Polymer Chain Conformations
A polymer chain in solution doesn't hold a single fixed shape. It constantly fluctuates between many possible spatial arrangements, and the average of these arrangements determines the chain's effective size and how it interacts with its environment. The conformation a polymer adopts depends on the balance between segment-segment interactions and segment-solvent interactions.
Types of Polymer Chain Conformations
Three broad categories describe the conformations polymers adopt:
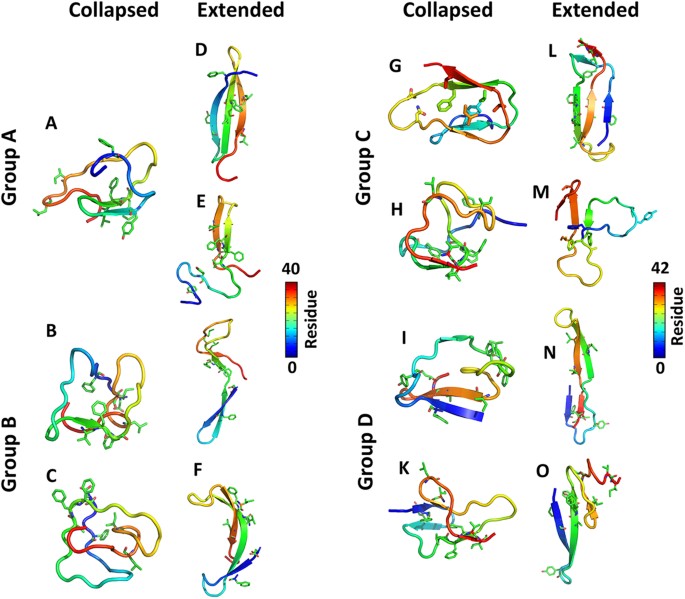
- Random coil conformation: The most common case. The polymer backbone has enough flexibility that thermal energy drives each segment into a roughly random orientation relative to its neighbors. The result is a disordered, coiled structure. Think of cooked spaghetti in water: no preferred direction, just a loose tangle.
- Extended conformation: The chain stretches out when repulsive interactions between segments dominate (as in a highly charged polyelectrolyte) or when an external force is applied (like stretching a rubber band). The chain occupies much more space than a random coil of the same molecular weight.
- Collapsed conformation: The chain folds into a compact, globular structure when segment-segment attractions overpower segment-solvent interactions. This happens in poor solvent conditions, where the polymer minimizes its contact with the surrounding solvent.
Impact of Polymer Chain Conformations on Physical Properties
The conformation a polymer adopts has direct consequences for measurable properties:
- Solubility: Extended conformations maximize polymer-solvent contact, increasing solubility. Collapsed conformations minimize that contact, reducing solubility and often leading to phase separation.
- Viscosity: Random coils in solution create significant chain entanglements, raising the viscosity. Collapsed globules occupy less hydrodynamic volume and entangle less, lowering viscosity. (Note: highly extended chains in dilute solution can also raise viscosity because of their large hydrodynamic volume.)
- Mechanical strength: In bulk materials, chains that can align and extend (as in drawn fibers) produce higher tensile strength. Disordered random coil arrangements yield more flexible but weaker materials.
Radius of Gyration and Polymer Size
Definition and Calculation of Radius of Gyration
The radius of gyration () quantifies how far, on average, the segments of a polymer chain are from its center of mass. It gives you a single number that captures the overall spatial extent of the chain.
where is the number of segments, is the position of the -th segment, and is the position of the center of mass.
For an ideal (freely jointed) chain with segments of length , this simplifies to:
This scaling is the baseline for an unperturbed chain. Deviations from this scaling tell you about solvent quality and excluded volume effects (more on that below).
Relationship between Radius of Gyration and Polymer Size
- A larger means the chain is more spread out (expanded or extended conformation).
- A smaller means the chain is more compact (collapsed conformation).
More generally, scales with chain length as , where the Flory exponent depends on solvent conditions:
| Condition | Conformation | |
|---|---|---|
| Good solvent | (0.588) | Swollen coil |
| Theta solvent | Unperturbed random coil | |
| Poor solvent | Collapsed globule |
can be measured experimentally using small-angle X-ray scattering (SAXS), small-angle neutron scattering (SANS), or static light scattering (SLS). These techniques probe the spatial correlations within the chain and extract from the low-angle scattering regime (Guinier analysis). Dynamic light scattering (DLS) measures the hydrodynamic radius , which is related to but distinct from .
