Quantum Numbers and Electron Configuration
Quantum numbers and electron configurations describe how electrons are arranged within atoms. They specify each electron's energy level, orbital shape, and orientation, which in turn determine an atom's chemical behavior, bonding, and placement on the periodic table.
The rules governing electron configurations (the Pauli exclusion principle, Hund's rule, and the Aufbau principle) explain how electrons fill orbitals in a predictable way. Mastering these rules lets you predict an element's reactivity, its magnetic properties, and the patterns that organize the periodic table.
Quantum Numbers for Electron States
Defining the Four Quantum Numbers
Every electron in an atom is described by four quantum numbers. Together, they act like an address system: no two electrons in the same atom can share all four values.
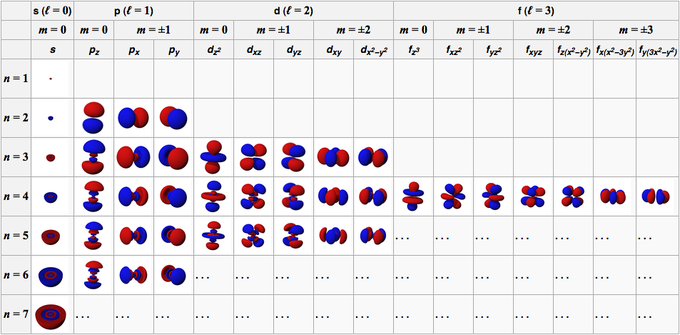
- Principal quantum number () represents the main energy level (shell).
- Determines overall energy and average distance from the nucleus
- Takes positive integer values:
- Higher means higher energy and a larger orbital
- Azimuthal (angular momentum) quantum number () describes the subshell and orbital shape.
- Values range from to
- Each value maps to a letter: , , ,
- The shape matters because it affects how electrons in that subshell interact with other atoms
- Magnetic quantum number () specifies the orbital's orientation in space.
- Ranges from to in integer steps
- This gives the number of orbitals per subshell: 1 for s, 3 for p, 5 for d, 7 for f
- Spin quantum number () represents the electron's intrinsic angular momentum.
- Only two possible values: (spin-up) or (spin-down)
- Two electrons sharing an orbital must have opposite spins
Significance and Application of Quantum Numbers
These four numbers uniquely define the state of each electron in an atom. That uniqueness is enforced by the Pauli exclusion principle: no two electrons can share the same set of all four quantum numbers.
The theoretical foundation comes from the Schrödinger equation. Its solutions yield wavefunctions (orbitals), and each wavefunction corresponds to a specific combination of , , and . The probability distributions of these wavefunctions tell you where an electron is most likely to be found.
Quantum numbers also explain atomic spectra. When an electron transitions between states with different quantum numbers, it absorbs or emits a photon whose energy equals the difference between those states.
Electron Configuration Rules
Pauli Exclusion Principle
The Pauli exclusion principle states that no two electrons in an atom can have the same set of four quantum numbers. The practical consequence: each orbital holds at most two electrons, and those two must have opposite spins ( and ).
This sets the maximum electron capacity of each subshell:
- s subshell: 1 orbital × 2 = 2 electrons
- p subshell: 3 orbitals × 2 = 6 electrons
- d subshell: 5 orbitals × 2 = 10 electrons
- f subshell: 7 orbitals × 2 = 14 electrons
These capacities directly shape periodic trends in atomic radius, ionization energy, and other properties, because they determine how many electrons fit in each shell before a new one begins filling.
Hund's Rule and Orbital Filling
Hund's rule says that when filling degenerate orbitals (orbitals with the same energy within a subshell), electrons occupy separate orbitals with parallel spins before any pairing occurs. This maximizes total spin and produces a more stable configuration.
Consider carbon () with ground state configuration . The two 2p electrons each go into a separate 2p orbital with the same spin direction, rather than doubling up in one orbital.
