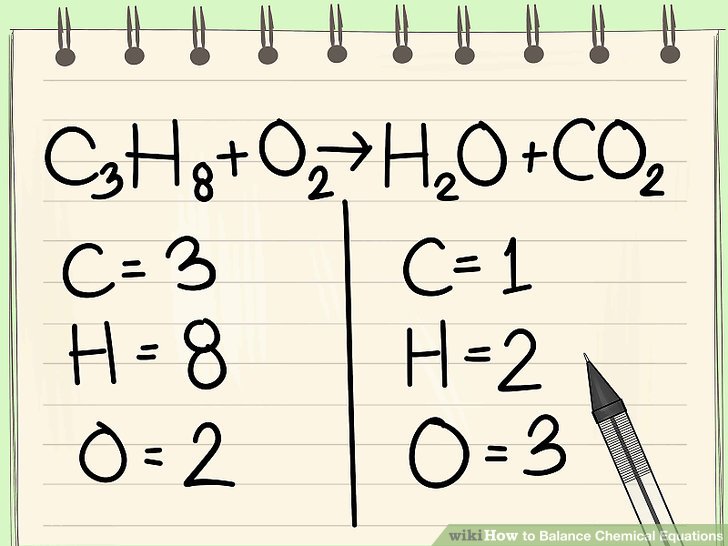Chemical reactions can be represented in multiple ways: through chemical equations, net ionic equations, and particulate models. Just as net ionic equations provide information about interactions with ions in aqueous solutions, chemical reaction equations are a great way to visually track physical changes and chemical reactions.
Before we can examine these equations to observe these changes, we need to learn how to properly set up the equation for a chemical reaction. In other words, we need to learn how to balance equations. If an equation isn't balanced, our answers will be incorrect. But why is it bad if it's unbalanced, and how can we learn to balance equations easily?
Image Courtesy of Quizizz
Why Do We Balance Reactions?
The law of conservation of mass is a fundamental principle in chemistry that states that the amount of matter stays constant in a closed system. In short, matter cannot be neither created nor destroyed in thin air.
A chemical reaction is a closed system, so the number of atoms that are produced must be equal to the number of atoms put in. The atoms can still rearrange, though, which is all a chemical reaction really does! In conclusion, we need to make sure that the number of elements in the reactants is the same as those in the products, even if the products are different than what we started with.
Let’s look at an example to make sense of this.
Example #1
Here’s the chemical equation that represents the synthesis of carbon dioxide: CO (g) + O₂ (g) → CO₂ (g)
Step 1
Get a general feel of the equation, and make sure it’s not already balanced. Take a look at the products and the reactants. Note the number of each element on both sides. If the number of one element is equal on both sides, it’s balanced.
In this case, there is 1 carbon atom on both sides, so carbon is balanced. However, there are 3 oxygen molecules on the reactant side and 2 on the product side, so oxygen is not.
Step 2
Look at the elements that are only in one compound on both sides of the equation and are already balanced. In this case, it’s carbon. From this, we know that both of those compounds with carbon will have to have the same coefficient. Thus, we can leave the coefficients of CO and CO₂ as 1 for now.
Remember that we can only change the coefficients of the molecules. If we change the subscript, we would change the molecule completely. For example, 2NO2 represents two molecules of nitrogen dioxide, but N2O4 represents one molecule of dinitrogen tetroxide. Coefficients should always be a whole number.
Step 3
Find elements that are only in one compound on both sides but have different numbers of atoms on each side. Balance those elements. In our example, we don’t have any in this category (don’t worry 😌, the next example will have one!).
Step 4
Look for elements that appear in multiple compounds on one side. Balance those elements. In our equation, that’s oxygen. Since there is less oxygen on the product side, let’s increase the coefficient of CO₂ to 2:
CO (g) + O₂ (g) → 2CO₂ (g)
As stated in step two, since both sides have an equal number of carbon, both CO and CO2 must have the same coefficient so that carbon remains balanced. Thus, we have to increase the coefficient of CO to 2 as well.
