This unit is all about learning and identifying different chemical reactions. One of the learning objectives of unit four in the AP Chemistry course is to "identify a reaction as acid-base, oxidation-reduction, or precipitation."
Types of Reactions
Throughout this unit's study guides, you've gotten a taste of net ionic equations and precipitation reactions, as well as titrations and acid-base reactions. Oxidation-reduction reactions are going to be reviewed in-depth in the rest of this unit. For now, here is a quick rundown of these three types of reactions:
-
Acid-base reactions are chemical reactions that involve the transfer of a proton from one molecule to another. They often involve the transfer of a proton from a strong acid to a strong base, resulting in the formation of a salt and water. For more about acid-base reactions, check out the next study guide.
-
Oxidation-reduction reactions, also known as redox reactions, are chemical reactions in which the atoms of one or more elements are oxidized (lose electrons) and reduced (gain electrons). In these reactions, electrons are transferred from the species that is oxidized to the species that is reduced. The species that loses electrons is oxidized, while the species that gains electrons is reduced.
While you may encounter the terms "reducing agent" (the species that is oxidized) and "oxidizing agent" (the species that is reduced), understanding these specific terms will not be assessed on the AP exam.
To track electron transfer in redox reactions, we assign oxidation numbers to atoms:
- Elements in their elemental form have oxidation number 0
- Oxygen is usually -2 (except in peroxides where it's -1)
- Hydrogen is usually +1 (except in metal hydrides where it's -1)
- The sum of oxidation numbers equals the charge on the species
- Combustion reactions are a type of redox reaction and you've already learned about them! The specifics and must-know information about redox reactions will be covered later in this unit.
-
Precipitation reactions are chemical reactions in which two or more soluble reactants combine to form an insoluble product, which is known as a precipitate. We'll focus on precipitation reactions in this study guide!
Precipitation Reactions Explained
When ions in aqueous solutions react, they may produce an insoluble (undissolvable) or barely soluble solid ionic compound. This solid product is called a precipitate.

Solubility Rules
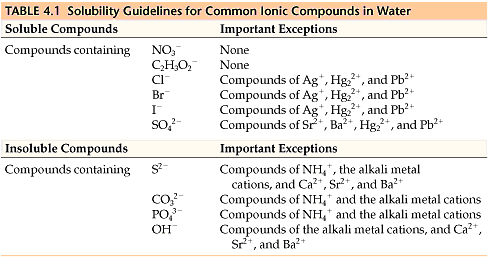
All sodium, potassium, ammonium, and nitrate salts are soluble in water, so they aren't precipitates. You don't need to know any other solubility rules for the AP, but it doesn't hurt to be familiar with common soluble and insoluble compounds. Table 4.1 is a table of solubility for common ions in water. Usually, the question will tell you if the compound is soluble and which solution it's soluble in.
Net Ionic Equations
We went over this in key topic 4.2, but let's do a quick overview!
The best steps to follow when writing a net ionic equation are:
- Figure out which compounds are soluble and insoluble using solubility rules.
- Balance the chemical equation. It may already be balanced, but it also may not, so you always have to check.
- Write the complete ionic equation by dissociating soluble compounds into ions.
- Omit the spectator ions and write the final net ionic equation of the given reaction. Make sure you include the phase of matter each compound is in.
Concentration of Ions
Knowing how to write the net ionic equation for a precipitation reaction is just the first step! Let's take a look at a concentration of ions question, where you calculate how much of each ion is present after a precipitation reaction.
Question: 20.0 mL of [0.100] NaCl (aq) reacts with 30.0 mL of [0.0400] Pb(C₂H₃O₂)₂ (aq).
Part a: What is the mass of the solid formed?
Part b: What are the concentrations of ions at the end of the reaction?
Step #1
Since they didn't give us the equation, let's write it ourselves!
