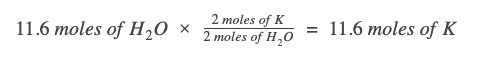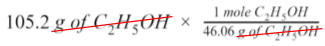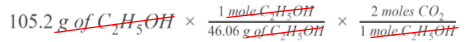What Is Stoichiometry Useful For?
In the previous section, we looked at qualitatively analyzing chemical reactions. Now, we’ll learn how to quantitatively analyze reactions using stoichiometry! It may seem like a lot of math at first, but once you do more practice, you’ll be more confident and become a stoichiometry master

Key Concepts to Understanding Stoichiometry
In earlier units, you learned about moles, molar mass/molar volume, molarity, and Avogadro's number. Stoichiometry is all about using mole ratios and these measurements and manipulating them to get to our desired unit. First, let's recap some key concepts to understand in stoichiometry:
- Balanced chemical equation: A balanced chemical equation is a written representation of a chemical reaction that shows the reactants on the left side and the products on the right side, with the number of atoms of each element balanced on both sides of the equation.
- Mole: The mole is a unit of measurement in chemistry that represents the amount of a substance. One mole of a substance is equal to Avogadro's number, which is 6.022 x 10^23 atoms or molecules.
- Stoichiometric coefficients: The coefficients in a balanced chemical equation represent the relative amounts of the reactants and products in a chemical reaction. The coefficients can be used to calculate the amount of reactants and products that are required or produced in a chemical reaction.
- Stoichiometric calculations: Stoichiometric calculations involve using the balanced chemical equation and the mole concept to predict the amount of reactants and products that are required or produced in a chemical reaction. These calculations are based on the principle of the conservation of mass, which states that the total mass of the reactants in a chemical reaction is equal to the total mass of the products.
As you've probably noticed, stoichiometry is used specifically to quantify the amount of reactants and products used in a chemical reaction. Therefore, it becomes especially helpful in the laboratory and becomes second nature to chemists.
Mole Ratios
A mole ratio is a ratio of the amounts of two or more substances in a chemical reaction, expressed in moles. Without a mole ratio, it'd be difficult to use stoichiometry to quantify values in the laboratory. It’s important to comfortably know the relationship between certain units as we do mole ratios. Below is a list of these measurements:
- At STP, one mole of an ideal gas fills 22.4 L of volume. This is also known as molar volume and this value can be found on the AP Chemistry reference sheet.
- As discussed above, one mole = 6.022 x 10^23 particles. Avogadro's number is also on the AP Chemistry reference sheet.
- One mole is represented by the molar mass (the number of grams there are in a mole). Each element's molar mass is on the periodic table. If we take a look at hydrogen, there are 1.008 grams in one mole.
You can find mole ratios by looking at a chemical reaction. In the reaction below, 1 mole of C2H5OH (ethanol) and 1 mole of oxygen gas are needed to produce 2 moles of carbon dioxide and 3 moles of water. A sample mole ratio would be 1 mole of O2 to 2 moles of CO2. Keep in mind that 2 moles of CO2 to 1 mole of O2 would mean the same thing even though this may seem backward.
Practice Stoichiometry Problems
Question 1
How many moles of potassium metal is required to fully react with 11.6 moles of water?
Step 1
Write the chemical reaction, if it’s not given. Be sure to balance it to satisfy the law of conservation of mass. 2K (s) + 2H₂O (l) → 2KOH (aq) + H₂ (g)
Step 2
Identify the known measurement. In this case, we’re given the value of 11.6 moles of wate.
Step 3
Since our given value is already in moles, we just need to write the appropriate mole ratio. We want a ratio that cancels out the known measurement units (moles of H2O) and brings in the unit we want (moles of K).
Looking at the chemical equation, for every 2 moles of H2O, we need 2 moles of K metal.
Note: Writing out the units and the molecule the value belongs to in the dimensional analysis will help you exponentially. Please do it!
One way to make sure you’re writing the correct mole ratio is to keep track of the units in your work. Since we need the unit "moles of K," we have to ensure that every other unit gets canceled out:
Question 2
If you have 105.2 g of ethanol (C₂H₅OH), what is the maximum volume of carbon dioxide that can form at STP?
Step 1
Write the chemical equation and balance it: C₂H₅OH (l) + 3O₂ (g) → 2CO₂ (g) + 3H₂O (l)
Step 2
Identify the known measurement: 105.2 g of ethanol.