2CO (g) + O₂ (g) → 2CO₂ (g)
Now let’s make sure it really is balanced:
| Reactants | Products |
|---|---|
| Carbon: 2 | Carbon: 2 |
| Oxygen: 4 | Oxygen: 4 |
Sweet! Both the reactants and products have the same number of each atom. We followed the Law of Conservation of Mass! 🥳
Example #2
Here’s the equation: Li (s) + N₂ (g) → Li₃N (s)
Step 1
Like before, let’s make sure the equation’s not already balanced. Take a look at the products and the reactants and note the number of atoms of each element on both sides.
There is 1 lithium molecule on the reactant side and 3 on the product side. There are 2 nitrogen molecules on the reactant side and 1 on the product side. Neither lithium nor nitrogen is balanced, so we must find a way to balance both.
Step 2
Look at the elements that are only in one compound on both sides of the equation and have the same number of atoms on both sides. Balance those elements. In our example, no such element exists, meaning no matter what we change, we will not have to make sure that we are unbalancing an element (like we would have carbon in the last example).
Step 3
Find elements that are only in one compound on both sides but have different numbers of atoms on each side. Balance those elements. In our example, both lithium and nitrogen fall into this category. In this situation, it’s more like a guess-and-check, but we can go about it strategically.
Since there are 3 lithium molecules on the right, let’s increase the coefficient of Li to 3.
3Li (s) + N₂ (g) → Li₃N (s)
Lithium is all balanced! Now, looking at nitrogen, we notice that there are 2 on the left and only 1 on the right. Let’s increase the coefficient of Li₃N to 2.
3Li (s) + N₂ (g) → 2Li₃N (s)
Nitrogen looks good! But wait…lithium is no longer balanced! Fear not, we have the power to change coefficients until the chemical equation is completely balanced. Since there are 6 molecules of lithium on the right, let’s increase the coefficient of Li to 6 on the reactant side.
6Li (s) + N₂ (g) → 2Li₃N (s)
Step 4
Look at the elements that are in more than one compound. In this example, there is no element.
Time to double-check our work. Try to always do this, as you may have "unbalanced" one element when trying to balance another.
| Reactants | Products |
|---|---|
| Lithium: 6 | Lithium: 6 |
| Nitrogen: 2 | Nitrogen: 2 |
Steps to Balancing Equations
Woohoo! We balanced another equation! It’s probably evident that the more practice you do, the better you’ll be at balancing equations. Be sure to keep the steps we used in mind:
- Make sure the equation isn’t already balanced
- Find the elements that are only in one compound on both sides and have the same number of atoms on both sides. These must have equal coefficients.
- Look at the elements that are only in one compound on both sides and have different numbers of atoms on both sides. Balance them.
- Look at the elements that are in more than one compound. Balance them.
- Finally, double-check your answer and make sure that the number of element X on the reactants side is the same as on the products side. The key to this is satisfying the law of conservation of mass.
Particulate Representations of Reactions
Balanced chemical equations can be translated into particulate models - visual representations showing individual atoms, molecules, or ions before and after a reaction. These models help us visualize what's happening at the molecular level during a chemical reaction.
Key Principles for Particulate Models
-
Conservation of atoms: Just like in balanced equations, the same number of each type of atom must appear on both sides
-
Proper representation:
- Atoms are shown as spheres (often color-coded by element)
- Molecules show atoms bonded together
- Ions can be shown with + or - charges
-
Consistent scale: Use the same relative sizes and colors for atoms throughout
Example: CO + O₂ → CO₂
Let's visualize our first balanced equation: 2CO (g) + O₂ (g) → 2CO₂ (g)
In a particulate model:
- Before reaction: Show 2 CO molecules (each with 1 carbon bonded to 1 oxygen) and 1 O₂ molecule (2 oxygens bonded together)
- After reaction: Show 2 CO₂ molecules (each with 1 carbon bonded to 2 oxygens)
- The total count: 2 carbon atoms and 4 oxygen atoms on each side
Example: Formation of Lithium Nitride
For the reaction 6Li (s) + N₂ (g) → 2Li₃N (s):
- Before reaction: Show 6 separate Li atoms and 1 N₂ molecule (2 nitrogens bonded)
- After reaction: Show 2 formula units of Li₃N (each containing 3 Li⁺ ions and 1 N³⁻ ion in an ionic structure)
- Note how the atoms rearrange but the total number remains constant
Physical Processes vs. Chemical Reactions
Particulate models can also represent physical processes:
- Dissolving: Show solid ionic compound separating into individual ions surrounded by water molecules
- Phase changes: Show particles getting closer (condensation) or farther apart (evaporation)
- Mixing: Show different types of molecules distributed throughout the space
In physical processes, the chemical identity doesn't change - only the arrangement or state of the particles changes.