The Aufbau principle governs the order in which orbitals fill: lower-energy orbitals are filled first. The standard filling sequence is:
A useful mnemonic is the diagonal rule: write the subshells in rows (1s / 2s 2p / 3s 3p 3d / …) and read along diagonals from upper-right to lower-left.
Exceptions arise in some transition metals because half-filled or fully filled d subshells carry extra stability. For example:
- Chromium (Cr): instead of the expected
- Copper (Cu): instead of
In both cases, shifting one electron from 4s into 3d achieves a half-filled or fully filled d subshell, which lowers the overall energy.
Ground vs Excited State Configurations
Ground State Configurations
The ground state is the lowest-energy arrangement of electrons in an atom or ion. You find it by applying all three rules together: Aufbau principle, Pauli exclusion principle, and Hund's rule.
Valence electrons (those in the outermost shell) are the most important for chemical behavior. For sodium (Na, ), the ground state is . That single 3s valence electron explains why sodium so readily loses one electron to form .
Noble gas shorthand makes long configurations easier to write. You replace the inner-shell electrons with the symbol of the preceding noble gas in brackets. Chlorine (Cl), for instance, becomes instead of .
Anomalous ground state configurations (like Cr and Cu above) occur when the added stability of a half-filled or fully filled d subshell outweighs the normal filling order.

Excited State Configurations
An excited state occurs when one or more electrons occupy higher-energy orbitals than the ground state prescribes. This happens when the atom absorbs energy (from a photon, heat, or an electric discharge).
Excited states are temporary. For sodium, an excited state could look like , where the valence electron has been promoted from 3s to 3p. When that electron drops back down, the atom emits a photon. Sodium's characteristic yellow-orange flame color comes from exactly this transition.
The same principle is at work in neon signs: electrical energy excites neon atoms, and the specific wavelengths emitted as electrons relax produce the red-orange glow.
Multiple excited states are possible for any atom, depending on how much energy is absorbed and which transitions are allowed by selection rules. Hydrogen's Balmer series, for example, consists of visible-light emissions from electrons falling to from higher levels.
Electron Configuration and the Periodic Table
Periodic Table Organization
The periodic table is organized around electron configurations. Elements in the same group share similar valence electron arrangements, which is why they have similar chemical properties. Each new period corresponds to filling a new principal energy level.
- s-block (groups 1 and 2): valence electrons in s orbitals
- Alkali metals (group 1): configuration
- Alkaline earth metals (group 2): configuration
- p-block (groups 13–18): valence electrons in p orbitals
- Halogens (group 17) have , one electron short of a filled shell
- Noble gases (group 18) have completely filled s and p subshells, making them largely unreactive
- d-block (transition metals): partially filled d orbitals
- This gives rise to variable oxidation states and colored compounds
- Example: Iron (Fe) is , with a partially filled 3d subshell
- f-block (lanthanides and actinides): partially filled f orbitals
- Example: Cerium (Ce) is
Periodicity and Electron Configuration Trends
Atomic properties follow periodic trends that trace directly back to electron configuration:
- Atomic radius generally decreases across a period (increasing nuclear charge pulls electrons closer) and increases down a group (new shells are added farther from the nucleus).
- Ionization energy generally increases across a period and decreases down a group. Removing an electron gets harder when the nucleus holds the remaining electrons more tightly.
- Electronegativity follows a similar pattern: it increases across a period and decreases down a group. Halogens, which are one electron short of a full octet, have the highest electronegativities.
The octet rule says atoms tend to gain, lose, or share electrons to achieve a noble gas configuration. Exceptions are common for elements with accessible d or f orbitals, which can expand beyond eight valence electrons.
Magnetic properties also connect to electron configuration:
- Paramagnetic materials have one or more unpaired electrons and are attracted to external magnetic fields (e.g., oxygen with two unpaired electrons in its orbitals).
- Diamagnetic materials have all electrons paired and are weakly repelled by magnetic fields (e.g., helium with configuration ).