NaCl + Pb(C₂H₃O₂)₂ → NaC₂H₃O₂ + PbCl₂
Always check the equation is balanced! This one isn't, so let's balance it out and make sure each ion is in equal amounts on both sides. 2NaCl + Pb(C₂H₃O₂)₂ → 2NaC₂H₃O₂ + PbCl₂
Step #2
With precipitation reactions and concentration of ions questions, there will always be an insoluble product. In this case, it is either NaC₂H₃O₂ or PbCl₂. Since sodium is always soluble, PbCl₂ is the precipitate in this question.
2NaCl (aq) + Pb(C₂H₃O₂)₂ (aq) → 2NaC₂H₃O₂ (aq) + PbCl₂ (s)
Step #3
Now that we have the equation and know the precipitate, let's get into the math itself. The question provided us with the volumes and molarities for each reactant. Using these two pieces of information, we can find the number of moles of NaCl and Pb(C₂H₃O₂)₂.
Molarity = moles / volume in L - We have to convert the volumes we have into L by dividing by 1000.
0.100 = x moles of NaCl / 0.020 L → x = 0.00200 moles of NaCl (aq)
0.0400 = x moles of Pb(C₂H₃O₂)₂ / 0.030 L → x = 0.00120 moles of Pb(C₂H₃O₂)₂ (aq)
Step #4
Using the number of moles we solved for above, we can now use stoichiometry to answer part a.
But wait! Which number do we do stoich with: 0.00200 or 0.00120?
This is where the limiting reactant (LR) comes into play. The limiting reactant in a reaction is the substance that limits the amount of products produced. Basically, there are different amounts of each reactant. One reactant is more abundant, right?
The reactant that there is less of eventually stops the reaction and limits it since the reactant runs out. The other reactant is called the excess since there is still some of it left over, unreacted.
To find the LR, we have to do stoichiometry with both amounts. Convert each reactant into the precipitate:
Since there are less moles of PbCl₂ using NaCl as a reactant, NaCl is the LR. Pb(C₂H₃O₂)₂ is the excess.
Now we know how many moles of NaCl, Pb(C₂H₃O₂)₂, and PbCl₂ we have, wecan answer part a for real now!
0.00100 mol PbCl₂ x 278.2 g/mol = 0.278 g of PbCl₂
Step #5
Yay, we did half the problem! Let's move on to solving for the concentrations of ions. In order for us to do this, we have to know the moles of each ion and the volumes of each ion.
Let's think this through conceptually a bit. After PbCl₂ (s) forms, what is left in the solution?
Looking back at the LR, either Na⁺ or Cl⁻ will have a final concentration of 0 since one of them will be completely used up. Since Cl⁻ is in the precipitate, Cl⁻ has a final concentration of 0. All of the chloride anions in the solution have been used up to form as much precipitate as possible.
That was easy! 1/4 of part b is complete. 😊
The ion that is in the LR and precipitate ALWAYS has a final concentration of 0. Think of it as being 100% used up, so there is none of it left.
Step #6
In this next step, we can solve for the concentrations of two ions: Na⁺ and C₂H₃O₂. These are considered spectator ions since they aren't in the precipitate. To find their concentrations, we have to use both the 0.00100 mol of PbCl₂ from using NaCl and the 0.00120 mol of PCl₂ from using Pb(C₂H₃O₂)₂.
The first number can help us find Na⁺ whereas the second can help us find C₂H₃O₂⁻.
Na+: All you have to do now is find the volume, but we have to multiply the number of moles by 2 since NaCl has an initial coefficient of 2. This is where balancing the reaction comes in! To find the volume, we just have to add 20.0 mL and 30.0 mL and convert to liters.
(0.00100)(2) / 0.050 L = 0.0400 M of Na⁺
C₂H₃O₂⁻: We have to multiply by 2 here as well since there was a subscript on the reactant side of the equation.
(0.00120)(2) / 0.050 L = 0.0480 M of C₂H₃O₂⁻
Step #7
We have one last ion we have to calculate the concentration of: Pb⁺². This is slightly harder to find, but with some practice, you got this! 😌
To find the excess amount of lead, convert the LR to the soluble product. Here, we would convert 0.00200 moles of NaCl to find the moles reacted. Since there is a 1:2 mole ratio, 0.00100 moles reacted. Then, we would subtract by the excess number of moles (found in step 4), which is 0.00120.