Tips for Drawing Particulate Models
-
Start with the balanced equation - this tells you exactly how many of each particle to draw
-
Use a legend - show what each colored sphere represents
-
Keep it simple - you don't need to show every detail, just the essential features
-
Check conservation - count atoms before and after to ensure nothing was created or destroyed
-
Show proper bonding - molecules should have atoms connected, while separated ions should be apart
Review Activity
Part A: Balance the following equations and include the states of matter of both reactants and products:
- Na₃PO₄ + AgNO₃ → Ag₃PO₄ + NaNO₃
- A reaction between iron (III) oxide and carbon monoxide
- The combustion of ethane (C₂H₆)
- The synthesis of sulfur trioxide
- The decomposition of potassium chlorate
Part B: Practice with Particulate Models
- Draw a particulate representation of the synthesis of water: 2H₂ (g) + O₂ (g) → 2H₂O (g)
- Given a particulate diagram showing 4 NH₃ molecules and 3 O₂ molecules as reactants, draw the products after the reaction (assuming complete reaction)
- Sketch a particulate model showing NaCl dissolving in water (physical process)
Answers to Balancing Practice Problems
Question 1) Na₃PO₄ (aq) + 3AgNO₃ (aq) → Ag₃PO₄ (s) + 3NaNO₃ (aq)
Question 2) Fe₂O₃ (s) + 3CO (g) → 2Fe (s) + 3CO₂ (g)
Question 3) 2C₂H₆ (g) + 7O₂ (g) → 4CO₂ (g) + 6H₂O (l)
Question 4) 2SO₂ (g) + O₂ (g) → 2SO₃ (g)
Question 5) 2KClO₃ (s) → 2KCl (aq) + 3O₂ (g)
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
balanced chemical equation | A chemical equation where the number of atoms of each element is equal on both the reactant and product sides. |
chemical reaction | A process in which substances are transformed into different substances through the breaking and forming of chemical bonds. |
particulate model | A representation of matter showing individual atoms, molecules, or ions and their interactions to describe chemical processes at the molecular level. |
physical process | A change in the state or properties of matter that does not alter the identity of the substances involved. |
symbolic representation | A depiction of chemical reactions using chemical formulas, symbols, and equations to represent reactants and products. |
Frequently Asked Questions
What's the difference between a balanced chemical equation and a particulate diagram?
A balanced chemical equation is a symbolic, macroscopic description: formulas, stoichiometric coefficients, and phase symbols that show reactants → products while conserving atoms and charge (e.g., molecular, complete ionic, or net ionic forms). A particulate diagram is a microscopic picture showing individual atoms, ions, or molecules (dots, circles, or shapes) before and after the reaction—showing how particles collide, rearrange, which ions stay as spectators, and the actual count of species that matches the coefficients. Key differences: equations give stoichiometry and conservation of mass numerically; particulate diagrams show spatial arrangement, identity of spectator ions, and whether species are dissociated in solution. On the AP exam you’re expected to translate between these representations (CED 4.3.A.1): check that particle counts match coefficients and that charges/mass are conserved. For practice and worked examples, see the Topic 4.3 study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4). For more practice problems, go to Fiveable’s AP Chem practice page (https://library.fiveable.me/practice/ap-chemistry).
How do I draw molecules and atoms in a chemical reaction diagram?
Think of a chemical reaction diagram as a “particulate translation” of the balanced equation: show the same kinds and numbers of atoms/ions before and after, use groupings to show molecules, and include phase/aqueous dissociation when relevant (that’s exactly what the CED expects for 4.3.A). Practical tips: - Atoms = colored or labeled dots; molecules = clusters of connected dots (H2 as two H dots joined; H2O as one O with two Hs). - Stoichiometric coefficients = number of particle drawings (3 H2 → draw three H2 molecules). - For aqueous salts, draw dissociation into ions for complete ionic diagrams, then cancel identical spectator ions to get the net ionic. Label spectator ions. - Include phase symbols (s, l, g, aq) near the equation and show bonds or proximity to indicate collisions if illustrating kinetics. - Always conserve atoms/charge between left and right. Want guided examples and practice? See the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to show particles when we already have the chemical equation?