Step 3
Since our given value is in grams, we need to convert it to moles. To do this, we will calculate the molar mass (g/mol) of C₂H₅OH using the molar mass of each element found on the periodic table: 2(12.01g) + 6(1.008g) + 16.00g = 46.07 g/mol
Using this value, we can write a mole ratio to get rid of the unit of grams of ethanol.
Step 4
Uh oh! 😩 We don’t want moles of C₂H₅OH--we want the volume of carbon dioxide produced. Now that we have moles, though, we can go over to CO₂ with a mole ratio. Looking at the balanced chemical equation, for every 1 mole of C₂H₅OH, we can make up to 2 moles of CO₂.
Step 5
Almost there! We have a unit of CO₂, but we want its volume. Since this reaction occurs at STP, we can use the value of the molar volume at STP.
Good work! With 105.2g of ethanol, we could make 102. L of CO₂.
You will see stoichiometry used with the ideal gas law (PV=nRT) and molarity (moles/mass or moles/volume, often denoted as M). Check out the practice problems to see how you’ll see them in problems.
🎥 Watch AP Chemistry teacher Mónica Gracida teach the basic concepts of stoichiometry.
General Steps
Here is a neat list of the general steps for tackling stoichiometry problems:
- Write out the balanced chemical equation, if not given.
- Identify the known measurements given to you.
- If the known measurement is in grams, convert it to moles.
- Multiply by a mole ratio using the coefficients in the chemical equation.
- Repeat steps 3-4 as many times as necessary.
- Cross out your units and circle the final one to make sure you set it up correctly!
Try on your Own!
The following reaction occurs at STP:
How many particles of BrF will be produced with 160.0g of Br2?
Answer
Step 1 includes using the molar mass of Br2 to convert to moles of Br2. Step 2 is the mole ratio of Br2 to BrF using their respective stoichiometric coefficients. Step 3 uses Avogadro's number to convert the number of moles to the number of particles of BrF.
🎥 Watch Serene Fang go over gas stoichiometry, discussing STP, limiting reactants, the ideal gas law, and gas collection over water.
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
atom conservation | The principle that atoms cannot be created, destroyed, or changed during a chemical process, so the total number and type of atoms remain constant. |
balanced chemical equation | A chemical equation where the number of atoms of each element is equal on both the reactant and product sides. |
coefficients | The numbers placed in front of chemical formulas in a balanced equation that indicate the relative proportions of reactants and products involved in the reaction. |
ideal gas law | The equation PV = nRT that relates pressure, volume, number of moles, and temperature of an ideal gas. |
molarity | A measure of solution concentration expressed as the number of moles of solute dissolved per liter of solution. |
mole concept | A fundamental chemistry concept that relates the number of particles (atoms, molecules, or ions) to measurable quantities through Avogadro's number. |
product | Substances formed as a result of a chemical reaction. |
reactant | Substances that are consumed in a chemical reaction to form products. |
stoichiometric calculations | Quantitative calculations using balanced chemical equations and mole ratios to determine amounts of reactants consumed or products formed in a chemical reaction. |
Frequently Asked Questions
What is stoichiometry and why do we need to learn it?
Stoichiometry is the quantitative bookkeeping of chemical reactions—using a balanced equation’s coefficients as mole ratios to relate amounts of reactants and products. Because atoms are conserved (conservation of mass), you can calculate how many moles (or grams, liters, or molarity) of product form from a given reactant amount, or how much reactant is needed for a desired product. Key ideas: mole concept, molar mass, Avogadro’s number, limiting vs. excess reactant, theoretical and percent yield, and using mole ratios with the ideal gas law or solution molarity for gas/solution problems. Stoichiometry is tested in AP Unit 4 (LO 4.5.A) and appears on both multiple-choice and free-response questions that emphasize mathematical routines, so you’ll want fluency converting units and setting up mole-ratio conversion factors. For targeted review, see the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and practice sets (https://library.fiveable.me/practice/ap-chemistry).
How do you balance chemical equations for stoichiometry problems?
