Statistical mechanics bridges the gap between microscopic particle behavior and macroscopic thermodynamic properties. It uses probability theory to explain how countless individual particles collectively produce observable phenomena like temperature, pressure, and entropy. In the history of science, this represented a major conceptual shift: thermodynamics no longer had to rely solely on bulk measurements, because the behavior of heat and energy could now be derived from the statistics of atoms and molecules.
Entropy, already a key concept in classical thermodynamics, gained a fundamentally new meaning through statistical mechanics. Rather than just a bookkeeping quantity in heat engines, entropy became interpretable as a measure of how many microscopic arrangements correspond to a given macroscopic state. This reframing, largely due to Ludwig Boltzmann in the 1870s, helped explain why entropy tends to increase and gave the second law of thermodynamics a probabilistic foundation.
Statistical Mechanics Principles
Fundamentals and Thermodynamic Connections
Statistical mechanics uses probability theory to study systems composed of enormous numbers of particles, connecting their microscopic properties to macroscopic thermodynamic quantities like temperature and pressure. The key move is treating individual particle behavior as unknowable in detail but predictable in aggregate.
The fundamental postulate of statistical mechanics states that all accessible microstates of an isolated system in equilibrium are equally probable. A microstate is one specific arrangement of all the particles' positions and momenta. This postulate forms the basis for deriving thermodynamic properties from microscopic behavior: if every microstate is equally likely, then the macroscopic state you observe is simply the one consistent with the most microstates.
Key Concepts and Laws
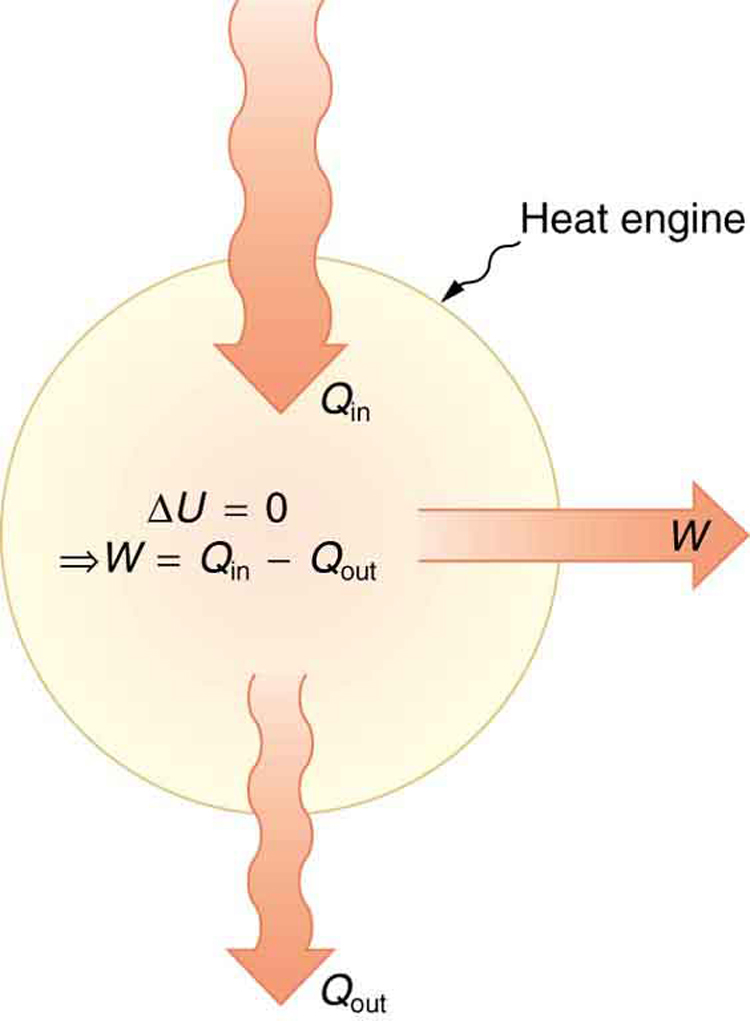
The partition function is a central mathematical tool in statistical mechanics. It sums over all possible states of a system, with each state weighted by its Boltzmann factor (, where is the state's energy, is Boltzmann's constant, and is temperature). From the partition function, you can calculate thermodynamic quantities such as energy, entropy, and free energy.