A balanced chemical equation tells you what reacts and what forms, but a particulate (particle-level) picture shows what’s actually happening to atoms, ions, and molecules—which is exactly what Learning Objective 4.3.A expects you to do on the AP exam (translate symbolic equations into consistent particulate models). Particle drawings reveal collisions, spectator ions, dissociation in aqueous solution, and which species change oxidation state or form a precipitate. That matters for writing complete ionic and net ionic equations, identifying spectator ions, and explaining why a precipitate, gas, or neutral molecule forms (keywords from the CED: aqueous dissociation, spectator ions, precipitation reaction, stoichiometric coefficients). Use particle models to check conservation of atoms and charge and to visualize collision frequency for kinetics questions. For practice, work problems in the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4); you’ll also find lots of practice questions at (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain how to convert a chemical equation into a particle model?
Start by reading the balanced equation and phase symbols—that tells you which species are molecules, ions, solids, liquids, or gases. Then follow these steps: 1. Identify aqueous electrolytes and dissociate them into ions (complete ionic form). E.g., 2 NaCl(aq) → 2 Na+(aq) + 2 Cl−(aq). Use solubility rules for precipitation. 2. Keep stoichiometric coefficients equal to particle counts: coefficient 2 means draw two of that particle. 3. For molecular species (H2O(l), CO2(g), O2(g)), draw whole molecules, not separated atoms. For ionic solids (AgCl(s)), show neutral lattice or paired ions that form precipitate. 4. Remove spectator ions to get the net ionic equation and draw only reacting particles for the particulate net-ionic model. 5. Show collisions/arrangements if asked (orient molecules, show electrons or charge where relevant) and conserve atoms and charge. On the AP exam this maps to LO 4.3.A (representing reactions with particulate models). For a step-by-step example and practice, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and try problems from the unit page (https://library.fiveable.me/ap-chemistry/unit-4) or the 1000+ practice questions (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about how to represent ionic compounds in particulate diagrams - do I draw the individual ions?
Short answer: yes—draw the individual ions, but how you draw them depends on phase and context. - Solid ionic compound (s): show a repeating lattice of cations and anions arranged together (no free ions). Indicate ratio set by formula unit (e.g., two Na+ for every S2– in Na2S). - Aqueous solution (aq): show dissociated, separate ions (Na+, Cl–) spaced apart and ideally surrounded by water or small “solvation” markers. Include charges and correct stoichiometric counts. - When translating equations to complete ionic or particulate diagrams, show all soluble strong electrolytes as ions, label spectator ions, and cancel them to get the net ionic picture (CED keywords: complete ionic equation, net ionic equation, spectator ions, aqueous dissociation). - Always keep charges, stoichiometric coefficients, and phase symbols consistent to conserve mass and charge. For a quick walkthrough and examples, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM). For extra practice, Fiveable has hundreds of AP Chem problems (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when they ask for a "consistent particulate model" on the AP exam?
A "consistent particulate model" means your drawing or description of atoms, ions, and molecules must match the symbolic equation exactly—species, phases, charges, and stoichiometric coefficients—and obey conservation of mass and charge. Practically: if the equation shows NaCl(aq) break into Na+ and Cl–, your particulate sketch must show dissociated ions (not intact NaCl molecules). Show spectator ions separately in a complete ionic picture and remove them for a net ionic model. Use correct counts of particles for stoichiometric coefficients, include phase symbols (s, l, g, aq) in your reasoning, and, when relevant, show collisions or bonds forming/breaking to indicate reaction mechanism. AP graders expect this translation (CED LO 4.3.A). For worked examples and tips, check the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I know how many of each atom to draw in my particle representation?