Balancing equations is just making sure atoms are conserved so you can use the coefficients as mole ratios in stoichiometry. Quick method: write the unbalanced formulae, count atoms of each element on both sides, and adjust integer coefficients (start with elements that appear once, leave H and O for last). Recount and repeat until all elements match and coefficients are in lowest whole-number ratio. Those coefficients are your stoichiometric coefficients—they give mole ratios used to convert between moles (or mass, volume, molarity) of reactants and products. For problems, convert grams/volume to moles, use the balanced-equation mole ratio, then convert back to desired units; include checks for limiting reactant, theoretical yield, percent yield. This aligns with CED 4.5.A: coefficients give proportional information for calculations. Want more worked examples? See the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and practice problems (https://library.fiveable.me/practice/ap-chemistry).
I'm confused about mole ratios - can someone explain how they work in stoichiometry?
Mole ratios come straight from the balanced equation—the coefficients tell you how many moles of each substance react or form. Treat those coefficients as conversion factors. Quick steps you’ll use on AP stoichiometry problems (Topic 4.5): 1. Balance the equation. Example: 2 H2 + O2 → 2 H2O gives H2:O2: H2O = 2:1:2. 2. Convert your given amount to moles (grams → mol by molar mass; molecules → mol by Avogadro’s number; volume → mol if gas with PV = nRT; or use M × V for solutions). 3. Use the mole ratio (from coefficients) to convert moles of the known substance to moles of the unknown. E.g., 3.00 mol H2 × (1 mol O2 / 2 mol H2) = 1.50 mol O2 needed. 4. Convert moles back to the requested unit (grams, liters, molecules). Mole ratios also tie into limiting reactant, theoretical yield, percent yield, and mix with ideal gas law or molarity in multi-step AP problems (CED 4.5.A.1–4.5.A.3). For more examples and practice, see the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and the AP practice question bank (https://library.fiveable.me/practice/ap-chemistry).
What's the difference between theoretical yield and actual yield in stoichiometry calculations?
Theoretical yield is the maximum amount of product you can make from given reactants using the balanced equation and mole ratios—it assumes the reaction goes perfectly with the limiting reactant fully consumed. Actual (or experimental) yield is what you actually collect in the lab (usually less because of incomplete reactions, side reactions, losses, or measurement error). Percent yield links them: % yield = (actual yield / theoretical yield) × 100%. Example: if theoretical yield is 10.0 g but you isolate 7.5 g, percent yield = (7.5/10.0)×100 = 75%. On the AP, you’ll use the balanced equation, find the limiting reactant, calculate theoretical yield (moles → mass), then compare to experimental mass for percent yield (Topic 4.5 keywords: limiting reactant, theoretical yield, percent yield). For extra practice, check the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and more practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do we always have to start stoichiometry problems by balancing the equation first?
You always balance the equation first because stoichiometry is just bookkeeping of atoms and moles—atoms are conserved. A balanced equation gives stoichiometric coefficients that tell you exact mole ratios (conversion factors) between reactants and products, so you can convert moles of one substance into moles of another. If the equation isn’t balanced you’ll use the wrong ratios and get wrong answers for limiting reactant, theoretical yield, percent yield, or any mole-based calculation. On the AP exam you’ll be expected to use those coefficients for conversions and to justify amount changes (CED 4.5.A, EK 4.5.A.1–3). For practice, review the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and try problems from the Unit 4 page (https://library.fiveable.me/ap-chemistry/unit-4) or the AP practice set (https://library.fiveable.me/practice/ap-chemistry) to get fluent with mole ratios and limiting-reactant setups.
How do I convert from grams to moles to grams in stoichiometry problems?
1) Always start with a balanced chemical equation—the stoichiometric coefficients give the mole ratios (CED 4.5.A.2). 2) Convert grams of your given substance to moles: moles = mass (g) ÷ molar mass (g·mol⁻¹). Use the periodic table to get molar mass. 3) Use the mole ratio from the balanced equation to convert moles of the given to moles of the target: moles_target = moles_given × (coef_target / coef_given). 4) Convert moles of the target to grams: mass_target (g) = moles_target × molar mass_target (g·mol⁻¹). Example template: mass A → nA = massA / M_A → nB = nA × (b/a) → massB = nB × M_B. AP tips: always show the balanced equation, include units and sig figs, and check for limiting reactant/theoretical or percent yield when multiple reactants are present (CED keywords). For a quick review, see the Topic 4.5 stoichiometry guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and practice lots of problems (https://library.fiveable.me/practice/ap-chemistry).
What does it mean when they say "atoms must be conserved" in chemical reactions?