All four laws of thermodynamics (zeroth through third) can be derived from the principles of statistical mechanics. This was historically significant because it showed that thermodynamics wasn't a separate branch of physics but rather a consequence of the mechanical behavior of particles governed by probability. James Clerk Maxwell, Ludwig Boltzmann, and Josiah Willard Gibbs were the central figures in establishing this connection during the second half of the 19th century. Maxwell developed the velocity distribution for gas molecules in the 1860s, Boltzmann formulated the statistical interpretation of entropy in the 1870s, and Gibbs provided the most rigorous and general mathematical framework in his 1902 treatise Elementary Principles in Statistical Mechanics.
Entropy: A Statistical Perspective

Entropy as a Measure of Multiplicity
From a statistical viewpoint, entropy measures the number of microscopic configurations (microstates) that correspond to a system's macroscopic state. A state with many possible microstates has high entropy; a state with few has low entropy. You'll often hear entropy described as "disorder," but the more precise term is multiplicity: the count of microstates compatible with the observed macroscopic conditions.
The Boltzmann entropy equation makes this quantitative:
Here, is entropy, is Boltzmann's constant ( J/K), and is the number of microstates. The logarithm is doing important work here: it makes entropy additive. If you combine two independent systems, their total entropy is the sum of their individual entropies, even though the total number of microstates is the product (since ). This equation is carved on Boltzmann's tombstone in Vienna, reflecting its importance to the history of physics.
Spontaneous Processes and the Second Law
In any spontaneous process, the total entropy of the universe (system plus surroundings) increases. The statistical interpretation explains why: the system naturally evolves toward the macroscopic state consistent with the greatest number of microstates, simply because that state is overwhelmingly more probable.
This explains everyday irreversible processes:
- Heat flows from hot to cold, not the reverse, because the combined system has far more microstates when energy is spread out evenly.
- Gases mix when a partition is removed because the mixed state has astronomically more microstates than the separated state.
The reverse of these processes isn't forbidden by any fundamental law of particle mechanics. It's just so improbable as to be effectively impossible for any macroscopic system.
Entropy also provides insight into the arrow of time. The laws of mechanics are time-reversible at the particle level, yet macroscopic processes clearly have a preferred direction. The statistical interpretation resolves this: the direction of increasing entropy defines the forward direction of time. This was a genuinely puzzling issue in the late 19th century and generated significant debate, including the famous Loschmidt paradox (1876): if the microscopic equations of motion are time-reversible, how can entropy always increase? Boltzmann's resolution relied on probabilistic reasoning rather than strict mechanical necessity, arguing that while individual reversed trajectories are possible, they are fantastically unlikely for systems with many particles.
Microscopic Behavior and Macroscopic Properties
Particle Distributions and Thermodynamic Variables
Different statistical distributions describe how particles distribute themselves across energy states, depending on the type of particle:
- The Maxwell-Boltzmann distribution describes the probability distribution of particle speeds in a classical ideal gas at thermal equilibrium. From it, you can calculate macroscopic properties like average speed, root-mean-square speed, and most probable speed. This distribution applies well to ordinary gases at everyday temperatures and was the earliest of the three, developed by Maxwell in 1860 and extended by Boltzmann.
- The Fermi-Dirac distribution applies to fermions (particles with half-integer spin, like electrons and protons). Fermions obey the Pauli exclusion principle, so no two can occupy the same quantum state. This is crucial for understanding electrons in metals, semiconductor behavior, and the structure of white dwarf stars, where electron degeneracy pressure prevents gravitational collapse.