0.00120 - 0.00100 = 0.00020 moles of Pb⁺² unreacted. Then we just divide by the volume in liters, so 0.00020 moles / 0.050 L = 0.0040 M of Pb⁺².
Final Answers
Part a: 0.278 g of PbCl₂
Part b: [Cl⁻] = 0
[Na⁺] = 0.0400
[C₂H₃O₂⁻] = 0.0480
[Pb⁺²] = 0.0040
This is a very difficult question but once you practice and understand it conceptually, you'll begin to be able to get through it faster. It is honestly a lot in one question and probably won't be tested like this. However, knowing it will strengthen your overall stoichiometry skills, so it doesn't hurt! 🙃
Vocabulary
The following words are mentioned explicitly in the College Board Course and Exam Description for this topic.Term | Definition |
|---|---|
acid-base reaction | A chemical reaction involving the transfer of one or more protons (H⁺ ions) between chemical species. |
aqueous solution | A solution in which water is the solvent. |
combustion | A type of oxidation-reduction reaction in which a species reacts with oxygen gas to produce products such as carbon dioxide and water. |
electron transfer | The movement of one or more electrons from one chemical species to another in a redox reaction. |
hydrocarbons | Organic compounds composed of carbon and hydrogen atoms that undergo complete combustion to produce carbon dioxide and water. |
insoluble | Unable to dissolve in a solvent, such as water; describes compounds that form as precipitates. |
ionic compound | Compounds formed by the electrostatic attraction between positively charged cations and negatively charged anions. |
oxidation numbers | Numbers assigned to atoms in reactants and products to track electron transfer and identify oxidized and reduced species in a redox reaction. |
oxidation-reduction reaction | A chemical reaction involving the transfer of one or more electrons between chemical species, indicated by changes in oxidation numbers. |
oxidized | The process in which a species loses electrons in a redox reaction. |
precipitation reaction | A chemical reaction in which ions in aqueous solution combine to produce an insoluble or sparingly soluble ionic compound. |
proton transfer | The movement of a proton (H⁺) from one species to another in an acid-base reaction. |
reduced | The process in which a species gains electrons in a redox reaction. |
sparingly soluble | Slightly soluble in a solvent; describes ionic compounds that form precipitates when ions combine in aqueous solution. |
Frequently Asked Questions
What's the difference between acid-base, redox, and precipitation reactions?
Acid-base: a proton (H+) is transferred between species—think Bronsted-Lowry. Look for H+ moving from an acid to a base (e.g., HCl + NH3 → NH4+ + Cl−). Redox: electrons are transferred; identify by changes in oxidation numbers. If an atom’s oxidation number increases it’s oxidized (lost e−); if it decreases it’s reduced (gained e−). Combustion (hydrocarbons → CO2 + H2O) is a redox subclass. Precipitation: two aqueous ionic solutions mix and form an insoluble (or sparingly soluble) solid—a precipitate. Write the net ionic equation to remove spectator ions; remember Na+, K+, NH4+, and NO3− salts are always soluble (so won’t precipitate). On the AP exam you should identify these by proton transfer (acid-base), oxidation-number changes (redox), or formation of an insoluble solid/net ionic equation (precipitation). For practice and a quick Topic 4.7 review, see Fiveable’s study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and extra practice problems (https://library.fiveable.me/practice/ap-chemistry).
How do I know if a reaction is transferring protons or electrons?
Short answer: look for H+ moving vs. oxidation-number changes. Acid–base (Bronsted–Lowry) = proton transfer: if a species donates H+ to another (HCl + H2O → H3O+ + Cl− or net ionic H+ + OH− → H2O), it’s an acid–base reaction. Redox = electron transfer: assign oxidation numbers to atoms before and after—if any change, electrons moved (Zn + Cu2+ → Zn2+ + Cu is redox: Zn goes 0 → +2, Cu goes +2 → 0). Precipitation reactions you’ll spot by formation of an insoluble solid from ions (use solubility rules in the CED). For AP exam work, the CED explicitly says identify acid–base vs. redox vs. precipitation (4.7.A); use net ionic equations to reveal H+ transfer or oxidation-number changes. Want more practice? Check the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and the AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why do we need to assign oxidation numbers to atoms in chemical reactions?