Start from the balanced chemical equation—the stoichiometric coefficients tell you how many formula units (molecules or formula units) to represent in your particulate model. Make sure your drawing obeys conservation of mass (same total number of each element on both sides). Steps: 1. Balance the equation. The coefficients = number of particles to draw. (e.g., 2 H2 + O2 → 2 H2O means draw two H2 molecules, one O2, and two H2O molecules → total atoms: 4 H and 2 O on both sides.) 2. For aqueous reactions, write the molecular equation, then dissociate strong electrolytes into ions for a complete ionic particulate model. Draw ions separately (Na+, Cl–) and label spectator ions—they appear unchanged on both sides and aren’t in the net ionic diagram. 3. For net ionic drawings, omit spectators and show only species that actually change (precipitate, gas, acid–base protons, etc.). AP tip: the CED expects a consistent particulate model that matches balanced symbolic equations (stoichiometric coefficients, aqueous dissociation, spectator ions). For extra practice, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM), Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and plenty of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do some particle diagrams show molecules as connected spheres and others show them separately?
Good question—those pictures are just showing two different particulate models. - Connected spheres mean atoms are bonded together as one molecule (a molecular representation). Use that when the symbolic equation shows a covalent molecule or an undissociated product (e.g., H2O, CO2). - Separate spheres show individual particles not bonded to each other: free ions in solution (complete ionic or net ionic representations), atoms after dissociation, or uncombined reactants. That style highlights spectator ions and aqueous dissociation (important for writing complete and net ionic equations). On the AP CED this maps to 4.3.A—you should be able to translate a balanced equation into a consistent particulate model (molecular vs ionic vs collision diagrams). Pay attention to phase symbols and stoichiometric coefficients: they tell you whether to draw bonded molecules or separate ions. For practice, check the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and more problems at (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between representing a physical change and a chemical change with particles?
For a particulate representation, the key difference is whether the identities of the particles change. For a physical change (e.g., melting, dissolving) you show the same molecules/ions before and after—maybe rearranged, separated, or with different spacing, but with no new species formed. For a chemical change you must show bonds broken and new bonds formed: different molecular species or ions appear (use molecular, complete ionic, or net ionic particulate models to show that). Always conserve atoms and charge (stoichiometric coefficients) and indicate phase/solubility (aq, s, g, l). For aqueous reactions, show dissociated ions and label spectator ions in complete ionic diagrams, then remove them for the net ionic particulate picture. On the AP exam, you may be asked to translate balanced symbolic equations into consistent particulate models and identify spectators/precipitate—practice doing molecular → complete ionic → net ionic (CED 4.3.A; keywords: spectator ions, aqueous dissociation, precipitation). For a quick refresher, check the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM), unit overview (https://library.fiveable.me/ap-chemistry/unit-4), and extra practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I represent aqueous solutions in particulate models - do I need to show water molecules?
Short answer: usually no—you don’t have to draw every water molecule in a particulate model of an aqueous solution. For AP Topic 4.3 you should show the dissolved species that matter for the reaction: separated ions labeled (aq), intact molecules that don’t dissociate, and any protons or neutral species involved in the chemistry. Water can be shown implicitly (use (aq) phase labels) or explicitly only when you need to show solvation/hydration shells, hydrogen-bonding, or a proton transfer mechanism (e.g., acid–base collisions). Tips tied to the CED: translate the balanced equation into a particulate view that conserves mass and stoichiometry (LO 4.3.A). For complete ionic particulate models show all ions present; for net ionic remove spectator ions. If you draw particles, keep correct counts (stoichiometric coefficients) and show collisions for reactive encounters. For more examples and practice, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how to show the before and after of a reaction using particle diagrams.
Think of a particle diagram as a “count-and-type” picture that must match the balanced equation and the particulate model in the CED (LO 4.3.A). Steps to show before and after: - Start with the balanced chemical equation and stoichiometric coefficients. Draw that many particles of each species (molecules or ions). Conservation of mass means number and type of atoms must match before and after. - For aqueous reactants, show dissociation into ions (complete ionic form) and draw spectator ions separately. Then remove spectators to show the net ionic diagram (precipitates or new molecules shown). - Use different shapes/colors for atom types and label phases (s, l, g, aq). For a precipitation: show separated ions before, then clustered pairs forming solid particles after. - For gas evolution or acid–base, show H+ transfer or new gas molecules appearing. - If asked on the exam, explicitly state which representation you’re drawing (molecular, complete ionic, or net ionic) and keep counts clear. Practice by converting 3 representations for one reaction—molecular → complete ionic → net ionic. For step-by-step examples, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
What are the rules for drawing particulate representations of chemical reactions?