“Atoms must be conserved” means every atom you start with in the reactants still exists in the products—none are created or destroyed. In practice that’s why you must balance chemical equations: the number of each element’s atoms on the left equals the number on the right. Example: 2 H2 + O2 → 2 H2O has 4 H and 2 O on both sides. For AP Chem stoichiometry (CED 4.5.A.1–A.2) balanced equations give stoichiometric coefficients that are mole ratios—those ratios let you calculate product amounts from reactant amounts (and vice versa), find limiting reactants, theoretical yields, and percent yield. This conservation principle underpins mole-conversion factors, ideal-gas and molarity-linked stoichiometry too. Review examples and practice problems in the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and more practice at the unit page (https://library.fiveable.me/ap-chemistry/unit-4) or practice problem bank (https://library.fiveable.me/practice/ap-chemistry).
Can someone walk me through a basic stoichiometry calculation step by step?
Start with a balanced equation and a clear question. Example: how many grams CO2 form from 16.0 g CH4 in the reaction CH4 + 2 O2 → CO2 + 2 H2O? Step 1—convert given mass to moles: mol CH4 = 16.0 g ÷ 16.04 g·mol⁻¹ = 0.998 mol (use molar mass; show units). Step 2—use the mole ratio (stoichiometric coefficients): from the balanced equation 1 mol CH4 → 1 mol CO2, so mol CO2 = 0.998 mol. Step 3—convert moles product to grams: mass CO2 = 0.998 mol × 44.01 g·mol⁻¹ = 43.9 g CO2 (report with correct sig figs). Notes tied to the CED: always start with a balanced equation, use mole ratios from coefficients (4.5.A.2), show unit conversions and the mole concept (4.5.A.1). For exam free-response, write each step, label units, and state limiting reactant/theoretical yield/percent yield when those are asked. Need more practice? Check the AP Chem Stoichiometry study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and try problems at (https://library.fiveable.me/practice/ap-chemistry).
How do you know which reactant is the limiting reagent in stoichiometry problems?
You find the limiting reagent by using the balanced equation and mole ratios. Steps: (1) Balance the reaction. (2) Convert the masses/volumes/concentrations of each reactant to moles. (3) Use the stoichiometric coefficients to compute how many moles of product (or of some other reactant) each reactant could make/consume. (4) The reactant that makes the fewest moles of product (or that runs out first) is the limiting reagent; the other(s) are in excess. Quick tip: you can also divide moles available by the coefficient for each reactant (moles available / stoichiometric coefficient). The smallest result identifies the limiting reagent. That’s exactly the mole-ratio reasoning in CED 4.5.A (use coefficients to relate amounts and calculate product/theoretical yield). Want practice? Work through the stoichiometry study guide on Fiveable (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and try problems from the AP unit page (https://library.fiveable.me/ap-chemistry/unit-4) or the practice set (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how to use coefficients from balanced equations in calculations - help?
Think of coefficients as mole-conversion factors—they tell you the ratio of moles of each substance in a balanced equation. Quick recipe (use for gram → mole → mole ratio → gram, or for gases/solutions with PV=nRT or molarity): 1) Start with given amount (often grams, L gas, or molarity). 2) Convert to moles (grams ÷ molar mass; or for gas use n = PV/RT; or for solution use moles = M × L). 3) Use the balanced equation coefficients as a mole ratio to convert: moles of A × (coefficient of B / coefficient of A) = moles of B. 4) Convert moles of B to the desired units (grams, volume, etc.). Remember to check for limiting reactant if more than one reactant is given (compare moles needed vs available). For AP free-response, show each step, units, and the mole-ratio step clearly—graders expect that (CED 4.5.A.1–4.5.A.3). For extra practice and worked examples, see the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2), the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and practice problems (https://library.fiveable.me/practice/ap-chemistry).
What's the connection between stoichiometry and the ideal gas law?
Stoichiometry and the ideal gas law connect whenever one (or more) reactants or products is a gas. Use PV = nRT to convert measured gas conditions (P, V, T) into moles (n), then treat those moles like any other quantity in stoichiometric calculations—use the balanced equation and mole ratios to find limiting reactants, theoretical yield, or percent yield. For example, measure CO2 volume at STP → compute nCO2 with PV = nRT → use the reaction’s coefficients to find moles (and mass) of reactant consumed or product formed. You can also work backwards: from grams → moles → predict gas volume or partial pressure. AP CED explicitly expects you to combine stoichiometry with the ideal gas law for gas stoichiometry problems (Topic 4.5). Practice applying this flow—PV = nRT ↔ mole ratio ↔ molar mass—in both MC and free-response questions. For a focused review, see the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do molarity calculations relate to stoichiometry problems?