You assign oxidation numbers because they’re a bookkeeping tool that tells you whether electrons are lost or gained in a reaction—which is exactly what defines a redox reaction in the AP CED (4.7.A.2–4.7.A.4). By comparing oxidation numbers of each atom before and after, you can (1) spot which species is oxidized (its oxidation number increases) and which is reduced (its oxidation number decreases), (2) write balanced half-reactions and balance electrons, and (3) identify combustion reactions (a subclass of redox) and predict products. Oxidation numbers make electron transfer explicit even when electrons aren’t shown in the equation, so they’re essential for identifying redox vs. acid–base or precipitation reactions (learning objective 4.7.A). For practice identifying and balancing redox problems, see the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and try problems at https://library.fiveable.me/practice/ap-chemistry. Note: the CED says “reducing/oxidizing agent” terms won’t be assessed, but knowing oxidation-number changes is.
I'm confused about how to tell which species gets oxidized and which gets reduced in a redox reaction.
Quick rule: oxidation = loss of electrons (oxidation number increases); reduction = gain of electrons (oxidation number decreases). To tell which species does which, assign oxidation numbers to each atom in reactants and products (CED 4.7.A.4–4.7.A.3). Whichever atom’s oxidation number goes up was oxidized (it lost e–); whichever goes down was reduced (it gained e–). Fast method: 1) Write balanced equation (or half-reactions). 2) Assign oxidation numbers to the atoms of interest. 3) Compare reactant vs product values: increase = oxidation, decrease = reduction. Example: Zn + Cu2+ → Zn2+ + Cu: Zn: 0 → +2 (oxidized); Cu: +2 → 0 (reduced). If you prefer, split into half-reactions to see electron transfer explicitly. For AP exams, this oxidation-number method is what they expect for identifying redox (Topic 4.7; see the CED). For extra practice, check the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and try practice problems (https://library.fiveable.me/practice/ap-chemistry).
What happens during a precipitation reaction and how do I predict if a precipitate will form?
A precipitation reaction happens when two aqueous ionic solutions mix and an insoluble (or very sparingly soluble) ionic solid forms and falls out of solution. To predict a precipitate, write the possible product ions, then use solubility rules from the CED: all Na+, K+, NH4+, and NO3− salts are soluble, so they never precipitate (4.7.A.5). If a product is insoluble (like AgCl, PbSO4, BaSO4, etc.), a precipitate forms. Practically: (1) write the molecular equation, (2) break strong electrolytes into ions to make the complete ionic equation, (3) cancel spectator ions to get the net ionic equation showing the solid (s). If the net ionic equation includes a solid (e.g., Ag+(aq) + Cl−(aq) → AgCl(s)), a precipitate forms. Net ionic work and identifying spectator ions are tested on the AP (4.7.A). Need more practice? Check the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and plenty of practice problems (https://library.fiveable.me/practice/ap-chemistry).
Can someone explain combustion reactions in simple terms - are they always redox?
Short answer: combustion reactions are chemical reactions where a substance reacts with O2 (usually making CO2 and H2O for hydrocarbons). Per the AP CED, combustion is an important subclass of oxidation–reduction reactions (4.7.A.2), so yes—combustion reactions are redox. Why: in combustion you change oxidation numbers. Example: in complete combustion of methane, CH4 + 2 O2 → CO2 + 2 H2O, C goes from −4 to +4 (oxidized, loses electrons) and O (in O2, 0) goes to −2 (reduced, gains electrons). Assigning oxidation numbers to atoms before and after the reaction is the AP-approved way to spot the oxidized and reduced species (4.7.A.4). For AP review, see the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4). For lots of practice problems, try the Fiveable practice page (https://library.fiveable.me/practice/ap-chemistry).
How do I remember which salts are always soluble in water?