- The Bose-Einstein distribution applies to bosons (particles with integer spin, like photons). Bosons can pile into the same quantum state, which explains phenomena like the spectrum of blackbody radiation and, at very low temperatures, Bose-Einstein condensation, where a macroscopic fraction of particles occupies the lowest energy state.
The distinction between Fermi-Dirac and Bose-Einstein statistics became clear only with the development of quantum mechanics in the 1920s, representing a major refinement of Boltzmann and Gibbs's original classical framework.
The equipartition theorem states that, in thermal equilibrium, each quadratic degree of freedom in the system's energy contributes an average energy of . For example, a monatomic ideal gas has three translational degrees of freedom, giving an average kinetic energy per particle of . This theorem allows straightforward calculation of heat capacities. However, it famously fails at low temperatures: the measured heat capacities of diatomic gases drop below the values equipartition predicts, because rotational and vibrational modes "freeze out." This discrepancy was one of the early clues that classical physics was incomplete and helped motivate the development of quantum theory.
Linking Microscopic and Macroscopic Behavior
Statistical mechanics can derive the ideal gas law, , from first principles by considering how gas particles bounce off container walls. Pressure emerges from the average momentum transfer per collision, and temperature corresponds to average kinetic energy. This derivation demonstrated concretely that macroscopic gas laws are consequences of microscopic particle mechanics.
Fluctuations in thermodynamic properties (like small variations in energy or particle number) can also be analyzed statistically. For large systems, these fluctuations are tiny relative to the average values, which is why macroscopic thermodynamics works so reliably. But for very small systems, fluctuations become significant. Einstein's 1905 analysis of Brownian motion used exactly this kind of statistical fluctuation argument to provide strong evidence for the reality of atoms. By showing that the visible, jittery motion of pollen grains in water could be quantitatively explained by molecular collisions, Einstein turned the atomic hypothesis from a useful theoretical tool into something experimentally testable. Jean Perrin's subsequent experiments confirmed Einstein's predictions and earned Perrin the 1926 Nobel Prize.
Entropy, Probability, and the Arrow of Time
The Second Law and the Arrow of Time
The second law of thermodynamics states that the entropy of an isolated system never decreases over time. This establishes a clear directionality, known as the thermodynamic arrow of time, which aligns with our everyday experience that processes like breaking an egg or mixing cream into coffee don't spontaneously reverse.
The statistical interpretation gives this a probabilistic rather than absolute explanation: as a system evolves, it moves toward macroscopic states that correspond to vastly more microstates. The system doesn't "know" to increase entropy; it simply has an overwhelming probability of ending up in a high-entropy state because there are so many more ways to be disordered than ordered.
Entropy, Probability, and Irreversibility
The arrow of time can be understood as the system moving from less probable (lower multiplicity) states to more probable (higher multiplicity) states. Consider a simple example: if you have 100 gas molecules in a box, the chance of all 100 spontaneously clustering in one half is roughly in (about in ). For Avogadro's number of molecules (around ), the probability is so vanishingly small that you'd have to wait far longer than the age of the universe to see it happen.
The Boltzmann equation, , captures this connection precisely: states with more microstates (higher ) have higher entropy, and the system is overwhelmingly likely to be found in such states.
The irreversibility of processes like gas mixing or heat dissipation isn't absolute in principle. A reversed process (gas spontaneously unmixing, heat flowing from cold to hot without external work) doesn't violate any microscopic law of physics. It's just so extraordinarily unlikely for macroscopic systems that it never happens in practice. This distinction between "impossible in principle" and "impossible in practice" was one of the genuinely new ideas that statistical mechanics brought to physics.
On a cosmological scale, the arrow of time implies that the universe began in a state of remarkably low entropy. The progression from that ordered initial state toward higher entropy is consistent with cosmological observations of an expanding, cooling universe. Why the early universe had such low entropy remains an open question at the intersection of thermodynamics, cosmology, and fundamental physics. The physicist Roger Penrose has called this the "past hypothesis" and argued it requires a deeper explanation than current physics provides.