Factors Influencing Chain Dimensions
Chemical Structure and Flexibility of the Polymer Backbone
The chemical makeup of the repeat unit controls how easily the backbone can rotate and bend:
- Flexible backbones (e.g., polyethylene, with simple single bonds) allow many accessible rotational states per bond. These chains adopt compact random coils with relatively small for their molecular weight.
- Rigid backbones (e.g., Kevlar/poly(p-phenylene terephthalamide), with aromatic rings and amide linkages restricting rotation) resist bending. These chains have larger values and tend toward rod-like or extended conformations.
Backbone stiffness is often quantified by the persistence length (): the distance along the chain over which the chain "remembers" its initial direction. Flexible polymers have short persistence lengths (a few bond lengths); rigid polymers have long ones (tens of nanometers or more).
Degree of Polymerization and Chain Length
The degree of polymerization () is the number of repeat units in the chain. Since , longer chains are always larger, but the rate of growth depends on :
- In a good solvent, doubling increases by a factor of .
- In a theta solvent, doubling increases by .
Very short chains (oligomers, typically ) don't follow the same statistical scaling because there aren't enough segments for random-walk statistics to apply. Their properties can differ significantly from high-molecular-weight polymers of the same chemistry.
Interactions between Polymer Segments
Intramolecular interactions between segments shift the chain away from ideal random-coil behavior:
- Van der Waals forces: Weak, short-range attractions between all segments. These favor slightly more compact conformations and become collectively important in poor solvents.
- Hydrogen bonding: Directional and stronger than van der Waals forces. In polymers like nylon, intramolecular (and intermolecular) hydrogen bonds can stabilize specific local conformations and influence overall chain dimensions.
- Electrostatic interactions: Charged segments along the backbone (as in polyelectrolytes like polyacrylic acid) repel each other, driving chain expansion. The magnitude of this effect depends on ionic strength: adding salt screens the charges and allows the chain to contract.
Solvent Quality and Conformations
Good, Poor, and Theta Solvents
Solvent quality describes the net balance between polymer-solvent and polymer-polymer interactions:
- Good solvent: Polymer-solvent contacts are energetically favorable. The chain swells beyond its unperturbed dimensions because segments "prefer" to be surrounded by solvent rather than by other segments. Example: polystyrene in toluene.
- Poor solvent: Polymer-polymer contacts are preferred. The chain collapses to minimize solvent exposure. Example: polystyrene in methanol.
- Theta () solvent: The attractive and repulsive contributions to the excluded volume exactly cancel. The chain behaves as an ideal random coil with . Example: polystyrene in cyclohexane at 34.5°C (the theta temperature).
The theta condition is especially useful because it lets you measure unperturbed chain dimensions, which reflect only the intrinsic geometry of the backbone without solvent-mediated expansion or contraction.
Flory-Huggins Theory and Solvent Quality
The Flory-Huggins theory provides the thermodynamic framework connecting solvent quality to polymer conformation. It models the free energy of mixing as:
where and are the number of solvent and polymer molecules, and are their volume fractions, and is the Flory-Huggins interaction parameter.
The value of determines solvent quality:
| value | Solvent quality | Conformation |
|---|---|---|
| Good | Swollen coil | |
| Theta | Unperturbed coil | |
| Poor | Collapsed globule |
The first two terms in the free energy expression represent the entropy of mixing (always favoring dissolution), while the -dependent term captures the enthalpy of mixing (which can favor or oppose it).
Experimental Manipulation of Solvent Quality
You can tune solvent quality in the lab through several approaches:
- Temperature: For most polymer-solvent systems, raising the temperature improves solvent quality by increasing the entropic driving force for mixing. Some systems show lower critical solution temperature (LCST) behavior, where heating worsens solvent quality (e.g., poly(N-isopropylacrylamide) in water above ~32°C). Others show upper critical solution temperature (UCST) behavior, where heating improves it.
- Solvent composition: Mixing a good solvent with a poor solvent (co-solvent approach) lets you continuously tune and therefore the chain dimensions. This is commonly used in polymer precipitation and purification.
- Pressure: Less commonly used, but pressure can shift the balance of intermolecular interactions. This is most relevant for polymer solutions near critical conditions or in supercritical fluid solvents like .