Short rule to memorize: all salts containing Na+, K+, NH4+ (ammonium), and NO3− (nitrate) are always soluble in water. A quick mnemonic: “No Kittens Need Ammonia” → Nitrate, Potassium, Sodium, Ammonium (or use “NKSN” if you prefer). Tips to make it stick: - Practice by writing a few ionic equations and crossing out spectator ions—you’ll see Na+, K+, NH4+, NO3− never precipitate. - Remember the AP CED: only those families are required to be memorized for the exam (you won’t be tested on a full solubility table)—so focus your effort there. - Use net ionic problems: when you mix two aqueous salts, if one product would contain one of these ions, it stays soluble (a spectator). For extra practice, check the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and the AP Chem practice bank (https://library.fiveable.me/practice/ap-chemistry).
What's the actual difference between oxidation and reduction?
Oxidation and reduction are just electron bookkeeping. Oxidation = loss of electrons (oxidation number increases); reduction = gain of electrons (oxidation number decreases). In any redox reaction electrons flow from the species that’s oxidized (it loses electrons) to the species that’s reduced (it gains electrons). A reliable AP method to ID them is to assign oxidation numbers to atoms in reactants and products (CED 4.7.A.2–4.7.A.4). If an atom’s oxidation number goes up, it was oxidized; if it goes down, it was reduced. Remember combustion is a common redox type (reacting with O2). Note: the CED says the specific meanings of “reducing agent” and “oxidizing agent” won’t be tested, but knowing electron transfer and oxidation-number changes is. For more practice and examples, check the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz), the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4), and AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
Why does mixing two clear solutions sometimes make a solid precipitate?
When two clear aqueous solutions mix, their dissolved ions can pair up to form an ionic compound that’s insoluble (or sparingly soluble) in water—that solid is the precipitate. For example, AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq). The net ionic equation is Ag+(aq) + Cl−(aq) → AgCl(s); Na+ and NO3− are spectator ions. Precipitation depends on solubility: most Na+, K+, NH4+, and NO3− salts stay dissolved (CED 4.7.A.5), so precipitates form only when the new ion pair violates solubility expectations. On the AP Exam you should be able to identify precipitation reactions, write complete and net ionic equations, and point out spectator ions (CED Topic 4.7). For worked examples and guided practice, see the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and try extra problems at (https://library.fiveable.me/practice/ap-chemistry).
I don't understand how proton transfer works in acid-base reactions - can you give me an example?
Think of a Bronsted–Lowry acid as a proton (H+) donor and a base as a proton acceptor. Example (net ionic): HCl(aq) + NH3(aq) → NH4+(aq) + Cl−(aq). Here HCl donates H+ and NH3 accepts it—that proton transfer makes this an acid-base reaction (CED 4.7.A.1). You can also write HCl + H2O → H3O+ + Cl− to show water acting as the base. A quick way to spot these on the AP: look for H+ moving from one species to another (not electron transfer—that’s redox). On free-response or multiple choice you might be asked for the net ionic equation or to identify the acid/base by proton transfer (keep that CED language in mind). For more examples and practice, see the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and the AP Chem practice set (https://library.fiveable.me/practice/ap-chemistry).
How do I assign oxidation numbers to atoms step by step?
Step-by-step method to assign oxidation numbers (useful for spotting redox changes on the AP exam): 1. Know the common rules: - Element in pure form = 0 (O2, H2, Fe). - Monoatomic ion = its charge (Na+ = +1, Cl- = -1). - H is usually +1 (except in hydrides like NaH where H = -1). - O is usually -2 (except in peroxides where O = -1). - Group 1 metals = +1, group 2 = +2. 2. Write the formula and count atoms. 3. Assign oxidation numbers for atoms with fixed rules (H, O, alkali/alkaline earth). 4. Let unknowns be x and use the rule: sum of oxidation numbers = overall charge (0 for neutral compounds, ion charge for polyatomic ions). 5. Solve for x, then check by summing to the total charge. Quick example: Fe2O3. O = -2 × 3 = -6. Sum = 0, so 2·(Fe) + (-6) = 0 → 2Fe = +6 → Fe = +3. This is exactly how you identify oxidized vs reduced species (look for increase/decrease in oxidation numbers). For extra practice and AP-aligned problems, see the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and 1000+ practice questions (https://library.fiveable.me/practice/ap-chemistry).