Draw particulate diagrams so they match the symbolic equation exactly—same species, same stoichiometric ratios, and the same total number of atoms (conservation of mass). Key rules: - Start with a balanced chemical equation (include phase symbols). Use coefficients to set how many particles of each kind appear. - For aqueous strong electrolytes, show dissociation into individual ions in the particulate and complete ionic pictures (keep spectator ions separate). For net ionic, omit spectators and show only reacting ions. - Represent molecules vs. ions differently (neutral shapes for molecules, charged spheres or labeled ions for ions). Include charges and solvation if important (e.g., hydrated Na+). - Keep relative sizes/labels consistent so you can count atoms and charges at a glance. - Show gas evolution, precipitation, or bond-breaking/forming clearly (e.g., new particle groupings after reaction). - Use collisions/arrows if asked to show mechanism or collision representation. These are exactly the skills tested in Topic 4.3—see the AP study guide for examples (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and practice questions (https://library.fiveable.me/practice/ap-chemistry).
How do I represent gases vs liquids vs solids differently in particle models?
For particle models you show phase by how particles are drawn and how they move: - Solid: tightly packed, regular lattice, little vibrational motion arrows only. Draw each formula unit/molecule close together in fixed positions to show low kinetic energy and definite shape/volume. Use (s) in the equation. - Liquid: particles close but disordered with short random motion arrows and sliding past neighbors. Show no long-range order, variable spacing, and more collisions than a solid. Use (l). - Gas: particles far apart, randomly spaced, straight-line motion with frequent elastic-collision marks (arrows), large distances between particles to show high kinetic energy and compressibility. Use (g). Always keep the particulate diagram consistent with the balanced equation (stoichiometric coefficients = number of particles), conserve atoms, and show spectator ions if doing ionic/net ionic drawings. For collisions/kinetics questions, show arrows and collision geometry to explain reaction likelihood. For AP review, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4). For extra practice, try problems at (https://library.fiveable.me/practice/ap-chemistry).
Can you show me step by step how to go from 2H₂ + O₂ → 2H₂O to a particle diagram?
1) Count atoms from the balanced equation: 2 H2 gives 4 H atoms total; 1 O2 gives 2 O atoms total → products must have 4 H and 2 O. 2) Decide particle icons: pick a small circle (H) and a larger circle (O). Draw H2 as two small circles connected, O2 as two large circles connected. (Use different colors if you can.) 3) Sketch reactant side: draw two H2 molecules (so 2 pairs of H–H) and one O2 molecule (one O=O). Label phases if needed. This shows stoichiometric coefficients visually (2 H2, 1 O2). 4) Rearrangement to products: pair each O with two H atoms to make H–O–H. Since you had 4 H and 2 O, you can form two H2O molecules. Draw two H–O–H molecules (each O bonded to two H). 5) Check conservation of mass: same number of H and O atoms on both sides; same number of molecules when coefficients considered. Optionally show a “collision” arrow from reactants to products to emphasize reaction representation. This is exactly what Topic 4.3 asks: translate a balanced equation into a consistent particulate model (CED 4.3.A.1). For more examples and practice, see the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and tons of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do my particle diagrams need to match the coefficients in the balanced equation?
Because a balanced equation encodes the exact mole (and therefore particle) ratios required by conservation of mass, your particle diagram has to show those same ratios. Stoichiometric coefficients tell you the relative number of molecules (or ions) that react and form—so if the equation is 2 H2 + O2 → 2 H2O, your diagram must show twice as many H2 particles as O2 and end with the correct number of H2O molecules. Matching coefficients also keeps atom counts and charge balanced (important when you draw complete or net ionic particulate models and identify spectator ions). On the AP exam, Topic 4.3 expects you to translate balanced symbolic equations into consistent particulate models (CED 4.3.A.1); graders look for correct particle ratios, phases, and conservation of atoms/charge. For extra practice, check the Topic 4.3 study guide (https://library.fiveable.me/ap-chemistry/unit-4/representations-reactions/study-guide/CzoUpQyKbK27GRGVfXFM) and the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and try practice problems (https://library.fiveable.me/practice/ap-chemistry).