Molarity is just a way to express how many moles of a solute are in a given volume (M = mol/L), so it plugs straight into stoichiometry problems by giving you moles to use with mole ratios from a balanced equation. Typical steps: (1) convert volume of solution to moles using M × L; (2) use stoichiometric coefficients as mole-ratio conversion factors to find moles of product or other reactant; (3) convert those moles to grams, liters (via ideal gas law), or back to concentration as needed. That’s how titrations and solution stoichiometry are done on the exam: use molarity → moles → mole ratio → desired quantity (CED 4.5.A.2–4.5.A.3; keywords: mole ratio, molarity, solution stoichiometry, ideal gas law). For extra practice and worked examples check the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2), the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and lots of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do my stoichiometry answers never match the theoretical yield?
Short answer: your calculated stoichiometric “theoretical yield” is the maximum amount of product predicted by the balanced equation assuming complete reaction and pure reagents—real lab yields almost always fall below that. Common reasons your answers don’t match the theoretical yield: - math/setup errors: equation not balanced, wrong mole ratio, using grams instead of moles, or wrong molar mass (double-check units and sig figs). - limiting reagent mistakes: you must find the limiting reactant before calculating theoretical yield. - state/condition issues: gas or solution problems need PV = nRT or M = n/V conversions. - experimental losses: incomplete reaction, side reactions, impure reagents, product lost during transfer/filtration or drying → these lower actual yield. - reporting errors: percent yield = (actual yield/theoretical yield) × 100%; theoretical must be in the same units as actual. AP exam tip: show balanced equation, all conversions (g ↔ mol), identify limiting reagent, and box your percent yield—the College Board expects the full conversion chain (use CED stoichiometry keywords: mole ratio, limiting reactant, theoretical yield, percent yield). For a step-by-step checklist and practice problems, see the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and try more problems at (https://library.fiveable.me/practice/ap-chemistry).
What happens in stoichiometry when you have excess reactants?
If one reactant is in excess, it means another reactant is limiting—the limiting reactant determines how much product can form (the theoretical yield) because atoms are conserved and coefficients give mole ratios (CED 4.5.A). The excess reactant simply remains after the reaction goes to completion; you can calculate how much is left by: - Convert initial masses/volumes to moles. - Use the balanced equation’s mole ratio to find moles of limiting reactant consumed and moles of excess reactant required. - Subtract required moles from initial moles of the excess to get leftover moles (and convert to g if needed). Example: If equation is A + 2B → C and you start with 1.0 mol A and 3.0 mol B, A is limiting (needs 2.0 mol B but only 3.0 mol B available? actually B excess). Wait—work it each time with mole ratios. Remember to report theoretical yield, then percent yield if you have actual product. For step-by-step practice and AP-aligned examples, see the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2), the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and tons of practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I set up dimensional analysis for complex stoichiometry problems?
Think in chains of canceling units. Start by writing what you’re given and what you need, then build a series of conversion factors that cancel units until you’re left with the answer. Step-by-step: 1. Convert any mass/particles/volume/molarity to moles first (g ÷ molar mass; atoms ÷ Avogadro’s number; V_gas → n with PV = nRT; M × L → moles). 2. Use the balanced equation’s stoichiometric coefficients as a mole ratio to go from moles of one substance to moles of another. Example: grams A → mol A → mol B (× coefficient ratio) → grams B. 3. For solutions or gases combine molarity or ideal-gas conversions in the chain (e.g., L solution → mol solute → mol product → grams product). 4. For multi-reactant problems find the limiting reagent by computing product yield from each reactant. The smallest theoretical yield wins. 5. Finish: compute percent yield = (actual ÷ theoretical)×100, and include units and reasonable sig figs. On the AP exam show each conversion factor (molar mass, mole ratios, PV=nRT, M conversions) so graders can follow your math. For more worked examples and practice, check the Topic 4.5 study guide (https://library.fiveable.me/ap-chemistry/unit-4/stoichiometry/study-guide/GjwCuhOQRvWLb4rKjYD2) and thousands of practice problems (https://library.fiveable.me/practice/ap-chemistry).