What makes sodium, potassium, ammonium, and nitrate salts special for solubility?
Those ions are “always soluble” because their ionic interactions with water are strong enough that the lattice energy of any salt they form is overcome by hydration (ion–dipole) energy. Na+, K+, and NH4+ are small, singly charged cations and NO3– is a delocalized anion—together they dissolve easily, so salts containing them stay aqueous. For AP Chem this matters because they’re usually spectator ions in precipitation (net ionic) problems: if a salt contains one of these ions, it won’t be the solid that precipitates. The CED explicitly lists “All sodium, potassium, ammonium, and nitrate salts are soluble in water” (useful for identifying precipitation in Topic 4.7)—see the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz). For more practice on net ionic and precipitation questions, check the Unit 4 overview (https://library.fiveable.me/ap-chemistry/unit-4) and AP practice problems (https://library.fiveable.me/practice/ap-chemistry).
Is every reaction with oxygen gas automatically a combustion reaction?
No—a reaction involving O2 isn’t automatically a “combustion” reaction. On the AP CED, combustion is a specific subclass of oxidation–reduction reactions in which a species reacts with O2 (and for hydrocarbons, complete combustion gives CO2 + H2O). Key checks: does O2 act as the oxidizing agent (oxidation numbers change)? Is the process rapid and exothermic (flame or heat released)? If yes, it’s combustion. If O2 simply forms an oxide slowly (corrosion/rusting) or participates without the characteristic heat/combustion products, we’d call it a redox or synthesis/oxidation reaction instead. For more examples and AP-style practice, review Topic 4.7 (Types of Chemical Reactions) study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and try related practice problems (https://library.fiveable.me/practice/ap-chemistry).
How can I tell if electrons are being transferred just by looking at a chemical equation?
Short answer: assign oxidation numbers to every element on both sides and look for any changes. If an atom’s oxidation number increases, it’s oxidized (lost electrons); if it decreases, it’s reduced (gained electrons). Any net change in oxidation numbers = electron transfer → a redox reaction. Quick tips: metals that go from 0 to a positive number are oxidized (e.g., Zn → Zn2+); oxygen is usually −2 and hydrogen +1 (use CED rules). Combustion reactions are a common redox subclass (reacting with O2 to give CO2 and H2O). Don’t confuse this with acid–base: those transfer H+ (protons), not electrons. For practice, walk through a few balanced equations assigning oxidation numbers (see Topic 4.7 study guide: https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz). Want more practice problems? Fiveable has a big set here: https://library.fiveable.me/practice/ap-chemistry.
I missed lab today - how do precipitation reactions actually look when you do them in real life?
In the lab a precipitation reaction usually looks like instant cloudiness where two clear solutions meet, then a solid (precipitate) forms as tiny particles. Example: mix AgNO3(aq) and NaCl(aq)—both clear—and you’ll see a white, grainy AgCl(s) precipitate almost immediately. Color varies by compound: AgI is yellow, PbS is brown/black, many sulfate/carbonate precipitates are white. Sometimes the solid settles to the bottom (you can filter it), sometimes it stays suspended as a milky/colloidal mix. Rate depends on solubility—very insoluble salts form instantly; sparingly soluble ones form more slowly. On the AP side, treat these as ionic mixes: write the molecular, complete ionic, and net ionic equations (e.g., Ag+ + Cl– → AgCl(s)), and identify spectator ions. Remember the CED solubility hint: Na+, K+, NH4+, and NO3– salts are soluble (so those are usually spectators). For a quick refresher and practice (including writing net ionic equations), check the Topic 4.7 study guide (https://library.fiveable.me/ap-chemistry/unit-4/types-chemical-reactions/study-guide/0VTaPH2MhqYc3Azc3xJz) and AP Chem practice problems (https://library.fiveable.me/practice/ap-chemistry).